Abstract
Purpose: To determine whether the compromised intestinal villus blood flow in a rat model of endotoxemia could be improved by continuous infusion of the phosphodiesterase (PDE) inhibitor milrinone.
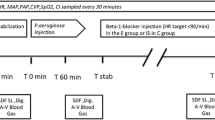
Methods: Twenty-four anesthetized and ventilated rats were laparotomized and an ileal portion was exteriorized and opened by an antimesenteric incision. The ileal segment was fixed with the mucosal surface upward. Microcirculatory parameters were assessed by intravital videomicroscopy. The animals were randomly assigned to receive one of three treatments: infusion ofEscherchia coli lipopolysaccharides without phosphodiesterase inhibitor pretreatment (=LPS group); or infusion of LPS with milrinone pretreatment (=milrinone group), or without infusion of LPS or milrinone (=control group). Macrohemodynamic parameters (MAP, HR) and microhemodynamic parameters of ileal mucosa (mean diameter of central arterioles=DA and mean erythrocyte velocity within the arterioles=VE) were measured 30 min before and at 0, 60, and 120 min after induction of endotoxemia. Mucosal villus blood flow was calculated from DA and VE.
Results: In the milrinone group MAP decreased 60 min after induction of endotoxemia whereas it remained stable in the control and the LPS group. In both groups given endotoxin VE decreased after start of LPS infusion. In contrast, DA decreased in the LPS group, but increased in the milrinone group after 120 min of endotoxemia. Thus, the endotoxin-induced decrease of intestinal villus blood flow was diminished but not fully restored by milrinone infusion.
Conclusion: Our results indicate that milrinone has some beneficial microcirculatory effects during endotoxemia. Although it contributed to systemic hypotension, it attenuated intestinal mucosal hypoperfusion.
Résumé
Objectif: Déterminer si le flot sanguin intestinal dont les grandes fonctions sont altérées dans un modèle d’endotoxémie chez le rat peut être amélioré par une perfusion continue d’un inhibiteur de la phosphodiestérase (PDE), la milrinone.
Méthode: Vingt-quatre rats sous anesthésie et ventilation ont été laparotomisés et une portion iléale a été extériorisée et ouverte par une incision antimésentérique. Le segment iléal a été fixé par la surface muqueuse extériorisée. Les paramètres microcirculatoires ont été évalués par vidéomicroscopie vitale. Les animaux ont été assignés à l’un des trois groupes de traitements: une perfusion de lipopolysaccharides d’Escherichia coli sans prétraitement avec l’inhibiteur de la phosphodiestérase (groupe LPS), ou une perfusion de LPS avec un prétraitement à la milrinone (groupe milrinone), ou sans perfusion de LPS ou de milrinone (groupe témoin). Les paramètres macrohémodynamiques (TAM, FC) et microhémodynamiques de la muqueuse iléale (diamètre moyen des artérioles centrales DA et vélocité moyenne des érythrocytes dans les artérioles VE) ont été mesurés 30 min avant l’induction de l’endotoxémie et à 0, 60 et 120 min après. Le flot sanguin des villosités muqueuses a été calculé à partir de DA et de VE.
Résultats: Dans le groupe milrinone, la TAM a baissé 60 min après l’induction de l’endotoxémie alors qu’elle est demeurée stable chez les témoins et dans le groupe LPS. Dans les deux groupes qui ont reçu l’endotoxine, VE a diminué après le début de la perfusion de LPS. Par ailleurs, DA était plus faible dans le groupe LPS, mais plus grand dans le groupe milrinone après 120 min d’endotoxémie. Donc, la baisse du flot sanguin intestinal induite par l’endotoxine a été réduite mais non complètement éliminée par la perfusion de milrinone.
Conclusion: Nos résultats indiquent que la milrinone présente certains effets microcirculatoires positifs pendant l’endotoxémie. Même si elle contribue à l’hypotension générale, elle limite l’hypoperfusion de la muqueuse intestinale.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Zimmerman JE, Knaus WA, Wagner DP, Sun X, Hakim RB, Nystrom P-O. A comparison of risks and outcomes for patients with organ system failure: 1982–1990. Crit Care Med 1996; 24: 1633–41.
Deitch EA. Multiple organ failure. Pathophysiology and potential future therapy. Ann Surg 1992; 216: 117–34.
Deitch EA. The role of intestinal barrier failure and basterial translocation in the development of systemic infection and multiple organ failure. Arch Surg 1990; 125: 403–4.
Fink MP, Kaups KL, Wang H, Rothschild HR. Maintenance of superior mesenteric arterial perfusion prevents increased intenstinal mucosal permeability in endotoxic pigs. Surgery 1991; 110: 154–60.
Berg RD. Bacterial translocation from the gastrointestinal tract. J Med 1992; 23: 217–44.
Carrico CJ, Meakins JL, Marshall JC, Fry D, Maier RV. Multiple-organ-failure syndrome. Arch Surg 1986; 121: 196–208.
Fink MP, Antonsson JB, Wang H, Rothschild HR. Increased intestinal permeability in endotoxic pigs. Mesenteric hypoperfusion as an etiologic factor. Arch Surg 1991; 126: 211–8.
Robertson DW, Beedle EE, Swartzendruber JK, et al. Bipyridine cardiotonics: the three-dimensional structures of amrinone and milrinone. J Med Chem 1986; 29: 635–40.
Barton P, Garcia J, Kouatli A, et al. Hemodynamic effects of IV milrinone lactate in pediatric patients with septic shock. A prospective, double-blinded, randomized, placebo-controlled, interventional study. Chest 1996; 109: 1302–12.
Heinz G, Geppert A, Delle Karth G, et al. IV milrinone for cardiac output increase and maintenance: comparison in nonhyperdynamic SIRS/sepsis and congestive heart failure. Intensive Care Med 1999; 25: 620–4.
Banner KH, Moriggi E, Da Ros B, Schioppacassi G, Semeraro C, Page CP. The effect of selective phosphodiesterase 3 and 4 isoenzyme inhibitors and established anti-asthma drugs on inflammatory cell activation. Br J Pharmacol 1996; 119: 1255–61.
Yoshimura T, Kurita C, Nagao T, et al. Effects of cAMP-phosphodiesterase isozyme inhibitor on cytokine production by lipopolysaccharide-stimulated human peripheral blood mononuclear cells. Gen Pharmacol 1997; 29: 633–8.
Molnar-Kimber K, Yonno L, Heaslip R, Weichman B. Modulation of TNF alpha and IL-1 beta from endotoxin-stimulated monocytes by selective PDE isozyme inhibitors. Agents Actions 1993; 39: C77–9.
Nielson CP, Vestal RE, Sturm RJ, Heaslip R. Effects of selective phosphodiesterase inhibitors on the polymor-phonuclear leukocyte respiratory burst. J Allergy Clin Immunol 1990; 86: 801–8.
Schmidt H, Secchi A, Wellmann R, et al. Effect of endotoxemia on intestinal villus microcirculation in rats. J Surg Res 1996; 61: 521–6.
Schmidt H, Secchi A, Wellmann R, Böhrer H, Bach A, Martin F. Effect of low-dose dopamine on intestinal villus microcirculation during normotensive endotoxemia in rats. Br J Anaesth 1996; 76: 707–12.
Schmidt W, Schmidt H, Bauer H, Gebhard MM, Martin E. Influence of lidocaine on endotoxin-induced leukocyte-endothelial cell adhesion and macromolecular leakagein vivo. Anesthesiology 1997; 87: 617–24.
Schmidt H, Secchi A, Wellmann R, Bach A, Böhrer H, Martin E. Dopexamine maintains intestinal villus blood flow during endotoxemia in rats. Crit Care Med 1996; 24: 1233–7.
Bennett ED. Dopexamine: much more than a vasoactive agent (Editorial). Crit Care Med 1998; 26: 1621–2.
Schmidt W, Häcker A, Gebhard MM, Martin E, Schmidt H. Dopexamine attenuates endotoxin-induced micro-circulatory changes in rat mesentery: role of beta2 adrenoceptors. Crit Care Med 1998; 26: 1639–45.
Bohlen HG, Gore RW. Preparation of rat intestinal muscle and mucosa for quantitative microcirculatory studies. Microvasc Res 1976; 11: 103–10.
Gore RW, Bohlen HG. Microvascular pressures in rat intestinal muscle and mucosal villi. Am J Physiol 1977; 233: H685–93.
Butcher EC, Weissman IL. Direct fluorescent labeling of cells with fluorescein or rhodamine isothiocyanate. I. Technical aspects. J Immunol Methods 1980; 37: 97–108.
Sarelius IH, Duling BR. Direct measurement of microverssel hematocrit, red cell flux, velocity, and transit time. Am J Physiol 1982; 243: H1018–26.
Bone RC. Gram-negative sepsis. Background, clinical features, and intervention. Chest 1991; 100: 802–8.
Bone RC, Balk RA, Cerra FB, et al. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. Chest 1992; 101: 1644–55.
Fink MP, Heard SO. Laboratory models of sepsis and septic shock. J Surg Res 1990; 49: 186–96.
Lindgren S, Almqvist P, Arvidsson D, Montgomery A, Andersson K-E, Haglund U. Lack of beneficial effects of milrinone in severe septic shock. Circ Shock 1990; 31: 365–75.
Vincent J-L Domb M, Van der Linden P, et al. Amrinone administration in endotoxin shock. Circ Shock 1988; 25: 75–83.
Secchi A, Wellmann R, Martin E, Schmidt H. Dobutamine maintains intestinal villus blood flow during normotensive endotoxemia: an intravital microscopic study in the rat. J Crit Care 1997; 12: 137–41.
Theuer CJ, Wilson MA, Steeb GD, Garrison RN. Microvascular vasoconstriction and mucosal hypoperfusion of the rat small intestine during bacteremia. Circ Shock 1993; 40: 61–8.
Spain DA, Wilson MA, Bar-Natan MF, Garrison RN. Nitric oxide synthase inhibition aggravates intestinal microvascular vasoconstriction and hypoperfusion of bacteremia. J Trauma 1994; 36: 720–5.
Fantini GA, Shiono S, Bal BS, Shires GT. Adrenergic mechanisms contribute to alterations in regional perfusion during normotensive E.coli bacteremia. J Trauma 1989; 29: 1252–7.
Wilson MA, Steeh GD, Garrison RN. Endothelins mediate intestinal hypoperfusion during bacteremia. J Surg Res 1993; 55: 168–75.
Lindgren S, Andersson K-E, Belfrage P, Degerman E, Manganiello VC. Relaxant effects of the selective phosphodiesterase inhibitors milrinone and OPC 3911 on isolated human mesenteric vessels. Pharmacol Toxicol 1989; 64: 440–5.
Möllhoff T, Loick HM, Van Aken H, et al. Milrinone modulates endotoxemia, systemic inflammation, and subsequent acute phase response after cardiopulmonary bypass (CPB). Anesthesiology 1999; 90: 72–80.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by a Research Fund of the Cooperative Research Committee of the Faculty of Medicine, University of Heidelberg, Germany.
Rights and permissions
About this article
Cite this article
Schmidt, W., Tinelli, M., Secchi, A. et al. Milrinone improves intestinal villus blood flow during endotoxemia. Can J Anaesth 47, 673–679 (2000). https://doi.org/10.1007/BF03019001
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03019001