Abstract
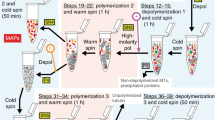
The soluble tubulin of human cerebral cortex, as assessed by [3H]colchicine binding of the 100,000g supernatant fraction, decreases drastically with age, 75 percent from age 0 to age 90. There is also a considerably lower concentration of high molecular weight proteins in the soluble fraction of postmortem human cerebral cortex than in that of nonhuman species. Human brain tubulin can be polymerized into microtubules with DEAE-dextran. The DEAE-dextran induced microtubules are stable to cold temperature (4°) and calcium. However, in the presence of 1 M glutamate, the microtubules become cold labile and depolymerize at 4°. Thus we have developed a novel method for purifying polymerization competent tubulin from fresh or frozen human cerebral cortex. Human brain tubulin purified by our novel method is very similar to tubulin from the brains of other mammals in molecular weight, amino acid composition, polymerization-depolymerization parameters, and structural dimensions of the microtubules formed.
Similar content being viewed by others
References
Soifer, D. (ed.) 1975. Ann. NY Acad. Sci., Vol. 253: The biology of cytoplasmic microtubules.
Butler, R. N. 1978. The National Institute of Mental Health Study. Pages 53–58,in Katzman, R., Terry, R. D., andBick, K. L. (eds.), Alzheimer's Disease: Senile Demential and Related Disorders (Aging Vol. 7), Raven Press, New York.
Terry, R. D., andDavies, P. 1980. Dementia of the Alzheimer's type. Ann. Rev. Neurosci. 3:77–95.
Grundke-Iqbal, I., Johnson, A. B., Wisniewski, H. M., Terry, R. D., andIqbal, K. 1979. Evidence that Alzheimer neurofibrillary tangles originate from neurotubules. Lancet 1:578–580.
Ghetti, B. 1979. Induction of neurofibrillary degeneration following treatment with maytansine in vivo. Brain Research 163:9–19.
Eng, L. F., Pratt, D., andWilson, L. 1974. Biochemical and pharmacological comparison of microtubule protein from human and chick brain. Neurobiology 4:301–308.
Yen, S. C., Gaskin, F., andTerry, R. D. 1981. Immunocytochemical studies of neurofibrillary tangles. Am. J. Pathol. 104:77–89.
Dahl, D., Selkoe, D. J., Pero, R. T., andBignami, A. 1982. Immunostaining of neurofibrillary tangles in Alzheimer's senile dementia with a neurofilament antiserum. J. Neurosci. 2:113–119.
Shelanski, M. L., Gaskin, F., andCantor, C. R. 1973. Microtubule assembly in the absence of added nucleotides. Proc. Natl. Acad. Sci. 70:765–768.
Weisenberg, R. V., andTimasheff, S. N. 1970. Aggregation of microtubule subunit protein: effect of divalent cations, colchicine and vinblastine. Biochem. 9:4110–4116.
Timasheff, S. N., andGrisham, L. M. 1980. In vitro assembly of cytoplasmic microtubules. Ann. Rev. Biochem. 49:565–591.
Redman, C. M., Banerjee, D., Howell, K., andPalade, G. E. 1975. Colchicine inhibition of plasma protein release from rat hepatocytes. J. Cell. Biol. 66:42–59.
Berk, B. C., andHinkle, P. M. 1980. Thyroid and brain microtubules: a comparison. J. Biol. Chem. 255:3186–3193.
Wilson, L. 1975. Microtubules as drug receptors—pharmacological properties of microtubule protein. Ann. NY Acad. Sci. 253:213–231.
Weber, K., andOsborn, M. 1969. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J. Biol. Chem. 244:4406–4412.
Lee, J. C., andTimasheff, S. N. 1975. The reconstitution of microtubules from purified calf brain tubulin. Biochem. 14:5183–5187.
Goodwin, T. W., andMorton, R. A. 1946. The spectrophotometric determination of tyrosine and tryptophan in proteins. Biochem. J. 40:628–632.
Haworth, W. M. 1915. A new method of preparing alkylated sugars. Chem. Soc. London J. 107:8–17.
Bradford, M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72:248–254.
Erickson, H. P., andVoter, W. A. 1976. Polycation-induced assembly of purified tubulin. Proc. Natl. Acad. Sci. 73:2813–2817.
Gaskin, F., Cantor, C. R., andShelanski, M. L. 1974. Turbidimetric studies of the in vitro assembly and disassembly of porcine neurotubules. J. Mol. Biol. 89:737–758.
Hamel, E., del Campo, A. A., Lowe, M. C. Waxman, P. G., andLin, C. M. 1982. Effects of organic acids on tubulin polymerization and associated guanosine 5′-triphosphate hydrolysis. Biochem. 21:503–509.
Bryan, J., andWilson, L. 1971. Are cytoplasmic microtubules heteropolymers? Proc. Natl. Acad. Sci. 68:1762–1766.
Eipper, B. A. 1974. Properties of rat brain tubulin. J. Biol. Chem. 249:1407–1416.
Feit, H., andBarondes, S. H. 1970. Colchicine-binding activity in particulate fractions of mouse brain. J. Neurochem. 17:1355–1364.
Job, D., Rauch, C. T., Fisher, E. H., andMargolis, R. L. 1982. Recycling of coldstable microtubules: evidence that cold stability is due to stoichiometric polymer blocks. Biochem. 21:509–515.
Lockwood, A. H. 1979. Molecules in mammalian brain that interact with the colchicine site on tubulin. Proc. Natl. Acad. Sci. 76:1184–1188.
Sherline, P., Schiavone, K., andBrocato, S. 1979. Endogenous inhibitor of colchicine-tubulin binding in rat brain. Science 205:593–595.
Bryan, J., Nagle, B. W., andDoenges, K. H. 1975. Inhibition of tubulin assembly by RNA and other polyanions: evidence for a required protein. Proc. Natl. Acad. Sci. 72:3570–3574.
Asnes, C. F., andWilson, L. 1979. Isolation of bovine brain microtubule protein without glycerol: polymerization kinetics change during purification cycles. Anal. Biochm. 98:64–73.
Author information
Authors and Affiliations
Additional information
Some aspects of this work have been published as an abstract in 1981. Fed. Proc. 40:1548.
Rights and permissions
About this article
Cite this article
Yan, Sc.B., Hwang, S., Rustan, T.D. et al. Human brain tubulin purification: Decrease in soluble tubulin with age. Neurochem Res 10, 1–18 (1985). https://doi.org/10.1007/BF00964768
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00964768