Abstract
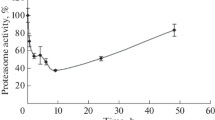
A single intraperitoneal injection of the human therapeutic drug bleomycin (BL) was administered to three groups of male Fischer 344 rats at time 0, and the incorporation of [35S]methionine (“synthesis”) and phosphorylation patterns of stress proteins (sps/hsps) from bone marrow cells were analyzed over time by two- dimensional electrophoresis and fluorography. Two groups of rats, young ad libitum (Y/AL — 3 months) and old ad libitum (O/AL — 28 months), had free access to rat chow, and a third group of old rats (O/CR — 28 months) were maintained on a caloric restricted intake (60% of the AL diet). The administration of BL in Y/AL, O/AL and O/CR animals activated the 35S- labeling of sp 90 which reached a peak at 4 hours. Labeling of sp 90 was significantly greater in Y/AL compared to O/AL, and the incorporation pattern of O/CR was intermediate to Y/AL and O/AL animals. All labeling of sp 90 in each group had disappeared by 10 hours after BL administration. Stress protein 70x (inducible form) in these three animal groups displayed a similar pattern of 35Sincorporation, but the amount of labeling was less than that of sp 90. No labeling of sp 70x remained by 13 hours after BL administration. Phosphorylation ([32P] phosphate incorporation) of sp 90 reached a maximum level at 2 hours in all animals, and 32P- labeling in Y/AL was significantly increased over O/AL and O/CR with an intermediate level found in O/CR animals. The turnover rate (phosphorylation/dephosphorylation) of sp 90 induced by BL was significantly suppressed and temporarily extended in O/AL as compared with O/CR, which implied that CR not only increased incorporation of sp 90, but also enhanced a utilization of the phosphate pool very similar to that seen in Y/AL animals. (Aging Clin. Exp. Res. 6: 121- 131, 1994)
Similar content being viewed by others
References
McCay C.M., Crowell M.F., Maynard L.A: Effect of retarded growth upon the length of life span and upon the ultimate body size. J. Nutr. 10: 63–70, 1935.
Masoro E.J.: Nutrition and aging — a current assessment. J. Nutr. 115: 842–848, 1985.
Pollard M., Luckert P.H.: Tumorigenic effects of direct-and indirect-acting chemical carcinogens in rats on a restricted diet. J. Natl. Cancer Inst. 74: 1346–1349, 1985.
Ruggeri B.A., Klurfeld D.M., Kritchevesky D.: Biochemical alterations in 7, 12-dimethylbenzlanthracene-induced mammary tumors from rats subjected to caloric restriction. Biochim. Biophys. Acta 929: 239–246, 1987.
Walford R.L.: Maximum life span. Norton, New York, 1983.
Pipkin J.L., Hinson W.G., Lyn-Cook L.E., Feuers R.J., Leakey J.E.A., Hart R.W., Casciano D.A.: The high mobility group of nuclear protein’s as biomarkers of age and caloric restriction in rats. Biochem. Int. 26: 953–961, 1992.
Pipkin J.L., Hinson W.G., Lyn-Cook L.E., Feuers R.J., Burns E.R., Hart R.W., Casciano D.A.: Histone variants, H1 subtypes and other chromatin proteins as biomarkers of age and caloric restriction in rats. Age 16: 97–103, 1993.
Richardson A., Butler J.A., Rutherford M.S., Sensei I., Gu M., Fernandes G., Chiang W.: Effect of age and dietary restriction on the expression of alpha 2u-globulin. J. Biol. Chem. 262: 12821–12825, 1987.
Nakamura K.D., Duffy P.H., Turturro A., Hart R.W.: The effect of dietary restriction on myc protooncogene expression in mice; a preliminary study. Mech. Ageing Dev. 48: 199–205, 1989.
Leakey J.E., Cunny H.C., Bazare J. Jr., Webb P.J., Feuers R.J., Duffy P.H., Hart R.W.: Effects of aging and caloric restriction on hepatic drug metabolizing enzymes in the Fischer 344 rat. I. The Cytochrome P-450 dependent monooxygenase system. Mech. Ageing Dev. 48: 145–155, 1989.
Leakey J.E., Cunny H.C., Bazare J. Jr., Webb P.J., Lipscomb J.C., Slikker W. Jr., Feuers R.J., Duffy P.H., Hart R.W.: Effects of aging and caloric restriction on hepatic drug metabolizing enzymes in the Fischer 344 rats. II. Effects on conjugating enzymes. Mech. Ageing Dev. 48: 157–166, 1989.
Leakey J.E., Bazare J., Harmon J.R., Feuers R.J., Duffy P.H., Hart R.W.: Effects of caloric restriction on hepatic drug metabolizing enzyme activities in the Fischer-344 rat. In: Fishbein L. (Ed.), Biological Effects of Dietary Restriction. Springer-Verlag, New York, 1991, pp. 207–216.
Lipman J.M., Turturro A., Hart R.W.: The influence of dietary restriction on DNA repair in rodents: a preliminary study. Mech. Ageing Dev. 48: 135–143, 1989.
Schlesinger M.J.M., Ashburner M., Tissieres: Heat shock from bacteria to man. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1982.
Ashburner M., Bonner J.J.: The induction of gene activity in Drosophila by heat shock. Cell 17: 241–254, 1979.
Courgeon A.M., Maisonhaute C., Best-Belpomme M.: Heat shock proteins are induced by cadmium in Drosophila cells. Exp. Cell Res. 153: 515–521, 1984.
Lawrence F., Robert-Gero M.: Induction of heat shock and stress proteins in promastigotes of three Leishmania species. Cell Biol. 82: 4414–4417, 1985.
Pipkin J.L., Anson J.F., Hinson W.G., Burns E.R., Wolff G.W.: Alterations in synthesis of the acid soluble proteins from nuclei of specific phases of the in vivo liver cell cycle after isoproterenol and sodium phŋobarbital administration: An electrophoretic study. Electrophoresis 6: 306–313, 1985.
Anson J.F., Laborde J.B., Pipkin J.L., Hinson W.G., Hansen D.K., Sheehan D.M., Young J.F.: Target tissue specificity of retinoic acid-induced stress proteins and malformations in mice. Teratology 44: 19–28, 1991.
Anson J.F., Hinson W.G., Pipkin J.L., Kwarta R.F., Hansen D.K., Young J.F., Burns E.R., Casciano DA: Retinoic acid induction of stress proteins in fetal mouse limb buds. Develop. Biol. 121: 542–547, 1987.
Buzin C.H., Bournais-Vardiabasis N.: Teratogens induce a subset of small heat shock proteins in Drosophila primary embryonic cell cultures. Proc. Natl. Acad. Sci. USA 81: 4075–4079, 1984.
Pipkin J.L., Hinson W.G., Anson J.F., Schol H.M., Lyn-Cook L.E., Burns E.R., Casciano DA: Synthesis and biochemical characteristics of nucleoproteins following a toxic dose of retinoic acid in cycling and differentiating H1-60 cells. Appl. Theor. Electrophoresis 2: 31–41, 1991.
Pipkin J.L., Anson J.F., Hinson W.G., Duffy P.H., Burns E.R., Casciano DA: The turnover of radiolabeled nuclear proteins in rats exposed to environmental and chemical stress. Toxicol. Lett. 39: 15–26, 1987.
Caltabiano M.M., Koestler T.P., Poste G., Greig R.G.: Induction of mammalian stress proteins by a triethylphosphine gold compound used in the therapy of rheumatoid arthritis. Biochem. Biophys. Res. Commun. 138: 1074–1080, 1986.
Carper S.W., Duffy J.J., Gerner E.W.: Heat shock proteins in thermotolerance and other cellular processes. Cancer Res. 47: 5249–5255, 1987.
Ang D., Liberek K., Skowyra D., Zylicz M., Georgopoulos C.: The biological role and regulation of the universally conserved heat shock proteins. J. Biol. Chem. 266: 24233–24236, 1991.
Itoh H., Tashima Y.: The stress (heat shock) protein. Int. J. Biochem. 23: 1185–1191, 1991.
Moseley P.L., York S.J., York J.: Bleomycin induces the hsp-70 heat shock promoter in cultured cells. Am. J. Resp. Cell Mol. Biol. 1: 89–93, 1989.
Hinson W.G., Pipkin J.L., Hudson J.L., Anson J.F., Tyrer H.: A micro-sample collection device for electrostatically sorted cells or particles and its preparative use for biochemical analysis. Cytometry 2: 390–394, 1982.
Pipkin J.L., Anson J.F., Hinson W.G., Burns E.R., Casciano D.A., Sheehan D.M.: Cell cycle–specific effects of sodium arsenite and hyperthermic exposure on incorporation of radioactive leucine and phosphate by stress proteins from mouse lymphoma cell nuclei. Biochem. Biophys. Acta 927: 334–344, 1987.
Herzenberg L.A., Sweet R.G., Herzenberg L.A.: Fluorescence activated cell sorting. Sci. Am. 234: 108–118, 1976.
Pipkin J.L., Anson J.F., Hinson W.G., Hudson J.L.: The effect of isoproterenol and hydroxyurea on the presence of ubiquitin and protein A24 in the rat salivary gland. Biophys. Acta 699: 155–163, 1982.
Pipkin J.L., Anson J.F., Hinson W.G., Schol H.M., Burns E.R., Casciano D.A.: Analysis of protein incorporation of radioactive isotopes in the Chinese hamster cell cycle by electronic sorting and micro-gel electrophoresis. Cytometry 7: 147–156, 1985.
Pipkin J.L., Anson J.F., Hinson W.G., Burns E.R., Casciano D.A.: Differential phosphate labeling of stress protein polypeptides in toxic dose response during Sphase of mouse lymphoma cells: A micro-electrophoretic study. Appl. Theor. Eledrophoresis 1: 77–84, 1989.
Pipkin J.L., Hinson W.G., Anson J.F., Schol H.M., Lyn-Cook L.E., Burns E.R., Casciano D.A.: Synthesis and phosphorylation of nuclear matrix proteins following a toxic dose of retinoic acid in cycling and differentiating HL-60 cells. Appl. Theor. Eledrophoresis 2: 17–29, 1991.
O’Farrell P.H.: High resolution two-dimensional electrophoresis of protein. J. Biol. Chem. 250: 4007–4021, 1975.
Hinson W.G., Pipkin J.L., Anson J.F., Burns E.R., Casciano D.A.: A micro-size polyacrylamide gel electrophoresis system. Reu. Sci. Instru. 58: 1736–1742, 1987.
Bonner W.M., Laskey R.A.: A film detection method for tritium labeled proteins and nucleic acids in polyacrylamide gels. Eur. J. Biochem. 46: 83–88, 1974.
Tahourdin C.S.M., Neihart N.K., Isenburg I., Bustin M.: Immunochemical detection of chromosomal protein HMG-17 in chromatin subunits. Biochem. J. 20: 910–915, 1981.
Towbin H., Staehelin T., Gordon J.: Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc. Natl. Acad. Sci. USA 76: 4350–4354, 1979.
Pipkin J.L., Anson J.F., Hinson W.G., Burns E.R., Casciano D.A.: Comparative immunochemical and structural similarities of five stress proteins from various tissue types. Comp. Biochem. Physiol. 89: 43–50, 1988.
Pipkin J.L., Hinson W.G., Lyn-Cook L.E., Burns E.R., Sheehan D.M., Casciano D.A.: A nuclear protein associated with lethal heat shock of HL-60 cells. Exp. Cell Res. 202: 167–173, 1992.
Pipkin J.L., Anson J.F., Hinson W.G., Schol H.M., Sheehan D.M.: Spleen cell phosphorylation and salt soluble nuclear protein from isoproterenol treated and sorted nuclei. J. Biochem. 95: 323–333, 1984.
Pipkin J.L., Anson J.F., Hinson W.G., Burns E.R., Casciano D.A.: Microscale electrophoresis of stress proteins induced by chemicals during the in vivo cell cycle. Electrophoresis 7: 463–471, 1986.
Hightower L.E.: Heat shock, stress proteins, chaperones, and proteotoxicity. Cell 66: 191–197, 1991.
Young R.A., Elliott T.J.: Stress proteins, infection, and immune surveillance. Cell 59: 5–8, 1989.
Sadis S., Raghavendra K., Hightower L.E.: Secondary structure of the mammalian 70-kilodalton heat shock cognate protein analayzed by circular dichroism spectroscopy and secondary structure prediction. Biochem. J. 29: 8199–8206, 1990.
Ellis R.J.: Molecular chaperones. Semin. Cell Biol. 1: 1–72, 1990.
Legagneux V., Morange M., Bensaude O.: Heat shock increases turnover of 90 kDa heat shock protein phosphate groups in HeLa cells. Fed. Eur. Biochem. Soc. 291: 359–362, 1991.
Ferris D.K., Harel-Bellan A., Morimoto R.I., Welch W.J., Farrar W.L.: Mitogen and lymphokine stimulation of heat shock proteins in T lymphocytes. Proc. Natl. Acad. Sci. USA 85: 3850–3854, 1988.
Freidman D.L., Ken R.: Insulin stimulates incorporation of 32P into nuclear lamins A and C in quiescent BHR-21 cells. J. Biol. Chem. 263: 1103–1106, 1988.
Gurley L.R., Walters R.A., Hildebrand C.E., Hohmany P.G., Barham S.S., Deaven L.L., Tobey R.A.: Histone phosphorylation related to chromatin structure. In: Brinkley B.R., Porter K.R. (Eds.), International Cell Biology. Rockefeller University Press, New York, 1977, pp. 420–429.
Feuers R.J., Casciano D.A.: Insulin and glucagon’s reciprocal mediation of a dual regulation mechanism for pyruvate kinase. Biochem. Biophys. Res. Commun. 124: 651–656, 1984.
Kleinsmith L.J.: Phosphorylation of nonhistone — proteins. In: Busch H. (Ed.), The Cell Nucleus. Vol. VI, Academic Press Inc., New York, 1978, pp. 221–261.
Welch W.J.: How cells respond to stress. Sci. Am. 268: 56–64, 1993.
Blake H.J., Fargnoli J., Gershon D., Holbrook N.J.: Concomitant decline in heat-induced hyperthermia and HSP70 mRNA expression in aged rats. Am. J. Physiol. 260: 663–667, 1991.
DeGuchi Y., Necoro S., Kishimoto S.: Age-related changes of heat shock protein gene transcription in human peripheral blood mononuclear cells. Biochem. Biophys. Res. Commun. 157: 580–584, 1988.
Famoli J., Kunishada T., Fornace A.J., Schneider E.L., Holbrook N.J.: Decreased expression of HSP 70 mRNA and protein after heat shock in cells of aged rats. Proc. Natl. Acad. Sci. USA 87: 846–850, 1990.
Wu B., Gu M.J., Heydari A.R., Richardson A.: The effect of age on the synthesis of two heat shock proteins in the HSP70 family. J. Gerontol. 48: 50–56, 1993.
Heydari A.R., Wu B., Takahashi R., Strong R., Richardson A.: Expression of heat shock protein 70 is altered by age and diet at the level of transcription. Mol. Cell Biol. 13: 2909–2918, 1993.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pipkin, J.L., Hinson, W.G., Feuers, R.J. et al. The temporal relationships of synthesis and phosphorylation in stress proteins 70 and 90 in aged caloric restricted rats exposed to bleomycin. Aging Clin Exp Res 6, 121–131 (1994). https://doi.org/10.1007/BF03324225
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03324225