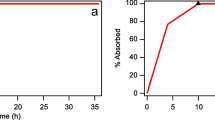
Summary
In order to assess the extent and the rate of absorption in bioavailability studies, area under the curve (AUC), experimental maximum concentration (Cmax) and experimental time to reach Cmax (Tmax), are used. But when slow-release formulations are considered, the drug concentration-time curves usually show multiple peaks, and it is difficult to compute a Cmax and Tmax value. In case a Cmax value is computed, important variability in this parameter results in high values in the residual variance of the ANOVA test. So in order to decrease the high variability, average parameters: average concentration (Cav), average maximum concentration (Cmax,av) and Cmax,av x 100/Cav (%Cmax,av), are proposed.
These new parameters were applied in a bioavailability study of slow-release amitriptyline formulation.
Similar content being viewed by others
References
Steinijans V.W. (1990): Pharmacokinetic, characterization of controlled-release formulations. Eur. J. Drug Metab. Pharmacokinet. 15: 173–181.
Bois F.Y., Tozer T.N., Hauck W.W., Chen M.L., Patnaik R., Williams R.L. (1994): Bioequivalence: performance of several measures of rate of absorption. Pharm. Res. 11: 966–974.
Wong S.H.Y., McCauley T. (1981): Reversed phase high performance liquid chromatographic analysis of tricyclic antidepressants in plasma. J. Liq. Chromatogr. 4: 849–862.
Martindale, The Extra Pharmacopoeia 29 ed., Reynolds JEF (editor). London, The Pharmaceutical Press 1989.
Schulz P., Dick P., Blaschke T.F., Hollister L. (1985): Discrepancies between pharmacokinetic studies of Amitriptyline. Clin. Pharmacokinet. 10: 257–268.
Vzquez M., Fagiolino P., Payss H., Rubio M., Aiache J.M., Mari#o E., Bioequivalence assessment from salivary data. Submitted.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fagiolino, P., Vázquez, M., Savio, E. et al. Average parameters in bioavailability studies: an application to slow-release amitriptyline formulation. European Journal of Drug Metabolism and Pharmacokinetics 23, 160–165 (1998). https://doi.org/10.1007/BF03189333
Issue Date:
DOI: https://doi.org/10.1007/BF03189333