Summary
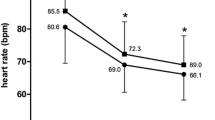
The clinical response to felodipine, in addition to a beta-blocker, was evaluated and compared with placebo in this double-blind cross-over study. Twenty patients with exertional angina pectoris completed the study. Felodipine reduced the number of angina attacks and the Glyceryl Trinitrate (GTN) consumption. The median exercise capacity was increased 33% after 4 weeks' felodipine treatment compared with placebo. At maximal exercise, systolic blood pressure and rate pressure product were reduced by felodipine while no change was seen in heart rate or ST-depression.
Felodipine reduced both supine and erect blood pressure. The mean supine blood pressure at rest was 138/82 mm Hg after four weeks' placebo treatment compared with 114/71 mm Hg after felodipine 5–10 mg b.i.d.
Felodipine has overall a modest but significant antianginal benefit when combined with a beta-blocker.
Similar content being viewed by others
References
Lynch P, Dargie H, Krikler S and Krikler D (1980) Objective assessment of antianginal treatment: A double-blind comparison of propranolol, nifedipine and their combination. Br Med J 280: 184–187
Braun S, Terdiman R, Berenfeld D and Laniado S (1985) Clinical and haemodynamic effects of combined propranolol and nifedipine therapy versus propranolol alone in patients with angina pectoris. Am Heart J 109: 478–485
Ljung B (1985) Vascular selectivity of felodipine. Drugs 29 [Suppl 2]: 46–58
Tweddel AC, Johnsson G, Pringle TH, Murray RG and Hutton I (1983) The systemic and coronary haemodynamic effects of felodipine in patients with coronary artery disease. Eur Heart J 4: 699–705
Detry J-MR, De Coster PM and Renkin J (1983) Haemodynamic effects of felodipine at rest and during exercise in exertional angina pectoris. Am J Cardiol 52: 453–475
Culling W, Ruttley MSM and Sheridan DJ (1984) Acute haemodynamic effects of felodipine during beta blockade in patients with coronary artery disease. Br Heart J 52: 431–434
Sheridan JV, Thomas P, Routledge PA and Sheridan DJ (1987) Effects of felodipine on haemodynamics and exercise capacity in patients with angina pectoris. Br J Clin Pharmacol 23: 391–396
Metcalfe MJ, Chan-Wah-Hak NS, Jennings K (1985) The effects of treatment with felodipine as a single agent in coronary artery disease. Br Heart J 61: 258–261
Anhoff M (1984) Determination of felodipine in plasma by gas chromatography with electron capture detection. J Pharm Biomed Anal 2: 519–526
Goldstein RE (1982) Coronary vascular responses to vasodilator drugs. Prog Cardiovasc Dis 24: 419–435
Adverse Drug Reaction Committee (ADRAC). Felodipine — Adverse cardiac effects. Australian Adverse Drug Reaction Committee Bulletin. December 1988
Adverse Reactions Advisory Committee (ADRAC). Nifedipine adverse cardiovascular effects. Australian Adverse Drug Reaction Committee Bulletin. March 1983
Wilhelmsen L, Berglund G, Elmfeldt D, Fitzsimons T, Holzgreve H, Hosie J, Hornkvist P-E, Pennert K, Tuomilehto J, Wedel H (1987) Beta-blockers versus diuretics in hypertensive men: main results from the HAPPHY trial. J Hypertens 5: 561–572
Chatterjee K, Rouleau JL, Parmley WW (1982) Haemodynamic and myocardial metabolic effects of captopril in chronic heart failure. Br Heart J 47: 233–238
Rouleau JL, Chatterjee K, Benge W, Parmley WW, Hiramatsu B (1982) Alterations in left ventricular function and coronary haemodynamics with captopril, hydralazine and prazosin in chronic ischaemic heart failure. Circulation 65: 671–678
Emanuelsson H, Holmberg S (1985) No adverse effects from high doses of felodipine to patients with coronary heart disease. Clin Cardiol 8: 329–336
The Canadian Felodipine Study Group (1988) Antihypertensive efficacy of the calcium-antagonist felodipine in patients with persisting hypertension on beta-blocker therapy. Br J Clin Pharmacol 26: 535–545
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lorimer, A.R., MacFarlane, P., Pringle, S. et al. The effects of felodipine in angina pectoris. Eur J Clin Pharmacol 38, 415–419 (1990). https://doi.org/10.1007/BF02336676
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02336676