Summary
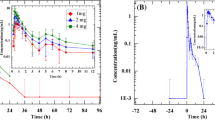
The pharmacokinetics of Cilazapril and its active metabolite cilazaprilat in plasma were investigated in an open study of 13 healthy male volunteers, aged 18 to 43 years. One capsule containing 2.5 mg Cilazapril was administered to each volunteer daily for 8 days. Plasma samples were obtained after the first and eighth doses. Concentrations of Cilazapril, cilazaprilat and activities of angiotensin converting enzyme (ACE) were measured by radioenzymatic methods. For Cilazapril, the values of apparent plasma clearance (about 15 1/h) and volume of distribution (around 28 1) were sufficiently high to suggest that significant pre-systemic hydrolysis to cilazaprilat occurred. There were no significant changes in these values after repeated dosing. There were small, but statistically significant, increases in mean peak concentrations, mean areas under concentration-time curves and mean trough concentrations from the first to the eighth dose. A steady state was achieved after eight doses with an accumulation of 20–30%. The mean effective half-life was approximately 9 h. Despite the accumulation of cilazaprilat in plasma, there were no significant differences in plasma ACE inhibition from the first to the eighth dose.
Similar content being viewed by others
References
Nussberger J., Fasanella d’Amore T., Porchet M., et al. (1987): Repeated administration of the converting enzyme inhibitor Cilazapril to normal volunteers. J. Cardiovasc. Pharmacol., 9, 39–44.
Cushman D.W., Cheung H.S. (1971): Spectrophotometric assay and properties of the angiotensin converting enzyme of rabbit lung. Biochem. Pharmacol., 20, 1637–1648.
Ryan R.W., Chung A., Ammons C., Carlton M.L. (1977): A simple radioassay for angiotensin-converting enzyme. Biochem. J., 167, 501–504.
Francis R.J., Brown A.N., Kler L., et al. (1987): Pharmacokinetics of the converting enzyme inhibitor Cilazapril in normal volunteers and the relationship to enzyme inhibition: Development of a mathematical model. J. Cardiovasc. Pharmacol., 9, 32–38.
Ryan J.W. (1983): Assay of peptidase and protease enzymes in vivo. Biochem. Pharmacol., 32, 2127–2137.
Williams P.E.O., Brown A.N., Rajaguru S., et al. (1989): Pharmacokinetics and bioavailability of Cilazapril in normal man. Br. J. Clin. Pharmacol., 27, 181S-188S.
Rosenthal E., Francis R.J., Brown A.N., et al. (1989): A pharmacokinetic study of Cilazapril in patients with congestive heart failure. Br. J. Clin. Pharmacol., 27, 267S-273S.
Till A.E. Gomez H.J., Hichens M., et al. (1984): Pharmacokinetics of repeated single oral doses of enalapril maleate (MK-421) in normal volunteers. Biopharm. Drug Dispos., 5, 273–280.
Lees K.R., Reid J.L., (1987): Age and the pharmacokinetics and pharmacodynamics of chronic enalapril treatment Clin. Pharmacol. Ther., 41, 597–602.
Beerman B., Junggren I., Cocchetto D., et al. (1985): Lisinopril steady state kinetics in healthy subjects. J. Clin. Pharmacol, 25, 471.
Rakhit A., Hurley M.E., Tipnis V., Coleman J., Rommel A., Brunner H.A. (1986): Pharmacokinetics and pharmacodynamics of pentopril a new angiotensin-converting-enzyme inhibitor in humans. J. Clin. Pharmacol., 26, 156–164.
Duchin K.L., Herman T.S., O’Leary K., Tu J., Nichola P. (1987): Steady-state (SS) kinetics of fosinopril in hypertensive patients. Clin. Pharmacol. Ther., 41, 227.
Lees K.R., Kelman A.W., Reid J.L. (1986): Effect of repeat dosing on concentration effect relationships with the angiotensin converting enzyme inhibitor Perindopril. Br. J. Clin. Pharmacol., 22, 243P.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Williams, P.E.O., Brown, A.N., Rajaguru, S. et al. Pharmacokinetics of cilazapril during repeated oral dosing in healthy young volunteers. Eur. J. Drug Metab. Pharmacokinet. 15, 63–67 (1990). https://doi.org/10.1007/BF03190129
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03190129