Summary
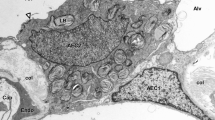
Surface proteins were compared in endothelial cells (EC) obtained from bovine peripheral lung, pulmonary artery and vein, and dorsal aorta using sodium dodecyl sulfate (SDS) polyacrylamide gel electrophoresis. Galactose-containing glycoproteins [molecular weight (Mr) 160–220 and 40 kDa] binding to theRicinus communis agglutinin (RCA) and peanut agglutinin (PNA) were selectively observed on pulmonary microvessel EC as compared to EC from pulmonary artery, pulmonary vein, and dorsal aorta. The unique RCA- and PNA-binding profiles of EC from the pulmonary artery and microvessels may be important in characterizing EC from different sites in the pulmonary circulation. The pulmonary microvessel EC monolayer was also 15-fold more restrictive to transendothelial flux of [14C]sucrose (Mr=342 Da) than the pulmonary artery EC monolayer. In contrast, the microvessel EC were only six- and twofold more restrictive to the flux of larger tracer molecules, ovalbumin (Mr 43 kDa) and albumin (Mr=69 kDa) than pulmonary artery EC. The greater restrictiveness of pulmonary microvessel EC monolayer indicates a major phenotypic difference in the cultured pulmonary microvessel EC barrier function.
Similar content being viewed by others
References
Auerbach, R.; Alby, L.; Morrissey, L. W., et al. Expression of organspecific antigens on capillary endothelial cells. Microvasc. Res. 29:401–411; 1985.
Belloni, P. N.; Nicolson, G. L. Differential expression of cell surface glycoproteins on various organ-derived microvascular endothelia and endothelial cell cultures. J. Cell. Physiol. 136:398–410; 1988.
Belloni, P. N.; Tressler, R. J. Microvascular endothelial cell heterogeneity: interactions with leukocytes and tumor cells. Cancer Metastasis Rev. 8:353–389; 1989.
Bocci, V. Efficient labelling of serum proteins with135I using chloramine-T. Int. J. Appl. Radiat. 15:449–456; 1964.
Cooper, J. A.; Del Vecchio, P. J.; Minnear, F. L., et al. Measurement of albumin permeability across endothelial monolayersin vitro. J. Appl. Physiol. 62:1076–1083; 1987.
Chung-Welch, N.; Patton, W. F.; Ameia Yen-Patton, G. P., et al. Phenotypic comparison between mesothelial and microvascular endothelial cell lineages using conventional endothelial cell markers, cytoskeletal protein markers andin vitro assays of angiogenic potential. Differentiation 42:44–53; 1989.
Del Vecchio, P. J.; Siflinger-Birnboim, A.; Shepard, J. M., et al. Endothelial monolayer permeability to macromolecules. Fed. Proc. 46:2511; 1987.
Folkman, J.; Haudenschild, C. C.; Zetter, B. R. Long-term culture of capillary endothelial cells. Proc. Natl. Acad. Sci. USA 76:5217–5221; 1979.
Gerritsen, M. E. Function heterogeneity of vascular endothelial cells. Biochem. Pharmacol. 36:2701–2711; 1987.
Horvath, C. J.; Ferro, T. J.; Jesmok, G., et al. Recombinant tumor necrosis factor increases pulmonary vascular permeability independent of neutrophils. Proc. Natl. Acad. Sci. USA 85:9219–9223; 1988.
Jaffe, E. A.; Hoyer, L. W.; Nachman, R. L. Synthesis of antihemophilic factor antigen by cultured human endothelial cells. J. Clin. Invest. 52:2757–2764; 1973.
Kumar, S.; West, D. C.; Ager, A. Heterogeneity in endothelial cells from large vessels and microvessels. Differentiation 36:57–70; 1987.
Montesano, R.; Orci, L.; Vassalli, P.In vitro rapid organization of endothelial cells into capillary-like networks is promoted by collagen matrices. J. Cell. Biol. 97:1648–1652; 1983.
Nicolson, G. L.; Winkelhake, J. L. Organ-specificity of blood-borne tumour metastasis by cell adhesion. Nature 255:230–232; 1975.
Ryan, U. S. Metabolic activity of pulmonary endothelium: modulation of structure and function. Ann. Rev. Physiol. 48:263–277; 1986.
Schroeter, D.; Spiess, E.; Paweletz, N., et al. A procedure for rupture-free preparation of confluently grown monolayer cells for scanning electron microscopy. J. Electron Microsc. Tech. 1:219–225; 1984.
Siflinger-Birnboim, A.; Del Vecchio, P. J.; Cooper, J. A., et al. Molecular sieving characteristics of the cultured endothelial monolayer. J. Cell. Physiol. 132:111–117; 1987.
Simionescu, M.; Simionescu, N.; Santoro, F., et al. Differentiated microdomains of the luminal plasmalemma of murine muscle capillaries: segmental variations in young and old animals. J. Cell. Biol. 100:1396–1407; 1985.
Schnitzer, J. E.; Siflinger-Birnboim, A.; Del Vecchio, P. J., et al. Segmental differentiation of permeability, protein glycosylation, and morphology of cultured bovine pulmonary vascular endothelium. J. Physiol. Lond. Submitted; 1992.
Taylor, R. F.; Pine, T. H.; Schwartz, S. M., et al. Neutrophil-endothelial monolayers grown on micropore filters. J. Clin. Invest. 67:584–587; 1981.
Tomasovic, S. P.; Simonette, R. A.; Wolf, D. A., et al. Co-isolation of heat stress and cytoskeletal proteins with plasma membranes. Int. J. Hyperthermia 5:173–190; 1989.
Towbin, H.; Staehelin, T.; Gordon, J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc. Natl. Acad. Sci. USA 76:4350–4354; 1979.
Voyta, J. C.; Via, D. P.; Butterfield, C. E., et al. Identification and isolation of endothelial cells based on their increased uptake of acetylated-low density lipoprotein. J. Cell. Biol. 99:2034–2040; 1984.
Williams, M. R.; McBride, D. Problems with radiolabelling egg albumin for the Farr ammonium sulfate technique for antibody estimation. J. Immunol. Methods 15:315–323; 1977.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
del Vecchio, P.J., Siflinger-Birnboim, A., Belloni, P.N. et al. Culture and characterization of pulmonary microvascular endothelial cells. In Vitro Cell Dev Biol - Animal 28, 711–715 (1992). https://doi.org/10.1007/BF02631058
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02631058