Abstract
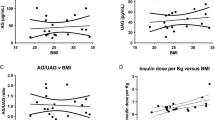
Ghrelin, the endogenous ligand for the GH secretagogue-receptor (GHS-R), in addition to its GH-releasing action, has orexigenic and adipogenic properties. These characteristics make ghrelin a potential hormone involved in the pathogenesis of obesity. Ghrelin levels are decreased in obese humans and it is unknown whether this decrease is responsible for the blunted GH secretion reported in visceral obesity. Since only few data are available on the potential feedback regulation by GH on systemic ghrelin concentrations, it remains to be established whether the correction of circulating GH concentrations in obese individuals affects ghrelin concentrations. To answer this question, we measured plasma ghrelin levels after a week of administration of low doses of recombinant human GH (rhGH) in a randomized, double-blind, placebo (PL)-controlled trial. This study was originally designed to evaluate the effects of GH replacement on lipid kinetics in visceral obese men. Six adult men with abdominal/visceral obesity (age 42±3 yr, body weight 107±10 kg, BMI 33±1 kg/m2, waist circumference 111±3 cm, mean±SE) were evaluated in the basal state (BS) and after one week of treatment with subcutaneous bedtime injections of either PL, 2.5 (GH2.5) or 3.3 (GH3.3) μg/kg/die of rhGH. In comparison to BS either PL, GH2.5 or GH3.3 did not significantly modify circulating ghrelin concentrations (p=0.77). In contrast, a significant increase of serum GH (p=0.0028), IGF-I (p=0.0033) and whole body rate of lipolysis (p=0.038, GH2.5; p=0.009, GH3.3) occurred, in comparison to BS or PL, after GH2.5 and GH3.3, without differences between the two treatments. These data demonstrate that in abdominal/visceral obese men a short-term treatment with very low doses of rhGH replacement, sufficient to augment the rate of lipolysis, do not modify circulating ghrelin levels.
Similar content being viewed by others
References
Bouchard C. Etiology of overweight and obesity. In: Bouchard C. (Ed.), Physical activity and obesity. Human Kinetics Publishers, Champaign, IL, 2000, pp. 7–10.
Arvat E., Maccario M., Di Vito L. et al. Endocrine activities of ghrelin, a natural growth hormone secretagogue (GHS), in humans: comparison and interactions with hexarelin, a non natural peptidyl GHS, and GH-relelasing hormone. J. Clin. Endocrinol. Metab. 2001, 86: 1169–1174.
Kojima M., Hosoda H., Date Y. et al. Ghrelin is a growthhormone releasing acylated from the stomach. Nature 1999, 402: 656–660.
Nakazato M., Murakami N., Date Y. et al. A role of ghrelin in the central regulation of feeding. Nature 2001, 409: 194–198.
Tschöp M., Weyer C., Tataranni A. et al. Circulating Ghrelin levels are decreased in human obesity. Diabetes 2001, 50: 707–709.
Shiiya T., Masamitsu N., Masanari M. et al. Plasma ghrelin levels in lean and obese humans and the effect of glucose on ghrelin secretion. J. Clin. Endocrinol. Metab. 2002, 87: 240–244.
Maccario M., Grottoli S., Procopio M. et al. The GH/IGFI axis in obesity: influence of neuroendocrine and metabolic factors. Int. J. Obes. Relat. Metab. Disord. 2000, Suppl. 2: 96–99.
De Boer H., Blok G.J., Van Der Veen E.A. Clinical aspects of growth hormone deficiency in adults. Endocr. Rev. 1995, 16: 63–86.
Johannsson G., Mårin P., Lönn L. et al. Growth hormone treatment of abdominally obese men reduces abdominal fat mass, improves glucose and lipoprotein metabolism, and reduces diastolic blood pressure. J. Clin. Endocrinol. Metab. 1997, 82: 727–734.
Lucidi P., Parlanti N., Piccioni F. et al. Short-term treatment with low doses of recombinant human GH stimulates lipolysis in visceral obese men. J. Clin. Endocrinol. Metab. 2002, 87: 3105–3109.
McGuire E., Helderman J., Tobin J., Andres R., Berman M. Effects of arterial venous sampling on analysis of glucose kinetics in man. J. Appl. Physiol. 1976, 41: 565–573.
Janssen J.A.M.J.L., van der Toorn F.M., Hoffland L.J. et al. Systemic ghrelin levels in subjects with growth hormone deficiency are not modified by one year of growth hormone replacement therapy. Eur. J. Endocrinol. 2001, 145: 711–716.
Matthews D.R., Hosker J.P., Rudenski A.S. et al. Homeostasis model assessment: insulin resistance and β-cell function from fasting glucose and insulin concentrations in man. Diabetologia 1985, 28: 412–419.
Wolfe R.R. Glycerol flux. In: Wolfe R.R. (Ed.), Radioactive and stable isotope tracers in biomedicine. Wiley-Liss Inc, New York, 1992, p. 325.
Wolfe R.R. Appendix A: lab methods. In: Wolfe R.R. (Ed.), Radioactive and stable isotope tracers in biomedicine. Wiley-Liss Inc, New York, 1992, pp. 427.
Horber F.F., Kahl J., Lecavalier L. et al. Determination of leucine and a-ketoisocaproic acid concentrations and specific activity in plasma and leucine specific activities in proteins using high-performance liquid chromatography. J. Chromatogr. 1989, 495: 81–94.
De Feo P., Gallai V., Mazzotta G. et al. Modest decrements in plasma glucose concentration cause early impairment in cognitive function and later activation of glucose counterregulation in the absence of hypoglycemic symptoms in normal man. J. Clin. Invest. 1988, 82: 436–444.
Wright J.C., Bartke A. Ghrelin administration decreases insulin secretion in Ames dwarf mice. 83rd Annual Meeting of Endocrine Society, Denver, USA, 2001, (Abstract) p.1, p.98.
van der Toorn F.M., Janssen J.A.M.J.L., de Herder W.W., et al. Central ghrelin production does not substantially contribute to systemic ghrelin concentrations; a study in two subjects with active acromegaly Europ. J. Endocrinol. 2002, 147:195–199.
Hansen T.K., Dall L., Hosoda H. et al. Weight loss increases circulating levels of ghrelin in human obesity. Clin. Endocrinol. (Oxf.) 2002, 56: 203–206.
Takaya K., Ariyasu H., Kanamoto N. et al. Ghrelin strongly stimulates growth hormone release in humans. J. Clin. Endocrinol. Metab. 2000, 85: 4908–4911.
Tassone F., Broglio F., Gottero C. et al. Effetti metabolici ed ormonali della somministrazione acuta di ghrelin in pazienti obesi. I Congresso Nazionale Società Italiana Obesità, Verona, Italy, 2002, pp. 78–198 (Abstract)
Cummings D.E., Purnell J.Q., Frayo R.S. et al. A preprandial rise in plasma ghrelin levels suggest a role in meal initiation in humans. Diabetes 2001, 50: 1714–1719.
Lucidi P., Murdolo G., Di Loreto C. et al. Ghrelin is not necessary for adequate hormonal counterregulation to insulininduced hypoglycemia. Diabetes 2002, 51: 2911–2914.
Broglio F., Arvat E., Benso A. et al. Ghrelin, a natural GH secretagogue produced by the stomach, induces hyperglycemia and reduces insulin secretion in humans. J. Clin. Endocrinol. Metab. 2001, 86: 5083–5086.
Date Y., Kojima M., Hosoda H. et al. Ghrelin, a novel growth hormone-releasing acylated peptide, is synthesized in a distinct endocrine cell-type in the gastrointestinal tracts of rats and humans. Endocrinology 2000, 141: 4255–4261.
Toshinai K., Mondal M.S., Nakazato M. et al. Upregulation of Ghrelin expression in the stomach upon fasting, insulin-induced hypoglycemia, and leptin administration. Biochem. Biophys. Res. Commun. 2001, 281: 1220–1225.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Murdolo, G., Lucidi, P., Di Loreto, C. et al. Circulating ghrelin levels of visceral obese men are not modified by a short-term treatment with very low doses of GH replacement. J Endocrinol Invest 26, 244–249 (2003). https://doi.org/10.1007/BF03345164
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03345164