Summary
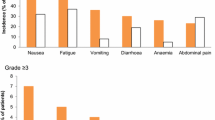
A Phase II study of Didemnin-B, a marine cyclic depsipeptide, was undertaken in patients with progressive epithelial ovarian cancer. The starting dose was 2.6 mg/m2. Fifteen patients received the drug, of whom twelve were evaluable. There were no responses observed in the twelve patients. The two most frequent toxicities were nausea and vomiting and anemia. On the basis of this trial, Didemnin-B is not felt to have significant effect with epithelial ovarian cancer.
Similar content being viewed by others
References
Rhineart KL Jr., Grever JB, Hughes RG, Swynenberg EG, Stringfellow DA, Kuentzel SL, Li LH: Didemnins: antiviral and antitumor depsipeptides from a Caribbean Tunicate Science 212:933, 1981
Chun HG, Davies B, Hoth D, Suffness M, Yun S: Didemnin-B: The first marine compound entering clinical trials as an antineoplastic agent. Invest New Drugs 4(3):279–84, 1986
Weiss GR, Arteaga C, Brown TD, Craig JB, Harman GS, Havlin KA, Koehler JH, Kuhu JG, Von Hoff DD: New anticancer agents. Ca Chemo Biol Res Modif 10:85–116, 1988
Ziang TL, Liu RL, Salman RF: Antitumor activity of didemnin-B in the human tumor stem cell assay. Cancer Chemotherapy Pharmacol 11:1, 1983
Miller A, Hoogstranten B, Staquet M, Winkler A: Reporting results of cancer treatment. Cancer 47:207–214, 1981
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cain, J.M., Liu, P.Y., Alberts, D.E. et al. Phase II trial of didemnin-B in advanced epithelial ovarian cancer. Invest New Drugs 10, 23–24 (1992). https://doi.org/10.1007/BF01275473
Issue Date:
DOI: https://doi.org/10.1007/BF01275473