Abstract
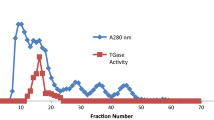
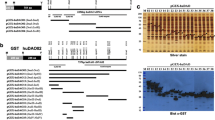
The catalytically inactivating subset within rabbit serum polyclonal antibody to the solubilized, purified 55,000 to 60,000 dalton active fragment of rat liver microsomal β-hydroxy-β-methylglutaryl coenzyme A reductase immunoinactivates this enzyme with little or no diminution of effect by enzyme catalytically inactivated by incubation of microsomes with ATP,Mg++. Reactivation of inactive enzyme with ethanol-treated rat liver phosphatase restores antibody affinity showing that the catalytically inactivating subset of antibody exhibits marked or complete affinity for the active enzyme over the ATP,Mg++-inactivated form. This means that immunoinactivation using this antibody is not a valid way of measuring changes in the specific activity of the enzyme via phosphorylation-dephosphorylation. Preference for the active enzyme has not been obvious because when different amounts of enzyme activity are used in immunotitrations of samples of low activity, apparent differences in specific activity are observed when none actually exist. If precautions are not taken, results are obtained supporting phosphorylation by using an antibody that is not capable of distinguishing it.
Similar content being viewed by others
References
Arebalo, R. E., Hardgrave, J. E., Noland, B. J., and Scallen, T. J. (1980).Proc. Natl. Acad. Sci. USA 77 6429–6433.
Arebalo, R. E., Tormanen, C. D., Hardgrave, J. E., Noland, B. J., and Scallen, T. J. (1982a).Proc. Natl. Acad. Sci. USA 79 51–55.
Arebalo, R. E., Hardgrave, J. E., and Scallen, T. J. (1982b).Fed. Proc. 41 1398.
Brandt, H., Capulong, Z. L., and Lee, E. Y. C. (1975).J. Biol. Chem. 250 8038–8044.
Beg, Z. H., and Brewer, H. B. (1982).Fed. Proc. 41 2634–2638.
Beg, Z. H., Stonik, J. A., and Brewer, H. B. (1978).Proc. Natl. Acad. Sci. USA 75 3678–3682.
Beg, Z. H., Stonik, J. A., and Brewer, H. B. (1980).J. Biol. Chem. 255 8541–8545.
Dugan, R. E., Baker, T. A., and Porter, J. W. (1982).Eur. J. Biochem. 125 497–503.
Edwards, P. A., Lemongello, D., and Fogelman, A. M. (1979).Biochim. Biophys. Acta 574 123–135.
Edwards, P. A., Lemongello, D., Kane, J., Shechter, I., and Fogelman, A. M. (1980).J. Biol. Chem. 255 3715–3725.
Gil, G., Sitges, M., Bove, J., and Hegardt, F. G. (1980).FEBS Lett. 110 195–199.
Gil, G., Sitges, M., and Hegardt, F. G. (1981).Arch. Biochem. Biophys. 210 224–229.
Goldfarb, S., and Pitot, H. C. (1971).J. Lipid Res. 12 512–515.
Hardgrave, J. E., Heller, R. A., Herrera, M. G., and Scallen, T. J. (1979).Proc. Natl. Acad. Sci. USA 76 3834–3838.
Jenke, H.-S., Lowel, M., and Berndt, J. (1981).J. Biol. Chem. 256 9622–9625.
Keith, M. L., Rodwell, V. W., Rogers, D. H., and Rudney, H. (1979).Biochem. Biophys. Res. Commun. 90 969–975.
Kleinsek, D. A., Jabalquinto, A. M., and Porter, J. W. (1980).J. Biol. Chem. 255 3918–3923.
Kleinsek, D. A., Dugan, R. E., Baker, T. A., and Porter, J. W. (1981).Methods Enzymol. 71 462–479.
Ness, G. C. (1983).Mol. Cell. Biochem. 53/54 299–306.
Ness, G. C., Spindler, C. D., and Benton, G. A. (1980).J. Biol. Chem. 255 9013–9016.
Ness, G. C., Benton, G. A., Deiter, S. A., and Wickham, P. S. (1982).Arch. Biochem. Biophys. 214 705–713.
Author information
Authors and Affiliations
Additional information
J. W. Porter died June 27th, 1984 after a valiant struggle against cancer.
Rights and permissions
About this article
Cite this article
Dugan, R.E., Porter, J.W. Antibody preference for the catalytically active form of β-hydroxy-β-methylglutaryl coenzyme A reductase. J Bioenerg Biomembr 16, 551–560 (1984). https://doi.org/10.1007/BF00743245
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00743245