Abstract
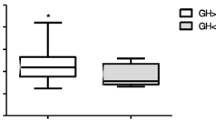
We investigated the melatonin (MT) circadian rhythm before and after somatostatin (octreotide) acute administration in ten subjects (4 M, 6 F; 23–52 yr old) with active acromegaly due to pituitary microadenoma. Blood samples were drawn every 2 hours over a 48-h span; after 24-h basal blood collection, octreotide (Sandostatin, Sandoz) 100 μg sc/8 h was administered. As control, 7 healthy adult subjects (3M, 4F; 26–50 yr old) were studied in basal condition over a 24-h span. Plasma MT and GH levels were measured by RIA in each sample, IGF-1 levels were measured by immunoradiometric assay in basal and after octreotide morning samples. The comparisons were made by Mann-U-Withney and Wilcoxon test as appropriate; the existence of a MT circadian rhythm was validated by cosinor analysis; GH and MT values were correlated by Pearson’s correlation coefficient. All of 7 control subjects and 2 of 10 acromegalics had significant 24-h MT rhythm. The area under curve (AUC), mesor and amplitude of the MT rhythms in acromegalics were significantly lower than in the controls (p< 0.001, 0.002 and 0.0006, respectively), with an earlier acrophase (median value: 22:14 vs 02:08 h of controls). Basal plasma IGF-1 levels and circadian GH concentrations were significantly increased in acromegalics in comparison with the control group. Octreotide administration significantly reduced GH, restoring a circadian MT rhythm in 5 of 10 acromegalics, with MT mean mesor and AUC not different from controls. Mean amplitude still remained lower than controls (p<:0.0006), with an earlier acrophase (median 00:01 h). No significant correlation was found between individual GH and MT levels. Our data indicate a reduction of MT circadian secretion in acromegaly, due especially to a blunted nocturnal increase with earlier MT peak; moreover, acute octreotide administration increase MT levels without modifying amplitude and phase of night-time secretion significantly. These findings suggest a negative interrelationship between GH and MT secretions or a facilitatory influence of somatostatin on daytime MT release only. This partial recovery of pineal secretion after octreotide in acromegalics could be a clinically significant contribution to improve their quality of life, considering that MT is involved in the regulation of several important functions
Similar content being viewed by others
References
Dieguez C, Page M.D., Scanlon M.F. Growth hormone neuroregulation and its alterations in diseases states. Clin. Endocrinol. (Oxf.) 28: 109, 1989.
Reiter R.J. The pineal gland. In: De Groot L.J. (Ed.) Endocrinology. W.B. Saunders, Philadelphia, 1989, p. 240.
Webb S.M., Puig-Domingo M. Role of melatonin in health and disease. Clin. Endocrinol. (Oxf.) 42: 221, 1995.
Smythe G.A., Lazarus L. Growth hormone responses to melatonin in man. Nature 184: 1373, 1974.
Valcavi R., Dieguez C, Azzarito C, Edwards C.A., Dotti C, Page M.D., Portioli I., Scanlon M.F. Effect of oral administration of melatonin on GH responses to GRF 1–44 in normal subjects. Clin. Endocrinol. (Oxf.) 26: 453, 1987.
Valcavi R., Zini M., Maestroni G.J., Conti A., Portioli I. Melatonin stimulates growth hormone secretion through pathways other than the growth hormone-releasing hormone. Clin. Endocrinol. (Oxf.) 39: 193, 1993.
Smythe G.A., Lazarus L. Suppression of human growth hormone secretion by melatonin and cyproheptadine. J. Clin. Invest. 54: 116, 1974.
Wright J., Aldhous M., Franey C, English J., Arendt J. The effects of exogenous melatonin on endocrine function in man. Clin. Endocrinol. (Oxf.) 24: 375, 1986.
Vaughan G.M., Allen J.P., Tullis W., Siler-Khodr T.M., De La Pena A., Sackman J.W. Overnight plasma profiles of melatonin and certain adenohypophyseal hormones in men. J. Clin. Endocrinol. Metab. 47: 566, 1978.
Rao M.L., Mager T. Influence of the pineal gland on pituitary function in humans. Psychoneuroendocrinology 12: 141, 1987
Van-Coevorden A., Meckel J., Laurent E., Kerkhofs M., L’Hermite-Baleriaux M., Decoster C., Neve P., Van-Cauter E. Neuroendocrine rhythms and sleep in aging men. Am. J. Physiol. 260: 651, 1991.
Grugni G., Maestroni G.J.M., Lissoni P., Barbaglia M., Moroni G., Mazzilli G., Morabito F. Study of the secretion of melatonin in idiopathic growth hormone deficiency (GHD). J. Endocrinol. Invest. 13: (Suppl. 3), 34, 1990.
Terzolo M., Piovesan A., Alt A., Codegone A., Pia A., Reimende G., Torta M, Paccotti P., Borretta G., Angeli A. Circadian profile of serum melatonin in patients with Cushing’s syndrome or acromegaly. J. Endocrinol. Invest. 18: 17, 1995.
Nelson W., Tong Y.L, Lee J-K., Halberg R Methods for cosinor-rhythmometry. Chronobiology 6: 305, 1979.
Reiter R.J. Pineal melatonin: cell biology of its synthesis and of its physiological interactions. Endocr. Rev. 12: 151, 1991.
Rosenthal N.E. Editorial: Plasma melatonin as a measure of the human clock. J. Endocrinol. Metab. 73: 225, 1991.
Weaver D.R., Stehele J.H., Stopa E.G., Reppert S. Melatonin receptors in human hypothalamus and pituitary: implications for circadian and reproductive responses to melatonin. J. Clin. Endocrinol. Metab. 76: 295, 1993.
Smythe G.A., Lazarus, L. Growth hormone regulation by melatonin and serotonin. Nature 244: 230, 1973.
Bellastella A., Colucci CF., D’Alessandro B., Lo Cicero M.L. L Dopa stimulated growth hormone release in the blind. J. Clin. Endocrinol. Metab. 44: 194, 1977.
Bellastella A., Parlato F., Sinisi A.A. Blindness impair plasma growth hormone response to L-Dopa but not to Arginine. J. Clin. Endocrinol. Metab. 70: 856, 1990.
Bellastella A., Sinisi A.A., Raiola C, Perrone L., Lorio S., Parlato F., Mazzucca A., Faggiano M. Blindness influences the growth of institutionalized prepubertal subjects. J. Endocrinol. Invest. 12: 605, 1989.
Bellastella A., Criscuolo T., Lorio S., Parlato F., Sinisi A.A., Sinisi A.M., Pasquali D., Pisano G. Normal plasma insulin-like growth factor I levels and impaired final stature in adult blind subjects. J. Endocrinol. Invest. 17: 37, 1994.
Smith J.A., O’Hara J., Schiff A.A. Altered diurnal serum melatonin rhythm in blind men (letter). Lancet 2: 933, 1981.
Lewy A.J., Newsome D.A. Different types of melatonin circadian secretory rhythm in some blind subjects. J. Clin. Endocrinol. Metab. 56: 1103, 1983.
Sack R.L, Lewy A.J., Blood M.L, Keith D.L, Nagakawa H. Circadian rhythm abnormalities in totally blind people: incidence and clinical significance. J. Clin. Endocrinol. Metab. 75: 127, 1992.
Bellastella A., Sinisi A.A., Criscuolo T., De Bellis A., Carella C, Lorio S., Sinisi A.M., Parlato F., Venditto T., Pisano G. Melatonin and the pituitary-thyroid axis status in blind adults: a possible resetting after puberty. Clin. Endocrinol. (Oxf.) 45: 707, 1995.
Richardson S.B., Hollander C.S., D’Eletto R., Greenleaf P.W., Thaw C. Acetylcholine inhibits the release of somatostatin from rat hypothalamus in vitro. Endocrinology 107: 122, 1980.
Martin J.M., Sattler C. Selectivity of melatonin pituitary inhibition for luteinizing hormone-releasing hormone. Neuroendocrinology 34: 112, 1982.
Reiter R.J. Functional aspects of the pineal hormone melatonin in combating cell and tissue damage induced by free radicals. Europ. J. Endocrinol. 134: 412, 1996.
Reppert S.M., Weaver D.R. Melatonin Madness. Cell 83: 1059, 1995.
Bengtsson B.-A. Acromegaly and neoplasia. J. Pediatr. Endocrinol. 6: 73, 1993.
Author information
Authors and Affiliations
Additional information
This work was supported in part by a grant from Ministero della Ricerca Scientifica e Tecnologica e dell’Universita (MURST 60%, Rome, Italy)
This work was presented in part at XXV Congresso Nazionale Società Italiana di Endocrinologia, Rome, May 22–25, 1994.
Rights and permissions
About this article
Cite this article
Sinisi, A.A., Pasquali, D., D’Apuzzo, A. et al. Twenty-four hour melatonin pattern in acromegaly: Effect of acute octreotide administration. J Endocrinol Invest 20, 128–133 (1997). https://doi.org/10.1007/BF03346890
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03346890