Summary
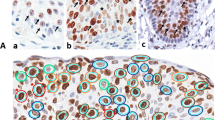
To evaluate proliferating cell nuclear antigen (PCNA) staining for assessing proliferative activity in routine pathology specimens of urinary bladder, the bladder carcinoma cell line J82 and a total of 122 specimens of normal bladder and urothelial lesions were stained with the antibody clone PC10 against proliferating cell nuclear antigen. In in vitro plateau cultures the proportion of PCNA-positive cells exceeded that of Ki-67-positive cells, and only very few cells were negative. In formalin-fixed tissues, the PCNA staining pattern, which should be confined to replicon units in the nucleus, was optimized by 1 h postfixation in an organic solvent (methacarn). Sections showed positive nuclear staining confined to basal and some suprabasal cells in normal urothelium and grade 1 dysplasias, but more generalized nuclear staining in all other neoplastic lesions. In addition, stromal cells adjacent to invasive tumors showed nuclear positivity in some instances. Using quantitative true color image analysis of sections counterstained with hemalum, the degree of brown staining of the PCNA reaction product is contrasted with the blue staining of the nuclear area. With this method low contrast specific staining not appreciated optically can be reliably detected. Image analysis data confirmed observations made on noncounterstained sections and showed significant differences between grade 1 and 2 dysplasias as well as between grade 1 dysplasia and all grades of papillary tumor. Furthermore, a significant difference in PCNA staining indices was found between grade 1 and 3 bladder carcinomas. The results indicate that PCNA staining using the PC10 antibody is not confined to the proliferative fraction of neoplastic urothelium. In contrast with data from normal tissue and malignant hematological neoplasms, the amount of PCNA is regulated differently in urothelial neoplasms, emphasizing the biological differences between the following two sets: mild dysplasia and moderate dysplasia; mild dysplasia and papillary carcinomas. The use of image analysis to standardize the detection process after controlled staining conditions is advisable in order to provide reliable data.
Similar content being viewed by others
References
Baserga R (1991) Growth regulation of the PCNA gene. J. Cell Sci 98: 433–436
Bravo R, Macdonald-Bravo H (1985) Changes in the nuclear distribution of cyclin (PCNA) but not its synthesis depend on DNA replication. EMBO J 4: 655–661
Bravo R, Macdonald-Bravo H (1987) Existence of two populations of cyclin/proliferating cell nuclear antigen during the cell cycle: association with DNA replication sites. J Cell Biol 105: 1549–1554
Diebold J, Lai MD, Loehrs U (1992) Analysis of proliferative activity in colorectal mucosa by immunohistochemical detection of proliferating cell nuclear antigen (PCNA). Methodological aspects and application to routine diagnostic material. Virchows Arch [B] 62: 283–289
Dierendonck JH van, Wijsman JH, Keijzer R, Velde CJH van de, Cornelisse CJ (1991) Cell-cycle-related staining patterns of antiproliferating cell nuclear antigen monoclonal antibodies. Comparisom with BrdUrd labeling and Ki-67 staining. Am J Pathol 138: 1165–1172
Fairman MP (1990) DNA polymerase delta/PCNA: actions and interactions (commentary). J Cell Sci 95: 1–4
Feitz WFJ, Beck HLM, Smeets AWGB, Debruyne FMJ, Vooijs GP, Herman CJ, Ramaekers FCS (1985) Tissue specific markers in flow cytometry of urological cancers: cytokeratins in bladder carcinoma. Int J Cancer 36: 349–358
Garcia RL, Coltrera MD, Gown AM (1989) Analysis of proliferative grade using anti PCNA/cyclin monoclonal antibodies in fixed, embedded tissues. Comparison with flow cytometric analysis (rapid communication). Am J Pathol 134: 733–739
Gerdes J, Schwab U, Lemke H, Stein H (1983) Production of a monoclonal antibody reactive with a human nuclear antigen associated with cell proliferation. Int J Cancer 31: 13–20
Hall PA, Levison DA, Woods AL, Yu CC-W, Kellock DB, Watkins JA, Barnes DM, Gillett CE, Camplejohn R, Dover R, Waseem NH, Lane P (1990) Proliferating cell nuclear antigen (PCNA) immunolocalization in paraffin sections: an index of cell proliferation with evidence of deregulated expression in some neoplasms. J Pathol 162: 285–294
Helpap B (1989) Pathologie der ableitenden Harnwege und der Prostata. Springer, Berlin Heidelberg New York
Jaskulski D, Gatti C, Travaki S, Calabretta B, Baserga R (1988) Regulation of the proliferating cell nuclear antigen cyclin and thymidine kinase mRNA levels by growth factors. J Biol Chem 263: 10175–10179
Knuechel R, Hofstaedter F, Sutherland RM, Keng PC (1990) Proliferation-associated antigens PCNA and Ki67 in two- and threedimensional experimental systems of human squamous carcinoma. Verh Dtsch Ges Pathol 74: 275–278
Liu Y-C, Marraccino RL, Keng PC, Bambara RA, Lord EM, Chou W-G, Zain SB (1989) Requirement for proliferating cell nuclear antigen expression during stages of the Chinese hamster ovary cell cycle. Biochemistry 28: 2967–2974
Masters JRW, Hepburn PJ, Walker L, Highman WJ, Trejdowiewics LK, Povey S, Parker M, Hill B, Riddle T, Franks L (1986) Tissue culture model of transitional cell carcinoma: charactertization of twenty-two urothelial carcinoma cell lines. Cancer Res 46: 3630–3636
Morris GF, Mathews MB (1989) Regulation of proliferating cell nuclear antigen during the cell cycle. J Cell Biol 264: 13856–13864
Mostofi FK, Sobin LH, Torloni H (1973) Histological typing of urinary bladder tumors. International histological typing of tumors, no. 10. WHO, Geneva
Neal DE, Sharples L, Smith K, Fennelly J, Hall RR, AL Harris (1990) The epidermal growth fictor receptor and the prognosis of bladder cancer. Cancer 65: 1619–1625
O'Reilly SM, Richard MA (1992) Clinical methods of assessing cell proliferation. In: Hall PA, Levison DA, Wright, NA (eds) Assessment of cell proliferation in clinical practice. Springer Berlin Heidelberg, pp 177–192
Scott RJ, Hall PA, Haldane JS, Van Noorden S, Price Y, Lane DP, Wright NA (1991) A comparison of immunohistochemical markers of cell proliferation with experimentally determined growth fraction. J Pathol 165: 173–178
Shipman-Appasamy P, Cohen KS, Prystowsky MB (1990) Interleukin 2-induced expression of proliferating cell nuclear antigen is regulated by transcriptional and posttranscriptional mechanisms. J Biol Chem 265: 19180–19184
Spiessl B, Beahrs OH, Hermanek P, Hutter RVP, Scheibe O, Sobin LH, Wagner G (1989) TNM atlas. Illustrated guide to the TNM/pTNM-classification of malignant tumours, 3rd edn., Springer, Berlin Heidelberg New York
Tachibana M, Deguchi N, Jitsukawa S, Baba S, Hata M, Tazaki H (1991) Quantification of cell kinetic characteristics using flow cytometric measurements of deoxyribonucleic acid and bromodeoxyuridine for bladder cancer. J Urol 145: 1159–1163
Terada T, Nakanuma Y (1992) Cell proliferative activity in adenornatous hyperplasia of the liver and small hepatocellular carcinoma. An immunohistochemical study demonstrating proliferating cell nuclear antigen. Cancer 70: 591–598
Toschi L, Bravo R (1988) Changes in cyclin/PCNA distribution during DNA repair synthesis. J Cell Biol 107: 1623–1628
Waseem NH, Lane DP (1990) Monoclonal antibody analysis of the proliferating cell nuclear antigen (PCNA). Structural conservation and the detection of a nucleolar form. J Cell Sci 96: 121–129
Wilkins BS, Harris S, Waseem NH, Lane DP, Jones DB (1992) A study of cell proliferation in formalin-fixed, wax-embedded bone marrow trephine biopsies using the monoclonal antibody PC10, reactive with proliferating cell nuclear antigen (PCNA). J Pathol 166: 45–52
Wolf HK, Dittrich KL (1992) Detection of proliferating cell nuclear antigen in diagnostic histopathology. J Histochem Cytochem 40: 1269–1273
Woods Al, Hall PA, Shepherd NN, Handby AM, Waseem NH, Lane DP. Levison DA (1991) The assessment of proliferating cell nuclear antigen (PCNA) immunostaining in primary gastrointestinal lymphomas and its relationship to histological grade, S+G2M phase fraction (flow cytometric analysis) and prognosis. Histopathology 19: 21–27
Yu CC-W, Woods A, Levison DA (1992) The assessment of cellular proliferation by immunohistochemistry: a review of currently available methods and their applications. Histochem J 24: 121–131
Author information
Authors and Affiliations
Additional information
Supported by the DFG project: Knuechel/Urothelcarcinom 263
Rights and permissions
About this article
Cite this article
Knuechel, R., Burgau, M., Rueschoff, J. et al. Proliferating cell nuclear antigen in normal urothelium and urothelial lesions of the urinary bladder: a quantitative assessment using a true color image analysis system. Virchows Archiv B Cell Pathol 64, 137–144 (1993). https://doi.org/10.1007/BF02915106
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02915106