Abstract
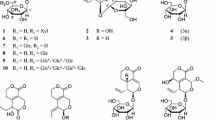
Seven known oleanolic acid glycosides (1–7) were isolated from the MeOH extract ofTiarella polyphylla. The structures were identified to be 3-O-(β-D-glucopyranosyl) oleanolic acid (1), 3-O-[β-D-glucopyranosyl-(1→3)-β-D-glucopyranosyl] oleanolic acid (2), 3-O-[β-D-glucopyranosyl-(1→2)-β-D-glucopyranosyl] oleanolic acid (3), 3-O-[β-D-glucopyranosyl-(1→3)-β-D-glucopyranosyl] oleanolic acid 28-O-β-D-glucopyranosyl ester (4), 3-O-[β-D-glucopyranosyl-(1→2)-β-D-glucopyranosyl] lleanolic acid 28-O-β-D-glucopyranosyl ester (5), 3-O-[a-L-rhamnopyranosyl-(1→3)-β-D-glucuronopyranosyl] oleanolic acid (6), and 3-O-[α-L-rhamnopyranosyl-(1→3)-β-D-glucuronopyranosyl] oleanolic acid 28-O-β-D-glucopyranosyl ester (7) on the basis of physicochemical and spectral data. These triterpene glycosides were tested for the anticomplement activity and hemolytic activity. Bisdesmosidic saponins,4, 5, and7, showed anticomplement activity; in contrast, monodesmosidic saponins,1–3, and6, showed direct hemolytic activity. Methyl esterified monodesmosidic saponins showed anticomplement activity at a low concentration and hemolytic activity at a high concentration.
Similar content being viewed by others
References Cited
Borel, C. and Hostettmann, K., Molluscicidal saponins fromSwartzia madagascariensis Desvaux.Helv. Chim. Acta., 70, 570–576 (1987).
Borel, C., Gupta, M. P. and Hostettmann, K., Molluscicidal saponins fromSwartzia simplex.Phytochemistry, 26, 2685–2689 (1987).
Davidyants, E. S., Putieva, Zh. M., Bandyukova, V. A. and Abubakirov, N. K., Triterpene glycosides ofSilphium perfoliatum. III. Structure of silphioside E.Chem. Nat. Compd., 20, 708–710 (1984).
Kabat, E. A. and Mayer, M. M. in Experimental immunochemistry Springfield, Illinois, 1961.
Kim, J. G. in Illustrated natural drugs encyclopedia, Nam-San-Dang Co. Seoul, 1984.
Kim, D. S., Oh, S. R., Lee, I. S., Jung, K. Y., Park, J. D., Kim, S. I. and Lee, H. K., anticomplementary activity of ginseng saponins and their degradation products.Phytochemistry, 47, 397–399 (1998).
Kuby, J. in Immunology, W. H. Freeman, New York, pp 336–355 (1992).
Nagao, T., Tanaka, R., Iwase, Y., Hanazono, H and Okabe, H., Studies on the constituents ofLuffa acutangula Roxb. I. Structures of acutosides A-G, oleanane-type triterpene saponins isolated from the herb.Chem. Pharm. Bull., 39, 599–606 (1991).
Oh, S. R., Jung, K. Y. and Lee, H. K.,In vitro anticomplementary activity of phenylpropanoids fromAgastache rugosa.Kor. J. Pharmacogn., 27, 20–25 (1996).
Romussi, G., Ciarallo, G., Falson, G. and Schneider, C., Constituents of Boraginaceae. Triterpene saponins fromAnchusa officinalis L.Lieb. Ann. Chem., 2028–2035 (1979).
Rother, K., Rother, U. and Hansch, G., The role of complement in inflammation.Path. Res. Pract., 180, 117–124 (1985).
Sati, O. P., Uniyal, S. K., Bahuguna, S. and Kikuchi, T., Clematoside-S, A triterpenoid saponin from the roots ofClematis grata.Phytochemistry, 29, 3676–3678 (1990).
Sotheeswaran, S., Bokel, M. and Kraus, W., A hemolytic saponin, randianin, fromRandia dumetorum.Phytochemistry, 28, 1544–1546 (1989).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, SH., Oh, S.R., Jung, K.Y. et al. Anticomplement activities of oleanolic acid monodesmosides and bisdesmosides isolated fromTiarella polyphylla . Arch Pharm Res 22, 428–431 (1999). https://doi.org/10.1007/BF02979071
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02979071