Abstract
Aim
In order to assess the influence of total-body irradiation (TBI) on the outcome and incidence of complication after bone marrow transplantation (BMT), we retrospectively analyzed our patients treated for acute leukemia and conditioned with TBI prior to BMT.
Patients and Methods
Between 1980 and 1993, 326 patients referred to our department with acute non-lymphoblastic leukemia (ANLL, n=182) and acute lymphoblastic leukemia (ALL, n=144) in complete remission underwent TBI either in single dose (190 patients: 10 Gy administered to the midplane, and 8 Gy to the lungs [STBI]) or in 6 fractions (136 patients: 12 Gy on 3 consecutive days, and 9 Gy to the lungs [FTBI]) before BMT. The male-to-female ratio was 204/122 (1.67), and the median age was 30 years (mean: 30 ± 11, range: 3 to 63). The patients were analyzed according to 3 instantaneous dose rate groups: 118 patients in the LOW group (≤0.048 Gy/min), 188 in the MEDIUM group (> 0.048 and ≤ 0.09 Gy/min), and 20 in the HIGH group (> 0.09 cGy/min). Conditioning chemotherapy consisted of cyclophosphamide (CY) alone in 250 patients, CY and other drugs in 54, and 22 patients were conditioned using combinations without CY. Following TBI, allogeneic and autologous BMT were realized respectively in 118 and 208 patients. Median follow-up period was 68 months (mean: 67 ± 29, range: 24 to 130 months).
Results
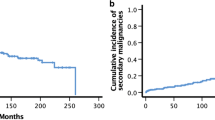
Five-year survival, LFS, RI and TRM rates were 42%, 40%, 47%, and 24%, respectively. Five-year LFS was 36% in the STBI and 45% in the FTBI group (p = 0.17). It was 36% in the LOW group, 42% in the MEDIUM group, and 30% in the HIGH group (p > 0.05). Five-year RI was 50% in STBI, 43% in FTBI, 55% in LOW, 41% in MEDIUM, and 44% in HIGH groups (STBI vs. FTBI, p = 0.48; LOW vs. MEDIUM, p = 0.03: MEDIUM vs. HIGH, p = 0.68). TRM was not influenced significantly by the different TBI techniques. When analyzing separately the influence of fractionation and the instantaneous dose rate either in ANLL or ALL patients, no difference in terms of survival and LFS was observed. Fractionation did not influence the 5-year RI both in ANLL and ALL patients. However, among the patients with ANLL, 5-year RI was significantly higher (58%) in the LOW group than the MEDIUM group (31%, p = 0.001), whereas instantaneous dose rate did not significantly influence the RI in ALL patients. The 5-year TRM rate was significantly higher in allogeneic BMT group both in ANLL (37%) and ALL (37%) patients than those treated by autologous BMT (ANLL: 15%, ALL: 18%: p = 0.002 and 0.02, respectively). The 5-year estimated interstitial pneumonitis (IP) and cataract incidence rates were 22% and 19%, respectively, in all patients. IP incidence seemed to be higher in the HIGH group (46%) than the MEDIUM (19%, p = 0.05) or LOW (25%, p = 0.15) groups. Furthermore, cataract incidence was significantly influenced by fractionation (STBI vs. FTBI, 29% vs. 9%; p = 0.003) and instantaneous dose rate (LOW vs. MEDIUM vs. HIGH, 0% vs. 27% vs. 33%; p < 0.0001). Multivariate analyses revealed that the best factors influencing the survival were 1st CR (p = 0.0007), age ≤ 40 years (p = 0.003), and BMT after 1985 (p = 0.008). The RI was influenced independently only by the remission status (p = 0.0002). On the other hand, the TRM rate was lower in patients who did not experience graft-vs.-host disease (GvHD, p < 0.0001), and in those treated after 1985 (p = 0.0005). GvHD was the only independent factor involved in the development of IP (p = 0.01). When considering the cataract incidence, the only independent factor was the instantaneous dose rate (p = 0.0008).
Conclusion
The outcome of BMT patients conditioned with TBI for acute leukemia was not significantly influenced by the TBI technique, and TRM seemed to be lower in patients treated after 1985. On the other hand, cataract incidence was significantly influenced by the instantaneous dose rate.
Zusammenfassung
Hintergrund
Um den Einfluß der Ganzkörperbestrahlung (TBI) auf die Prognose und die Inzidenz von Komplikationen bei Knochenmarktransplantationen (BMT) zu evaluieren, haben wir retrospektiv unser Patientengut, das bei akuter Leukämie vor der BMT mit der TBI behandelt wurde, analysiert.
Patienten und Methode
Von 1980 bis 1993 wurden 326 Patienten mit akuter nichtlymphatischer Leukämie (ANLL, n = 182) und akuter lymphatischer Leukämie (ALL, n = 144) in Vollremission in unserer Abteilung mit einer TBI vor einer BMT behandelt. Die TBI wurde entweder mit einer Einzeldosis (STBI; n = 190: 10 Gy L4, 8 Gy Lungen) oder in sechs Fraktionen (FTBI; n = 136: an drei aufeinanderfolgenden Tagen 12 Gy L4, 9 Gy Lungen) appliziert. Die Männer/Frauen-Ratio betrug 204/122 (1,67). und das mediane Alter betrug 30 Jahre (± 11, 3–63). Außerdem wurden die Patienten in Relation zu drei momentanen Dosisraten analysiert: 118 Patienten waren in der Gruppe mit niedriger Dosisrate (LDR; ≤ 0,048 Gy/min), 188 wurden mit mittlerer Dosisrate (MDR; > 0,048 und ≤ 0.09 Gy/min) und 20 mit einer hohen Dosisrate (HDR; > 0,09 Gy/min) bestrahlt. Die konditionierende Chemotherapie bestand aus Cyclophosphamid (CY) alleine bei 250 Patienten, CY und anderen Medikamenten bei 54 Patienten, und 22 Patienten wurden mit Kombinationen ohne CY behandelt. Nach der TBI wurden allogene und autologe BMT bei respektive 118 und 208 Patienten durchgeführt. Das mediane Follow-up betrug 68 Monate (67 ± 29,24 bis 130 Monate).
Ergebnisse
Das Fünf-Jahres-Überleben, das leukämiefreie Überleben (LFS), die Fünf-Jahres-Rezidivinzidenz (RI) und die therapiebedingte Mortalität (TRM) betrugen jeweils 42%, 40%, 47% und 24%. Das LFS betrug 36% in der STBI- und 45% in der FTBI-Gruppe (p = 0,17). Es betrug 36% in der LDR-, 42% in der MDR- und 30% in der HDR-Gruppe (p > 0,05). Die RI betrug 50% in der STBI-, 43% in der FTBI-, 55% in der LDR-, 41% in der MDR- und 44% in der HDR-Gruppe (STBI vs. FTBI, p = 0,48: LDR vs. MDR, p = 0,03; MDR vs. HDR, p = 0,68). Die TRM wurde durch die unterschiedlichen Bestrahlungstechniken nicht signifikant beeinflußt. Bei der getrennten Analyse des Einflusses der Fraktionierung und der momentanen Dosisrate bei ANLL- oder ALL-Patienten wurde kein Unterschied für das Überleben oder das LFS beobachtet. Die Fraktionierung beeinflußte die RI weder bei den ANLL- noch bei den ALL-Patienten. Jedoch war bei den ANLL-Patienten die RI signifikant höher (58%) in der LDR- als in der MDR-Gruppe (31%, p = 0,001), die momentane Dosisrate zeigte aber keinen signifikanten Einfluß auf die RI bei ALL-Patienten. Die Fünf-Jahres-TRM-Rate war in der allogenen BMT-Gruppe bei den ANLL- (37%) und ALL-Patienten (37%) signifikant höher als bei der autologen BMT-Gruppe (ANLL 15%, ALL 18%; respektive p = 0,002 und 0,02). Die Fünf-Jahres-Inzidenzraten für die interstitielle Pneumonitis (IP) und Katarakt betrugen 22% respektive 19% bei allen Patienten. Die IP-Inzidenz schien höher zu sein in der HDR- (46%) als in der MDR- (19%, p = 0,05) und LDR-Gruppe (25%, p = 0,15). Außerdem wurde die Kataraktinzidenz signifikant durch die Fraktionierung (STBI vs. FTBI, 29% vs. 9%, p = 0,003) und die momentane Dosisrate (LDR vs. MDR vs. HDR, 0% vs. 27% vs. 33%, p < 0,0001) beeinflußt. Multivarianzanalysen zeigten folgende Hauptfaktoren, die die Überlebensraten beeinflußten: erste Vollremission (p = 0,0007). Alter < 40 Jahre (p = 0,003) und BMT nach 1985 (p = 0,008). Die RI wurde unabhängig nur durch den Remissionsstatus beeinflußt. Außerdem war die TRM-Rate niedriger bei Patienten, die keine Graft-vs.-Host-Reaktion (GvHD, p < 0,0001) hatten, und bei den Patienten, die nach 1985 behandelt wurden. GvHD war der einzige unabhängige Faktor in der Entwicklung der IP (p = 0,01). Für die Kataraktinzidenz fand sich als einziger unabhängiger Faktor die momentane Dosisrate (p = 0,0008).
Schlußfolgerungen
Die Prognose der BMT-Patienten, die mit einer TBI bei akuter Leukämie behandelt wurden, wurde nicht durch die TBI-Technik signifikant beeinflußt, und die TRM schien bei den Patienten, die nach 1985 behandelt wurden, niedriger auszufallen. Außerdem wurde die Kataraktinzidenz durch die momentane Dosisrate signifikant beeinflußt.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Appelbaum F, L Fisher, ED Thomas. Chemotherapy vs. marrow transplantation for adults with acute nonlymphoblastic leukemia: a five-year follow-up. Blood 1988;72:179–84.
Baranov AE, GD Selidovkin, A Butturini et al. Hematopoietic recovery after 10-Gy acute total body irradiation. Blood 1994;83:596–9.
Barrett A. Total body irradiation (TBI) before bone marrow transplantation in leukemia: a co-operative study from the European Group for Bone Marrow Transplantation. Br J Radiol 1982;55:562–7.
Barrett A, MH Depledge, RL Powles. Interstitial pneumonitis following bone marrow transplantation after low dose rate total body irradiation. Int J Radiat Oncol Biol Phys 1983;9:1029–33.
Barrett A, J Nicholls, B Gibson. Late effects of total body irradiation. Radiother Oncol 1987;9:131–5.
Baume D, JM Cosset, JL Pico et al. Maladie veino-occlusive du foie après greffe de moelle osseuse: intérêt possible du fractionnement de l’irradiation corporelle totale. Presse Méd 1987;16:1759.
Belkacémi Y, M Ozsahin, F Pène et al. Cataractogenesis after total-body irradiation. Int J Radiat Oncol Biol Phys 1996;10:53–60.
Belkacémi Y, M Ozsahin, B Rio et al. Is veno-occlusive disease influenced by the total-body irradiation technique. Strahlenther Onkol 1995; 171:694–7.
Brauner R, M Fontoura, JM Zucker et al. Growth and growth hormone secretion after bone marrow transplantation. Arch Dis Child 1993; 68:458–63.
Brochstein JA, NA Kernan, S Groshen et al. Allogeneic bone marrow transplantation after hyperfractionated total-body irradiation and cyclophosphamide in children with acute leukemia. N Engl J Med 1987; 317:1618–24.
Calissendorff BM, P Bolme. Cataract development and outcome of surgery in bone marrow transplanted children. Br J Ophthalmol 1993;77:36–8.
Chalmers EA, AM Sproul, KI Mills et al. Effect of radiation dose on the development of mixed haemopoietic chimerism following T cell-depleted allogeneic bone marrow transplantation. Bone Marrow Transplant 1992;10:425–30.
Clift RA, CD Buckner, FR Appelbaum et al. Allogeneic marrow transplantation in patients with chronic myeloid leukemia in the chronic phase: a randomized trial of two irradiation regimens. Blood 1991;77:1660–65.
Cordonnier C, JF Bernaudin, P Bierling et al. Pulmonary complications occurring after allogeneic bone marrow transplantation: a study of 130 consecutive transplanted patients. Cancer 1986;58:1047–54.
Cosset JM, D Baume, JL Pico et al. Single dose versus hyperfractionated total body irradiation before allogeneic bone marrow transplantation: a non-randomized comparative study of 54 patients at the Institut Gustave-Roussy. Radiother Oncol 1989;15:151–60.
Cox D. R. Regression models and life tables. J R Stat Soc 1972;34:187–220.
Deeg HJ. Interstitial pneumonitis. In: Deeg HJ, Klingemann HG, Phillips GL, eds. A Guide to Bone Marrow Transplantation, 2nd edn, Berlin: Springer-Verlag, 1992:175–86.
Deeg HJ, N Flournoy, KM Sullivan et al. Cataracts after total body irradiation and marrow transplantation: a sparing effect of dose fractionation. Int J Radiat Oncol Biol Phys 1984;10:957–64.
Deeg JH, R Storb, G Longton et al. Single dose or fractionated total body irradiation and autologous marrow transplantation in dogs: effects of exposure rate, fraction size and fractionation interval on acute and delayed toxicity. Int J Radiat Oncol Biol Phys 1988;15:647–53.
Deeg HJ, KM Sullivan, CD Buckner et al. Marrow transplantation for acute nonlymphoblastic leukemia in first remission: toxicity and long-term follow-up of patients conditioned with single dose or fractionated total body irradiation. Bone Marrow Transplant 1986;1:151–7.
Dutreix J, E Gluckman, JM Brule. Biological problems of total body irradiation. J Eur Radiother 1982;3:163–73.
Evans RG, CL Wheatley, JR Nielsen. Modification of radiation-induced damage to bone marrow stem cells by dose rate, dose fractionation, and prior exposure to Cytoxan as judged by the survival of CFUs: Application to BMT. Int J Radiat Oncol Biol Phys 1988;14:491–5.
Ganem G, MF Saint-Marc Girardin, M Kuentz et al. Venocclusive disease of the liver after allogeneic bone marrow transplantation in man. Int J Radiat Oncol Biol Phys 1988;14:879–84.
Gorin NC, P Aegerter, B Auvert et al. Autologous bone marrow transplantation for acute myelocytic leukemia in first remission: a European survey of the role of marrow purging. Blood 1990;75:1606–14.
Kaplan EL, P Meier. Non-parametric estimation from incomplete observations. J Am Stat Assoc 1958;53:457–81.
Kim TH, PB McGlave, N Ramsay et al. Comparison of two total body irradiation regimens in allogeneic bone marrow transplantation for acute non-lymphoblastic leukemia in first remission. Int J Radiat Oncol Biol Phys 1990;19:889–97.
Kimler BF, CH Park, D Yakar et al. Radiation response of human normal and leukemic hematopoietic cells assayed by in vitro colony formation. Int J Radiat Oncol Biol Phys 1985;11:809–16.
Laver J. Radiobiological properties of human hematopoietic and stromal marrow cells. Int J Cell Cloning 1989;7:203–12.
Lazarus HM, GB Vogelsang, JM Rowe. Prevention and treatment of acute graft-versus-host disease: the old and the new. A report from the Eastern Cooperative Oncology Group (ECOG). Bone Marrow Transplant 1997;19:577–600.
Maraninchi D, JP Vernant, E Gluckman et al. Greffes de moelle allogéniques dans les leucémies aiguës myéloïdes: étude rétrospective chez 111 malades greffés en première rémission complète. Presse Méd 1986;1:2093–96.
McDonald GB, P Sharma, DE Matthews et al. Venocclusive disease of the liver after bone marrow transplantation: diagnosis, incidence and predisposing factors. Hepatology 1984;4:116–22.
McDonald GB, P Sharma, DE Matthews et al. The clinical course of 53 patients with venocclusive disease of the liver after marrow transplantation. Transplantation 1985;39:603–8.
Ozsahin M, Y Belkacémi, F Pène et al. Total-body irradiation and cataract incidence: a randomized comparison of two instantaneous dose rates. Int J Radiat Oncol Biol Phys 1994;28:343–7.
Ozsahin M, Y Belkacémi, F Pène et al. Interstitial pneumonitis following bone-marrow transplantation conditioned with cyclophosphamide and total-body irradiation. Int J Radiat Oncol Biol Phys 1996;34:71–7.
Ozsahin M, F Pène, JM Cosset et al. Morbidity after total body irradiation. Semin Radiat Oncol 1994;4:95–102.
Ozsahin M, F Pène, E Touboul et al. Total-body irradiation before bone marrow transplantation: results of two randomized instantaneous dose rates in 157 patients. Cancer 1992;69:2853–65.
Ozsahin M, LH Schwartz, F Pène et al. Is body weight a risk factor of interstitial pneumonitis after bone marrow transplantation? Bone Marrow Transplant 1992;10:97.
Peto P, MC Pike, P Armitage et al. Design and analysis of randomised clinical trials requiring prolonged observation of each patient: Part II. Br J Cancer 1977;35:1–39.
Rhee JG, CW Song, TH Kim et al. Effect of fractionation and rate of radiation dose on human leukemic cells, HL-60. Radiat Res 1985; 101:519–27.
Ringdén O, I Baryd, B Johansson et al. Increased mortality by septicemia, interstitial pneumonitis and pulmonary fibrosis among bone marrow transplant recipients receiving an increased mean dose rate of total body irradiation. Acta Radiol Oncol 1983;22:423–8.
Ringdén O, T Ruutu, M Remberger et al. A randomized trial comparing busulfan with total-body irradiation as conditioning in allogeneic marrow transplant recipients with leukemia: A report from the Nordic Bone Marrow Transplantation Group. Blood 1994;83:2723–30.
Ryalls M, HA Spoudeas, PC Hindmarsh et al. Short-term endocrine consequences of total body irradiation and bone marrow transplantation in children treated for leukemia. J Endocrinol 1993;136:331–8.
Sanders JE, CD Buckner, D Amos et al. Ovarian function following marrow transplantation for aplastic anemia or leukemia. J Clin Oncol 1988;6:813–8.
Scarpati D, F Frassoni, V Vitale et al. Total body irradiation in acute myeloid leukemia and chronic myelogenous leukemia: influence of dose and dose-rate on leukemia relapse. Int J Radiat Oncol Biol Phys 1989;17:547–52.
Schmidt GM, JC Niland, SJ Forman et al. Extended follow-up in 212 long-term allogeneic bone marrow transplant survivors. Transplantation 1993;55:551–7.
Shank B. Hyperfractionation versus single dose irradiation in human acute lymphocytic leukemia cells: application to TBI for marrow transplantation. Radiother Oncol 1993;27:30–5.
Shank B, FCH Chu, R Dinsmore et al. Hyperfractionated total body irradiation for bone marrow transplantation: results in seventy leukemia patients with allogeneic transplants. Int J Radiat Oncol Biol Phys 1983; 9:1607–11.
Shank B, RJ O’Reilly, I Cunningham et al. Total body irradiation for bone marrow transplantation: the Memorial Sloan-Kettering Cancer Center experience. Radiother Oncol Suppl 1990;1:68–71.
Sierra J, A Grañena, J García et al. Autologous bone marrow transplantation for acute leukemia: results and prognostic factors in 90 consecutive patients. Bone Marrow Transplant 1993;12:517–23.
Song CW, TH Kim, FM Khan et al. Radiobiological basis of total body irradiation with different dose rate and fractionation: repair capacity of hematopoietic cells. Int J Radiat Oncol Biol Phys 1981;7:1695–1701.
Sutton L, M Kuentz, C Cordonnier et al. Allogeneic bone marrow transplantation for adult acute lymphoblastic leukemia in first complete remission: factors predictive of transplant-related mortality and influence of total body irradiation. Bone Marrow Transplant 1993;12:583–9.
Tarbell NJ, DA Amato, JD Down et al. Fractionation and dose rate effects in mice: a model for bone marrow transplantation. Int J Radiat Oncol Biol Phys 1987;13:1065–69.
Tarbell NJ, EC Guinan, C Niemeyer et al. Late onset of renal dysfunction in survivors of bone marrow transplantation. Int J Radiat Oncol Biol Phys 1988;15:99–104.
Thomas ED. The use and potential of bone marrow allograft and whole-body irradiation in the treatment of leukemia. Cancer 1982;50:1449–54.
Thomas ED, RA Clift, J Hersman et al. Marrow transplantation for acute nonlymphoblastic leukemia in first remission using fractionated or single-dose irradiation. Int J Radiat Oncol Biol Phys 1982;8:817–21.
Thomas ED, R Storb, CD Buckner. Total body irradiation in preparation for marrow engraftment. Transplant Proc 1976;8:591–3.
Tichelli A. Late ocular complications after bone marrow transplantation. Nouv Rev Fr Hematol 1994;36:Suppl 1:S79–82.
Travis EL, LJ Peters, J McNeill et al. Effect of dose-rate on total body irradiation: lethality and pathologic findings. Radiother Oncol 1985;4:341–51.
Uckun FM, M Chandan-Langlie, W Jaszez et al. Radiation damage repair capacity of primary clonogenic blasts in acute lymphoblastic leukemia. Cancer Res 1994;53:1431–6.
Uckun FM, CW Song. Lack of CD24 antigen expression in B lincage acute lymphoblastic leukemia is associated with intrinsic radiation resistance of primary clonogenic blasts. Blood 1993;81:1323–32.
van Os R, H Thames, AWT Konings et al. Radiation-dose fractionation and dose-rate relationships for long-term repopulating hemopoietic stem cells in a murine bone marrow transplant model. Radiat Res 1993; 136:118–25.
Vriesendorp HM. Prediction of effects of therapeutic total body irradiation in man. Radiother Oncol Suppl 1990;1:37–50.
Vriesendorp HM. Radiobiological speculations on therapeutic total body irradiation. Crit Rev Oncol Hematol 1990;10:211–24.
Wagner JE Jr, GB Voselgang, WE Beschorner. Pathogenesis and pathology of graft vs. host disease. Am J Pediatr Hematol Oncol 1989;11:196–212.
Weiner RS, MM Bortin, RP Gale et al. Interstitial pneumonitis after bone marrow transplantation: assessment of risk factors. Ann Intern Med 1986;104:168–75.
Yan R., LJ Peters, EL Travis. Cyclophosphamide 24 hours before or after total body irradiation: effects on lung and bone marrow. Radiother Oncol 1991;21:149–56.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Belkacémi, Y., Pène, F., Touboul, E. et al. Total-body irradiation before bone marrow transplantation for acute leukemia in first or second complete remission. Strahlenther Onkol 174, 92–104 (1998). https://doi.org/10.1007/BF03038482
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03038482