Abstract
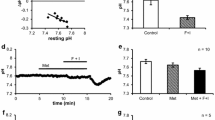
Intracellular pH (pHi) in the perfused rat mandibular gland was determined from the distribution of DMO (5,5-dimethyl-2,4-oxazolidinedione). In unstimulated glands, pHi averaged 7.12±0.02. Stimulation with a “standard” (submaximal) concentration (0.3 μmol/l) of acetylcholine (ACh) caused a fall in pHi to 6.81±0.06 over 60 min, but a maximal concentration (1.0 μmol/l) caused an initial rise in pHi to 7.60±0.02, followed by a fall to 7.45±0.02 over 60 min. After replacement of perfusate Cl with gluconate, the standard ACh concentration caused a rise in pHi to 7.50±0.02 followed by a fall to 7.27±0.04 after 60 min, concomitant with a 76% fall in secretory rate and a rise in salivary HCO3 concentration from 14±0.9 to 67±1.5 mmol/l. Furosemide (1 mmol/l) had a similar effect to gluconate replacement except that secretory rate fell only by 60%. Bumetanide (1 mmol/l), which inhibited secretion by 67%, did not cause pHi to rise following ACh stimulation but prevented the fall seen with ACh alone. Acetazolamide and methazolamide (1 mmol/l) had no effect on the salivary secretory response to ACh but they caused pHi to rise, respectively, to 7.20±0.03 and 7.43±0.02. Bumetanide and methazolamide together caused pHi to rise to 7.58±0.02 and reduced the secretory response to ACh by 91%. The disulfonic stilbene, SITS, caused pHi to rise to 7.26±0.03. Ouabain and amiloride both caused resting pHi to fall closer to equilibrium and largely abolished the gland's responsiveness to ACh. The results suggest that rat mandibular secretion depends both on a Na−K−2Cl symport and on Na−H and Cl−HCO3 antiports present in the basolateral membranes of the secretory cells in proportions of about 8 (Na−K−2Cl symport) to 5 (Na−H antiport) to 3 (Cl−HCO3 antiport).
Similar content being viewed by others
References
Cabantchik ZI, Knauf P, Rothstein A (1978) The anion transport system of the red blood cells: the role of membrane protein evaluated by the use of “probes”. Biochim Biophys Acta 515:239–302
Candia OA, Schoen HF, Low L, Podos SM (1981) Chloride transport inhibition by piretanide and MK-196 in bullfrog corneal epithelium. Am J Physiol 240:F25-F29
Case RM, Conigrave AD, Novak I, Young JA (1980) Electrolytes and protein secretion by the perfused rabbit mandibular gland stimulated with acetylcholine or catecholamines. J Physiol (Lond) 300:467–487
Case RM, Conigrave AD, Favaloro EJ Novak I, Thompson CH, Young JA (1982) The role of buffer anions and protons in secretion by the rabbit mandibular salivary gland. J Physiol (Lond) 322:273–286
Case RM, Hunter M, Novak I, Young JA (1984) The anionic basis of fluid secretion by the rabbit mandibular gland. J Physiol (Lond) 349:619–630
Cho AK, Curry SH, Jacobsen S (1969) Localization of basic drugs in the submaxillary gland. Biochem Pharmacol 18:2323–2330
Evans LAR, Pirani DP, Cook DI, Young JA (1986) Intraepithelial current flow in rat pancreatic secretory epithelia. Pflügers Arch 407 (Suppl 2):5107–5111
Evans MG, Marty A, Tan YP, Trautmann A (1986) Blockage of Ca-activated Cl conductance by furosemide in rat lacrimal glands. Pflügers Arch 406:65–68
Gard G, Christie JA, Cook DI, Young JA (1985) The action of loop diuretics on the submandibular anion self-exchange antiport. Proc Aust Physiol Pharmacol Soc 16:25P
Geck P, Pietrzyk C, Burckhardt BC, Pfeiffer B, Heinz E (1980) Electrically silent cotransport of Na, K, Cl in Ehrlich cells. Biochim Biophys Acta 600:432–447
Greger R, Schlatter E (1984) Mechanism of NaCl secretion in rectal gland of spiny dogfish (Squalus acanthias). I. Experiments in isolated in vitro perfused rectal gland tubules. Pflügers Arch 402:63–75
Greger R, Schlatter E (1984) Mechanism of NaCl secretion in rectal gland tubules of spiny dogfish (Squalus acanthias). II. Effect of inhibitors. Pflügers Arch 402:364–375
Greger R, Schlatter E, Wang F, Forrest JN (1984) Mechanism of NaCl secretion in rectal gland tubules of spiny dogfish (Squalus acanthias). III. Effect of stimulation of secretion by cyclic AMP. Pflügers Arch 402:376–384
Greger R, Schlatter E, Gögelein H (1985) Cl−-channels in the apical cell membrane of the rectal gland “induced” by cAMP. Pflügers Arch 406:446–448
Knickelbein R, Aronson PS, Schron CM, Seifter J, Dobbins JW (1985) Sodium and chloride transport across rabbit ileal brush border. II. Evidence for Cl−HCO3 exchange and mechanism of coupling. Am J Physiol 249:G236-G245
Martin CJ, Frömter E, Gebler E, Knauf B, Young JA (1973) The effects of carbachol on water and electrolyte fluxes and transepithelial electrical potential differences of the rabbit submaxillary main duct perfused in vitro. Pflügers Arch 341:131–142
Martinez JR, Cassity N (1985)36Cl fluxes in dispersed rat submandibular acini: effects of acetylcholine and transport inhibitors. Pflügers Arch 403:50–54
Murakami M, Imai Y, Seo Y, Morimoto T, Shiga K, Watari H (1983) Phosphorus nuclear magnetic resonance of perfused salivary gland. Biochim Biophys Acta 762:19–24
Murakami M, Novak I, Young JA (1986) The effects of choline on fluid secretion by the isolated perfused mandibular gland of the rat. Am J Physiol 251:G84-G89
Novak I, Young JA (1984) The effect of transport inhibitors and anion replacement on salivary secretion. In: Case RM, Lingard JM, Young JA (eds) Secretion: mechanisms and control. Manchester University Press, Manchester, pp 81–87
Novak I, Young JA (1986) Two independent anion transport systems in rabbit mandibular salivary glands. Pflügers Arch 407:649–656
Novak I, Dave C, Young JA (1984) The anionic basis of secretion by rat and rabbit mandibular glands. In: Case RM, Lingard JM, Young JA (eds) Secretion: mechanisms and control. Manchester University Press, Manchester, pp 77–80
Petersen OH (1971) Formation of saliva and potassium transport in the perfused cat submandibular gland. J Physiol (Lond) 216:129–142
Petersen OH (1980) The electrophysiology of gland cells. Academic Press, London
Petersen OH, Maruyama Y (1984) Calcium-activated potassium channels and their role in secretion. Nature 307:693–696
Poulsen JH, Kristensen LØ (1982) Is stimulation-induced uptake of sodium in rat parotid acinar cells mediated by a sodium/chloride co-transport system? In: Case RM, Garner A, Turnberg LA, Young JA (eds) Electrolyte and water transport across gastrointestinal epithelia. Raven, New York, pp 199–208
Putney JW, Borzelleca JF (1971) On the mechanism of14C-nicotine distribution in rat submaxillary gland in vitro. J Pharmacol Exp Ther 178:180–191
Putney JW, Borzelleca JF (1971) On the mechanisms of14C-salicylic acid distribution in rat submaxillary gland in vitro. J Pharmacol Exp Ther 177:263–275
Roos A, Boron WF (1981) Intracellular pH. Physiol Rev 61:296–434
Semenza G, Kinne R (eds) (1985) Membrane transport driven by ion gradients (Annals of the New York Academy of Sciences, vol 456), New York Academy of Sciences, New York
Seow FKT, Lingard JM, Young JA (1986) The anionic basis of fluid secretion by rat pancreatic acini in vitro Am J Physiol 250:G140-G148
Silva P, Stoff J, Field M, Fine L, Forrest JN, Epstein FH (1977) Mechanism of active chloride secretion by shark rectal gland: role of Na−K-ATPase in chloride transport. Am J Physiol 233:F298-F306
Suzuki K, Petersen OH (1985) The effect of Na+ and Cl− removal and of loop diuretics on acetylcholine-evoked membrane potential changes in mouse lacrimal acinar cells. Q J Exp Physiol 70:437–445
Tamarin A, Sreebny LM (1965) The rat submaxillary gland. A correlative study by light and electron microscopy. J Morphol 117:295–352
Vogh BP, Langham MR (1981) The effect of furosemide and bumetanide on cerebrospinal fluid formation. Brain Res 221:171–183
Waddell WJ, Butler TC (1959) Calculation of intracellular pH from the distribution of 5,5-dimethyl-2,4-oxazolidinedione (DMO). Application to skeletal muscle of the dog. J Clin Invest 38:720–729
Waugh WH (1977) Photometry of inulin and polyfructosan by the use of a cysteine/tryptophan reaction. Clin Chem 23:639–645
Welsh MJ (1983) Inhibition of chloride secretion by furosemide in canine tracheal epithelium. J Membr Biol 71:219–226
Young JA, Martin CJ, Asz M, Weber FD (1970) A microperfusion investigation of bicarbonate secretion by the rat submaxillary gland. The action of a parasympathomimetic drug on electrolyte transport. Pflügers Arch 319:185–199
Young JA, Cook DI, Lennep EW van, Roberts ML (1986) Secretion by the major salivary glands. In: Johnson L, Christensen J, Jackson M, Jacobson E, Walsh J (eds) Physiology of the gastrointestinal tract, 2nd edn. Raven Press, New York
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pirani, D., Evans, L.A.R., Cook, D.I. et al. Intracellular pH in the rat mandibular salivary gland: the role of Na−H and Cl−HCO3 antiports in secretion. Pflugers Arch. 408, 178–184 (1987). https://doi.org/10.1007/BF00581349
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00581349