Abstract
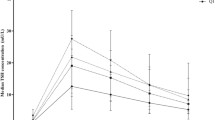
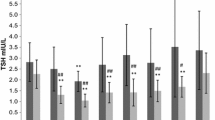
This study was performed on 36 obese subjects aged 8.5–17.4 yr, 14 boys and 22 girls (prepubertal: 5 boys and 5 girls [stage I, according to Tanner]; BMI: 35.5 ± 1.4 [mean ± SEM] and 35 ± 1.3 respectively; pubertal: 9 boys and 17 girls [stage IV–V]; BMI: 36.2 ± 1.8 and 36 ± 1.5 respectively) before and after 8 weeks of a 1000 kCal/day diet. The responses of serum TSH and PRL to TRH (200 μg iv as a bolus) were evaluated as Area Under the Curve (AUC) and net increase in respect to basal values (Δ TSH and Δ PRL). Serum T4, fT4 and rT3 were assayed at the baseline and T3 and fT3 at the baseline and 120’ after TRH injection. A similar analysis was performed on 14 age- and sex-matched lean subjects as controls. In females at baseline fT4 serum levels were greater than controls and were significantly reduced after weight loss; rT3 increased after weight loss in the whole study group. In patients of both sexes the PRL peak after TRH injection was earlier but not greater (15’) than in controls (30’). After weight loss PRL peak after TRH was found at 30’ (as controls) in females only. Taking into consideration the stage of pubertal development, the results were the following: a) in puberal girls, after weight loss, TSH and PRL peaks after TRH were delayed with respect to baseline and to the other considered subgroups; b) in prepubertal girls TSH and PRL peaks, Δ TSH, Δ PRL, AUC-TSH and AUC-PRL were blunted with respect to pubertal ones; c) the other considered variables were unchanged after the period of caloric deprivation. No correlation between BMI and the AUC of TSH and PRL was found. These data suggest that thyroid function is substantially normal in adolescent obese subjects and not influenced by a prolonged period of caloric restriction, even though a reduced hypothalamic dopaminergic tone on pituitary thyreotrophs and lactotrophs could cause subtle alterations on TSH and PRL release, partially influenced by gender and sexual development.
Similar content being viewed by others
References
Faggiano M., Carella C., Crisciuolo T., Jaquet P., Minozzi M. Massive obesity and thyroid function. In: Vague J., Boyer J. (Eds.), The regulation of adipose tissue mass. Excerpta Medica, Amsterdam, 1974, p. 295.
Martino E., Franchi F., Pinchera A., Romaldini J.H., Biagioni P., Loi A.M., Baschieri L. Effects of thyrotropin-releasing hormone and thyroxine levels in obesity. In: Vague J., Boyer J. (Eds.), The regulation of the adipose tissue mass. Excerpta Medica, Amsterdam, 1974, p. 291.
Duntas L., Hauner H., Rosenthal J., Pfeiffer E.F. Thyrotropin releasing hormone (TRH) immunoreactivity and thyroid function in obesity. Int. J. Obesity 15: 83, 1991.
Ford M.J., Cameron W.A., Ratcliffe D.B., Horn D.B., Toft A.D., Munro J.F. TSH response to TRH in substantial obesity. Int. J. Obesity 4: 121, 1980.
Donders S.H., Pieters G.F., Heerel J.G., Ross A.G., Smals H., Kloppenborg P.W.C. Disparity to thyrotropin (TSH) and prolactin responses to TSH-releasing hormone in obesity. J. Clin. Endocrinol. Metab. 61: 59, 1985.
Scaglione R., Averna M.R., Dichiara M.A., Barbagallo C.M., Mazzola G., Montalto G., Licata G., Notarbartolo A. Thyroid function and release of thyroid-stimulating hormone (TSH) and prolactin from the pituitary in human obesity. J. Int. Med. Res. 19: 389, 1991.
Amatruda J.M., Hochstein M., Hsu T.H., Lockwood D.M. Hypothalamic and pituitary dysfunction in obese males. Int. J. Obesity 6: 183, 1982.
Catania A., Cantalamessa L., Venegoni E., Orsatti A., Baldini M., Zanussi C. Evidence for differing dopaminergic activity in childhood- or adult-onset obesity. Clin. Endocrinol. (Oxf.) 22: 75, 1985.
Cavagnini F., Maraschini C., Pinto M., Dubini A., Polli E. Impaired prolactin secretion in obese patients. J. Endocrinol. Invest. 4: 149, 1981.
Wilcox R.G. Triiodothyronine, TSH and prolactin in obese women. Lancet 1: 1027, 1977.
Sannia A., Benna G.M. Prolactin response to stimulation in obesity. Horm. Metab. Res. 15: 411, 1983.
Atterrato A., Mascherpa G.F., Marino P., Morabito F. Studio dinamico della prolattinemia in obese adulte prima e dopo riduzione ponderale. Clin. Dietol. 12: 487, 1985.
Kopelman P.G., Pilkington T.R.E., White N., Jeffcoate S.L. Impaired hypothalamic control of prolactin secretion in massive obesity. Lancet. 1: 747, 1979.
Kopelman P.G., Pilkington T.R.E., Jeffcoate S.L., White N. Persistence of defective hypothalamic control of prolactin secretion in some obese women after weight reduction. Br. Med. J. 281: 358, 1980.
Cacciari E., Trejaville E., Balsamo A., Cicognani A., Pirazzoli P., Bernardi F., Zappulla F. Disordered prolactin secretion in the obese child and adolescent. Arch. Dis. Child. 56: 386, 1981.
Grimaldi F., Mazzolini A., Paterniti-Barbino R., Torossi I., Proto G., Bertolissi F. Alterata secrezione della prolattina nella obesità. Min. Endocrinol. 15: 267, 1990.
Croxson M.S., Hall T.D., Kletzky O.A., Jaramillo J.E., Nicoloff J.T. Decreased serum thyrotropin induced by fasting. J. Clin. Endocrinol. Metab. 45: 560, 1977.
Carlson M.E., Drenick E.J., Chopra I.J., Hershman J.M. Alterations in basal and TRH-stimulated serum levels of thyrotropin, prolactin and thyroid hormones in starved obese men. J. Clin. Endocrinol. Metab. 45: 707, 1977.
Azizi F. Effect of dietary composition on fasting-induced changes in serum thyroid hormones and thyrotropin. Metabolism 27: 935, 1978.
Burman K.D., Dimond R.C., Harvey G.S. Glucose modulation of alterations in serum iodothyronine concentrations induced by fasting. Metabolism 28: 291, 1979.
Burman K.D., Smallridge R.C., Osburne R. Nature of suppressed TSH secretion during undernutrition: effect of fasting and refeeding on TSH responses to prolonged TRH infusions. Metabolism 29: 46, 1980.
Rojdmark S., Rossner S. Decreased dopaminergic control of prolactin secretion in male obesity: normalization by fasting. Metabolism 40: 191, 1991.
Danforth E., Horton Jr. E.S., O’Connell M., Sims E.A.H., Burger A.G., Ingbar S.G., Braverman L., Vagenakis A.G. Dietary induced alterations in thyroid hormone metabolism during overnutrition. J. Clin. Invest. 64: 1336, 1979.
Mathieson R.A., Walberg J.L., Gwazdauskas F.C., Hinkle D.E., Gregg J.M. The effect of varying carbohydrate content of a very-low-caloric-diet on resting metabolic rate and thyroid hormones. Metabolism 35: 394, 1986.
Matzen L.E., Kvetny J. The influence of caloric deprivation and food composition on TSH, thyroid hormones and nuclear binding of T3 in mononuclear blood cells in obese women. Metabolism 38: 555, 1989.
Portnay G.I., O’Brian J.T., Bush J., Vagenakis A.G., Azizi F., Arky R.A., Ingbar S.G., Braverman L.E. The effect of starvation on the concentration and binding of thyroxine and triiodothyronine in serum and on the response to TRH. J. Clin. Endocrinol. Metab. 39: 191, 1974.
Danforth E., Desilets E.S., Horton E.S., Sims E.A.H., Burger A.G., Braverman L.E., Vagenakis A.G., Ingbar S.G. Reciprocal changes in serum triiodothyronine (T3) and reverse T3 (rT3) induced by altering the carbohydrate content of the diet. Clin. Res. 23: 573, 1975.
Lala V.R., Ray A., Jamias P., Te D., Orteza N., Fiscina B., Noto R. Prolactin and thyroid status in prepubertal children with mild to moderate obesity. J.Am. Coll. Nutr. 7: 361, 1988.
Strata A., Ugulotti G., Contini C., Magnati G., Pugnoli C., Tirelli F., Zuliani U. Thyroid and obesity: survey of some function tests in a large obese population. Int. J. Obesity 2: 333, 1978.
Hofman G.G., Schneider G., Strohmeier E., Pickardt C.R., Krich L. Thyroid hormones in obesity: thyroid function, effects on isolated fat cells of man and therapeutical application. In: Howard A. (Ed.), Recent advances in obesity research. I. Newman, London, 1975, p. 383.
Bray G.A. The myth of diet in the management of obesity. Am. J. Clin. Nutr. 23: 114, 1970.
Schmitt T., Luqman W., McCool C., Lenz F., Ahmad U., Nolan S., Stephan T., Sunder J.H., Danowski T.S. Unresponsiveness to exogenous TSH in obesity. Int. J. Obesity 1: 185, 1977.
Chopra I.J., Huang T., Beredo A. Evidence for an inhibitor of extrathyroidal conversion to 3,5,3’-triiodothyronine in sera of patients with non-thyroidal illness. J. Clin. Endocrinol. Metab. 60: 666, 1985.
Vagenakis A.G., Burger A., Portnay G.I., Rudolph M., O’Brian J.T., Azizi F., Arky A., Nicod P., Ingbar S.M., Braverman L.E. Diversion of peripheral thyroxine metabolism from activating to inactivating pathways during complete fasting. J. Clin. Endocrinol. Metab. 41: 191, 1975.
Reinhardt W., Holtermann D., Beuker G., Olbricht T., Jaspers C., Reinwein D. Effect of small doses of iodine on thyroid function during caloric restriction in normal subjects. Horm. Res. 39: 132, 1993.
Argenio G., Bernini G., Vivaldi M.S., Del Corso C., Monzani F., Baschieri L., Bertolozzi G., Santoni R., Franchi F., Luisi M. Effect of fenfluramine on prolactin and thyroid-stimulating-hormone response to thyrotropin-releasing-hormone in obese and normal women. Eur. J. Clin. Pharmacol. 39: 13, 1990.
Coiro V., Passeri M., Capretti L., Speroni G., Davoli C., Marchesi C., Rossi G., Camellini L., Volpi R., Roti E. Serotoninergic control of TSH and PRL secretion in obese men. Psychoneuroendocrinology 15: 261, 1990.
Kopelman P.G. Neuroendocrine function in obesity. Clin. Endocrinol. (Oxf.) 28: 675, 1988.
Jung R.T., Campbell R.G., James W.P.T., Callingham B.A. Altered hypothalamic sympathetic responses to hypoglycaemia in familial obesity. Lancet 1: 1043, 1982.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Guzzaloni, G., Grugni, G., Moro, D. et al. Thyroid-stimulating hormone and prolactin responses to thyrotropin-releasing hormone in juvenile obesity before and after hypocaloric diet. J Endocrinol Invest 18, 621–629 (1995). https://doi.org/10.1007/BF03349780
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03349780