Abstract
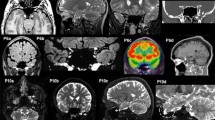
⊎ Background: The ability to name objects depends partly on visual perception. We used positron emission tomography (PET) to measure activity-related changes in regional cerebral blood flow (r-CBF) in order to identify regions of the brain activated during visual object naming. ⊎ Methods: Four right-handed volunteers were recruited. Following an intravenous injection of15O-labeled water, r-CBF was measured during visual object naming, counting numbers, and resting. PET and MRI images were coregistered, the size of the brain was proportionally adjusted in each axis to Talairach's and Tournoux's atlas, and the comparison of stimulated versus resting blood flow images revealed activated brain regions. ⊎ Results: In the subtraction of resting from naming, activation was observed in the bilateral primary visual cortex, bilateral fusiform gyrus, left lingual gyrus, bilateral inferotemporal cortex, bilateral inferior frontal gyrus, bilateral precentral gyrus, anterior cingulate gyrus, left parietal operculum, and left putamen. In the subtraction of counting from naming, most of the those areas were activated, but no significant activity was observed in the left lingual gyrus, left parietal operculum, or bilateral precentral gyrus (motor cortex). The areas activated with the paradigm included those dedicated to visual perception (primary and associate visual cortex), visual recognition (inferior temporal cortex), and phonological output (Broca's area). ⊎ Conclusion: Our results indicated that the major neural network from occipital lobe to frontal cortex, which is mainly involved in the ventral visual pathway, demonstrated activation in these tasks. Result of this study will serve as base line data for analyzing the findings in patients with impaired visual perception.
Similar content being viewed by others
References
Alexander MP (1990) Broca's area aphasias: aphasia after lesions including the frontal operculum. Neurology 40:353–362
Bender MB, Feldman M (1972) The so-called “visual agnosias.” Brain 95:173–186
Dejerine J (1891) Sur un cas de cecite verbal avec agraphie, suivie d'autopsie. C R Soc Biol (Paris) 3:197–201
Demonet JF (1993) Language functions explored in normal subjects by positron emission tomography: a critical review. Hum Brain Mapping 1:39–47
Demonet JF, Chollet F, Ramsay S, Cardebat D, Nespoulous JL, Wise R, Rascol A, Frackowiak R (1992) The anatomy of phonological and semantic processing in normal subjects. Brain 115:1753–1768
Fox PT, Perlmutter JS, Raichle ME (1985) A stereotactic method of anatomical localization for positron emission tomography. J Comput Assist Tomogr 9:141–153
Fox PT, Mintum MA, Raichle ME (1986) Mapping human visual cortex with positron emission tomography. Nature 323:806–809
Friston KJ, Frith CD, Liddle PF, Frackowiak RSJ (1991) Comparing functional (PET) images: the assessment of significant change. J Cereb Blood Flow Metab 11:690–699
Fujita I, Tanaka K, Minami I, Cheng K (1992) Columns for visual features of objects in monkey inferotemporal cortex. Nature 360:343–346
Haxby JV, Grady CL, Horwitz B, Ungerleider LG, Mishkin M, Carson RE, Herscovitch P, Schapiro MB, Rapoport SI (1991) Dissociation of object and spatial visual processing pathways in human extrastriate cortex. Proc Natl Acad Sci USA 88:1621–1625
Herscovitch P, Markham J, Raichle ME (1983) Brain blood flow measured with intravenous H2 15O. I. Theory and error analysis. J Nucl Med 24:782–789
Hinke RM, Hu X, Stillman AE, Kim SG, Merkle H, Salmi R, Ugurbil K (1993) Functional magnetic resonance imaging of Broca's area during internal speech. Neuroreport 4:675–678
Holms G (1918) Disturbances of vision by cerebral lesions. Br J Ophthalmol 2:353–384
Horton JC, Hoyt WF (1991) The representation of the visual field in human striate cortex. A revision of classic Holms map. Arch Ophthalmol 109:816–824
Iida H, Miura S, Kanno I, Murakami M, Yamamoto S, Amano M (1989) Design and elevation of Headtome-IV, a whole body positron emission tomograph, IEEE Trans Nucl Sci NS-37:1006–1010
Iwai E, Yukie M (1988) A direct projection from hippocampal field CAI to ventral area TE of inferotemporal cortex in the monkey. Brain Res 444:397–401
Kertesz A (1980) Western aphasia battery. University of Western Ontario Press, London, Ontario
Kikuchi R, Iwai E (1980) The locus of the posterior subdivision of the inferotemporal visual learning area in the monkey. Brain Res 198:347–360
Kiyosawa M, Itoh M, Nakagawa Y. Kobayashi N, Tamai M (1995) Effect of kanji and kana reading on cerebral blood flow patterns measured by PET. Jpn J Ophthalmol 39:198–205
Kiyosawa M, Mizuno K, Hatazawa J, Fukuda H, Yamada K, Ito M (1986) Metabolic mapping in hemianopsia using positron emission tomography with 18F-deoxyfluoroglucose. Am J Ophthalmol 101:310–319
Le Bihan D, Turner R, Zeffiro TA, Cuenod CA, Jezzard P, Bonnerot V (1993) Activation of human primary visual cortex during visual recall. A magnetic resonance imaging study. Proc Natl Acad Sci USA 90:11802–11805
Mesulam MM (1981) A cortical network for directed attention and unilateral neglect. Ann Neurol 10:309–325
Pardo JV, Pardo PJ, Janer KW, Raichle ME (1990) The anterior cingulate cortex mediates processing selection in the Stroop attentional conflict paradigm. Proc Natl Acad Sci USA 87:256–259
Petersen SE, Fox PT, Posner MI, Mintun M, Raichle ME (1988) Positron emission tomographic studies of the cortical anatomy of single-word processing. Nature 331:585–589
Posner MI, Petersen SE, Fox PT, Raichle ME (1988) Localization of cognitive operations in the human brain. Science 240:1627–1631
Salmelin R, Hari R, Lounasmaa OV, Sams M (1994) Dynamics of brain activation during picture naming. Nature 368:463–465
Senda M (1994) Mapping cortical functions using PET activation technique. In: Sugishita M (ed) New horizons in neuropsychology. Elsevier Science, Amsterdam, pp 23–34
Senda M, Kanno I, Yonekura Y, Fujiota H, Ishii K, Lyshkow H, Miura S, Oda K, Sadato N, Toyama H (1994) Comparison of anatomical standardization methods regarding the sensorimotor foci localization and between-subject variation in H2 15O PET activation: a three-center collaboration study. Ann Nucl Med 8:201–207
Sergent J, Ohta S, MacDonald B (1992) Functional neuroanatomy of face and object processing. Brain 115:15–36
Talairach J, Tournoux P (1988) A coplanar stereotactic atlas of human brain. Thieme, New York
Wise R, Chillet F, Hadar U, Friston K, Hoffner E, Frackowiak R (1991) Distribution of cortical neural networks involved in word comprehension and word retrieval. Brain 114:1803–1817
Zeki SM (1978) Functional specialization in the visual cortex of the rhesus monkey. Nature 274:423–428
Zeki SM (1993) The representation of the retina in the primary visual cortex. In: A vision of the brain. Blackwell, Oxford, pp 22–32
Zeki SM (1993) Functional specialization in human visual cortex. In: A vision of the brain. Blackwell, Oxford, pp 131–141
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kiyosawa, M., Inoue, C., Kawasaki, T. et al. Functional neuroanatomy of visual object naming: a PET study. Graefe's Arch Clin Exp Ophthalmol 234, 110–115 (1996). https://doi.org/10.1007/BF00695250
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00695250