Abstract
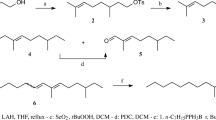
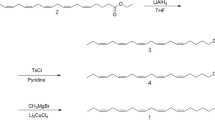
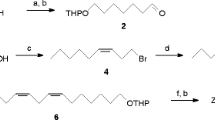
Although several sex pheromones of the family Geometridae have been characterized, investigations on Japanese species are limited. In order to obtain more information, screening using known sex pheromones and their analogs was carried out. The (Z,Z,Z)-3,6,9-triunsaturated and (Z,Z)-6,9-diunsaturated hydrocarbons with straight C19-C21 chains were synthesized by the Grignard coupling reaction as a key step starting from linolenic and linoleic acids, respectively. Oxidation of the homoconjugated trienes withm-chloroperoxybenzoic acid yielded a 1∶1∶1 mixture of three monoepoxy derivatives that could be separated by silica gel chromatography. The chemical structure of each positional isomer was confirmed using two-dimensional NMR techniques and MS measurements, which enabled characteristic fragment ions from the isomers to be identified. Field tests using lures incorporating only one of the above six hydrocarbons or nine epoxides were carried out in a forest in Tokyo. Consequently, attraction of male moths of 14 geometrid species in addition to four species in another family, the Noctuidae, was observed. It was concluded that hydrocarbons with a homoconjugated polyene system and the monoepoxy derivatives are important components of sex pheromones produced by Japanese lepidopterous insects, particularly the geometrid moths.
Similar content being viewed by others
References
Ando, T., Yoshida, S., Tatsuki, S., andTakahashi, N. 1977. Sex attractants for male Lepidoptera.Agric. Biol. Chem. 41:1485–1492.
Ando, T., Kuroko, H., Nakagaki, S., Saito, O., Oku, T., andTakahashi, N. 1981. Multicomponent sex attractants in systematic field tests of male Lepidoptera.Agric. Biol. Chem. 46:487–495.
Ando, T., Koike, M., Uchiyama, M., andKuroko, H. 1987. Lepidopterous sex attractants with a conjugated diene system.Agric. Biol. Chem. 51:2691–2694.
Ando, T., Ogura, Y., Koyama, M., Kurane, M., Uchiyama, M., andSeol, K.Y. 1988. Syntheses and NMR analyses of eight geometrical isomers of 10,12,14-hexadecatrienyl acetate, sex pheromone candidates of the mulberry pyralid.Agric. Biol. Chem. 52:2459–2468.
Arn, H.,Toth, M., andPriesner, E. 1986. List of Sex Pheromones of Lepidoptrea and Related Attractants. OILB-SROP/IOBC-WPRS, Secrétariat General, 149 Rue de Bercy, F-75595 Paris cedex 12; ISBN 92-9067-002-9, 123 pp.
Becker, D., Cyjon, R., Cossé, A., Moore, I., Kimmel, T., andWysoki, M. 1990. Identification and enantioselective synthesis of (Z,Z)-6,9-cis-3S,4R)-epoxynonadecadiene, the major sex pheromone component ofBoarmia selenaria.Tetrahedron Lett. 31:4923–4926.
Conner, W.E., Eisner, T., Meer, R.K.V., Guerrero, A., Ghiringelli, D., andMeinwald, J. 1980, Sex attractant of an arctiid moth (Utetheisa ornatrix): A pulsed chemical signal.Behav. Ecol. Sociobiol. 7:55–63.
Cossé, A.A., Cyjon, R., Moore, I., Wysoki, M., andBecker, D. 1992. Sex pheromone components of the giant looper,Boarmia selenaria Schiff. (Lepidoptera: Geometridae): Identification, synthesis, electrophysiological evaluation, and behavioral activity.J. Chem. Ecol. 18:165–181.
Fouquet, G., andSchlosser, M. 1974. Improved carbon-carbon linking by controlled copper catalysis.Angew. Chem. (Int. Ed. Engl.) 13:82–83.
Hansson, B.S., Szöcs, G., Schmidt, F., Francke, W., Löfstedt, C., andTóth, M. 1990. Electrophysiological and chemical analysis of sex pheromone communication system of the mottled umber,Erannis defoliaria (Lepidoptera: Geometridae).J. Chem. Ecol. 16:1887–1897.
Millar, J.G., Underhill, E.W., Giblin, M., andBarton, D. 1987. Sex pheromone components of three species ofSemiothisa (Geometridae), (Z,Z,Z)-3,6,9-heptadecatriene and two monoepoxydiene analogs.J. Chem. Ecol. 13:1371–1383.
Millar, J.G., Giblin, M., Barton, D., andUnderhill, E.W. 1990a. (3Z,6Z,9Z)-Nonadecatriene and enantiomers of (3Z,9Z)-cis-6,7-epoxynonadecadiene as sex attractants for two geometrid and one noctuid moth species.J. Chem. Ecol. 16:2153–2166.
Millar, J.G., Giblin, M., Barton, D., Morrison, A., andUnderhill, E.W., 1990b. Synthesis and field testing of enantiomers of 6Z,9Z-cis-3,4-epoxydienes as sex attractants for geometrid moths. Interactions of enantiomers and regioisomers.J. Chem. Ecol. 16:2317–2339.
Roelofs, W.L., Hill, A.S., Linn, C.E., Meinwald, J., Jain, S.C., Herbert, H.J., Smith, R.F. 1982. Sex pheromone of the winter moth, a geometrid with unusually low temperature precopulatory responses.Science 217:657–659.
Rossi, R., andVeracini, C.A. 1982. Insect pheromone components. Use of13C NMR spectroscopy for assigning the configuration of C=C double bonds of monoenic or dienic pheromone components and for quantitative determination ofZ/E mixtures.Tetrahedron 36:639–644.
Szöcs, G., Tóth, M., Bestmann, H.J., andVostrowsky, O. 1984. A two-component sex attractant for males of the geometrid mothAlsophila quadripunctata.Entomol. Exp. Appl. 38:287–291.
Underhill, E.W., Palaniswamy, P., Abrams, S.R., Bailey, B.K., Steck, W.F., andChisholm, M.D. 1983. Triunsaturated hydrocarbons, sex pheromone components ofCaenurgina erechtea.J. Chem. Ecol. 9:1413–1423.
Wong, J.W., Palaniswamy, P., Underhill, E.W., Steck, W.F., andChisholm, M.D. 1984. Sex pheromone components of fall cankerworm moth,Alsophila pometaria. Synthesis and field trapping.J. Chem. Ecol. 10:1579–1596.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ando, T., Ohsawa, H., Ueno, T. et al. Hydrocarbons with a homoconjugated polyene system and their monoepoxy derivatives: Sex attractants of geometrid and noctuid moths distributed in Japan. J Chem Ecol 19, 787–798 (1993). https://doi.org/10.1007/BF00985009
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00985009