Abstract
Purpose: To assess the effects of intraoperative infusion of dopexamine (a DA-1 and B2 adrenoreceptor agonist) on hemodynamic function, tissue oxygen delivery and consumption, splanchnic perfusion and gut permeability following aortic cross-clamp and release.
Methods: In a randomised double blind controlled trial 24 patients scheduled for elective infrarenal abdominal aortic aneurysm repair were studied in two centres and were assigned to one of two treatment groups.Group I received a dopexamine infusion starting at 0.5 µg·kg−1·min−1 increased to 2 µg·kg−1·min−1 maintaining a stable heart rate:Group II received a placebo infusion titrated in the same volumes following induction of anesthesia. Measured and derived hemodynamic data, tissue oxygen delivery and extraction and gut permeability were recorded at set time points throughout the procedure.
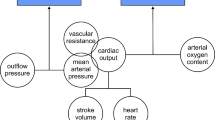
Results: Dopexamine infusion (0.5–2 µg·kg−1·min−1) was associated with enhanced hemodynamic function (MAP 65±5.5vs92±5.7 mm Hg,P=<0.05) only during the period of aortic cross clamping. However, during the most part of infrarenal abdominal aortic surgery, dopexamine did not reduce systemic vascular resistance index, mean arterial pressure nor oxygen extraction compared with the control group. The lactulose/ rhamnose permeation ratio was elevated above normal in both groups (0.22 and 0.29 in groups I and II respectively).
Conclusions: Dopexamine infusion (0.5 −2 µg·kg−1) did not enhance hemodynamic function and tissue oxygenation values during elective infrarenal abdominal aortic aneurysm repair.
Résumé
Objectif: Évaluer les effets d’une perfusion peropératoire de dopexamine, un agonist edes adrénorécepteurs DA-1 et B2, sur l’heémodynamie, l’apport et la consommation d’oxygène tissulaire, la perfusion splanchnique et la perméabilité intenstinale à la suite d’un clampage total de l’aorte et de son retrait.
Méthode: Il s’agit d’un essai ranomisé, contrôlé et à double insu auprès de 24 patients, provenant de deux centres et répartis en deux groupes, qui ont subi la réparation d’un anévrysme aortique abdominal sous-rénal. Le groupe I a reçu une perfusion de dopexamine amorcée à 0,5 µg·kg−1·min−1, augmentée à 2 µg·kg−1·min−1, maintenant une fréquence cardiaque stable; le groupe II a reçu une perfusion placebo de même volume après l’induction de l’anesthésie. Les données hémodynamiques mesurées et dérivées, l’apport et la consommation d’oxygène tissuulaire et la permeábilité intestinale ont été notés à des moments de mesure déterminés au cours de l’intervention.
Résultats: La perfusion de dopexamine (0,5 –2µg·kg−1) a stimulé la fonction hémodynamique (TAM 65±5,5vs 92±5,7 mm Hg,P=<0,05) pendant le clampage aortique seulement. Cependant, pendant la majeure partie de l’intervention aortique abdominale sous-rénale, la dopexamine n’a pas réduitl l’indice de résistance vasculaire générale, la tension artŕielle moyenne ou la consommation d’oxygène, ce sdonnées étant comparées avec celles du groupe témoin. Le ratio de dissémination de lactulose / rhamnose s’est élevé au-dessus de la normale dans les deux groupes (0,22 et 0,29 dans les groupes I et II respectivement).
Conclusion: La perfusion de dopexamine (0,5 –µg·kg−1·min−1) n’a pas amélioré l’hémodynamie ni haussé les valeurs d’oxygénation tissulaire pendant la réparation d’un anévrysme aortique abdominal sous-rénal.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Barash PG, Gullen BF, Stoeling RK. Clinical Anaesthesia, 2nd ed. Philadelphia: JB Lippincott, 1992: 1075–6.
Silverstein PR, Caldera DL, Cullen DJ, Davison JK, Darling RC, Emerson CW. Avoiding the hemodynamic consequences of aortic cross-clamping and unclamping. Anesthesiology 1979; 50: 462–6.
Attia RR, Murphy JD, Snider M, Lappas DG, Darling RC, Lowenstein E. Myocardial ischemia due to infrarenal aortic cross-clamping during aortic surgery in patients with severe coronary artery disease. Circulation 1976; 53: 961–5.
Gelman S, McDowell H, Varner P, et al. The reason for cardiac output reduction after aortic cross- clamping. Am J Surg 1988; 155: 578–86.
Lampe GH, Mangano DT. Anesthetic management for abdominal aortic reconstruction.In: Roizen MF (Ed.). Anesthesia for Vascular Surgery. New York: Churchill - Livingstone 1990: 265–84.
Cunningham AJ. Anaesthesia for abdominal aortic surgery — a review (Part I) Can J Anaesth 1989; 36: 426–44.
Brusoni B, Colombo A, Merlo L, Marchetti G, Longo T. Hemodynamic and metabolic changes induced by temporary clamping of the thoracic aorta. Eur Surg Res 1978; 10: 206–16.
Shenaq SA, Casar G, Chelly JE, Ott H, Crawford ES. Continuous monitoring of mixed venous oxygen saturation during aortic surgery. Chest 1987; 92: 796–99.
Gregoretti S, Gelman S, Henderson T, Bradley EL. Hemodynamics and oxygen uptake below and above aortic occlusion during crossclamping of the thoracic aorta and sodium nitroprusside infusion. J Thorac Cardiovasc Surg 1990; 100: 830–36.
Gelman S. The pathophysiology of aortic cross-clamping and unclamping. Anesthesiology 1995; 82: 1026–60.
Falk JL, Rackow EC, Blumenberg R, Gelfand M, Fein IA. Hemodynamic and metabolic effects of abdominal aortic crossclamping. Am J Surg 1981; 142: 174–7.
Hong S-AH, Gelman S, Henderson T. Angiotensin and adrenoceptors in the hemodynamic response to aortic crossclamping. Arch Surg 1992; 127: 438–41.
Maynard N, Bihari D, Beale R, et al. Assessment of splanchnic oxygenation by gastric tonometry in patients with acute circulatory failure. JAMA 1993; 270: 1203–10.
Garrett SA, Pearl RG. Improved gastric tonometry for monitoring tissue perfusion: the canary sings louder (Editorial). Anesth Analg 1996; 83: 1–3.
Meakins JL, Marshall JC. The gut as the motor of multiple system organ failure.In: Marston A, Bulkey GB, Fiddian-Green RG (Eds.). Splanchnic Ischaemia and Multiple Organ Failure. London: Edward Arnold, 1989: 339–48.
Fiddian-Green RG. Studies in splanchnic ischemia and multiple organ failure.In: Marston A, Bulkey GB, Fiddian-Green RG (Eds.). Splanchnic Ischaemia and Multiple Organ Failure. London: Edward Arnold, 1989: 349–63.
Leier CV. Regional blood flow responses to vasodilators and inotropes in congestive heart failure. Am J Cardiol 1988; 62: 86E-93E.
Smith GW, O’Connor SE. An introduction to the pharmacologic properties of Dopacard (dopexamine hydrochloride). Am J Cardiol 1988; 62: 9C-17C.
Mitchell PD, Smith GW, Wells E, West PA. Inhibition of uptake1 by dopexamine hydrochloridein vitro. Br J Pharmacol 1987; 92: 265–70.
Lier CV, Binkley PF, Carpenter J, Randolph PH, Unverferth DV. Cardiovascular pharmacology of dopexamine in low cardiac congestive heart failure. Am J Cardiol 1988; 62: 94–9.
Boyd O, Lamb G, MacKay CJ, Grounds RM, Bennett ED. A comparison of the efficacy of dopexamine and dobutamine for increasing oxygen delivery in high-risk surgical patients. Anaesth Intensive Care 1995; 23: 478–84.
Boyd O, Grounds RM, Bennett ED. A randomised clinical trial of the effect of deliberate perioperative increase of oxygen delivery on mortality in high risk surgical patients. JAMA 1993; 270: 2699–707.
Sinclair DG, Haslam PI, Quinlan GJ, Pepper JR, Evans TW. The effect of cardiopulmonary bypass on intestinal and pulmonary endothelial permeability. Chest 1995; 105: 718–24.
Sinclair DG, Houldsworth PE, Keogh B, Pepper J, Evans TW. Gastrointestinal permeability following cardiopulmonary bypass: a randomised study comparing the effects of dopamine and dopexamine. Intensive Care Med 1997; 23: 510–6.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McGinley, J., Lynch, L., Hubbard, K. et al. Dopexamine hydrochloride does not modify hemodynamic response or tissue oxygenation or gut permeability during abdominal aortic surgery. Can J Anesth 48, 238–244 (2001). https://doi.org/10.1007/BF03019752
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03019752