Abstract
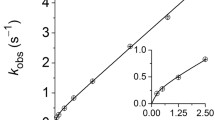
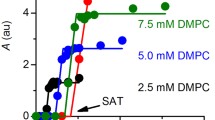
The binding of inorganic mercury Hg(II) to phospholipid headgroups has been investigated by phosphorus-31 nuclear magnetic resonance of phosphatidylethanolamine (PE), phosphatidylserine (PS) and phosphatidylcholine (PC) in water micellar and multilamellar phases. HgCl2 triggers the aggregation of phospholipid micelles, leading to a lipid-mercury precipitate that is no longer detectable by high-resolution31P-NMR. The remaining signal area corresponds to micelles in the soluble fraction and is a non-linear function of the initial mercury-to-lipid molar ratio. Kinetics of micelle aggregation are exponential for the first 15 min and show a plateau tendency after 120 min. Apparent Hg(H) affinities for phospholipid headgroups are in the order: PE > PS > PC. The same binding specificity is observed when HgCl2 is added to (1:1) mixtures of different micelles (PE + PC; PS + PC). However, mercury binding to mixed micelles prepared with two lipids (PE/PC or PS/PC) induces the aggregation of both lipids. Hg(II) also leads to a31P-NMR chemical shift anisotropy decrease of PC, PS and mixed (1:1) PE/PC multilamellar vesicles and markedly broadens PS spectra. This indicates that HgCl2 binding forces phospholipid headgroups to reorient and that the concomitant network formation leads to a slowing down of PS membrane collective motions. Formation of a gel-like lamellar phase characterized by a broad NMR linewidth is also observed upon HgCl2 binding to PE samples both in fluid (Lα) or hexagonal (HII) phases. The PE hexagonal phase is no longer detected in the presence of HgCl2. Mixed PE/PC dispersions remain in the fluid phase upon mercury addition, indicating that no phase separation occurs. Addition of excess NaCl leads to the appearance of the non-reactive species HgCl sup2−inf4 and induces the reversal of all the above effects.
Similar content being viewed by others
Abbreviations
- A(t):
-
time-dependence of peak area
- A40 :
-
peak area at t=40 min
- 1/β:
-
rate of peak area decrease
- Δδ:
-
isotropic chemical shift
- δ:
-
isotropic chemical shift change
- Δσ:
-
chemical shift anisotropy
- DPPC:
-
dipalmitoylphosphatidylcholine
- Hg(II):
-
inorganic mercury
- NMR:
-
nuclear magnetic resonance
- pCl:
-
−log [Cl−]
- PC:
-
phosphatidylcholine
- PE:
-
phosphatidylethanolamine
- PL:
-
phospholipid
- PS:
-
phosphatidylserine
- Ri :
-
mercury-to-lipid molar ratio
- MLV:
-
multilamellar vesicles
- SUV:
-
small unilamellar vesicles
References
Bevan Dr, Worrel WJ, Barfield KD (1983) The interaction of Ca2+, Mg2+, Zn2+, Cd2+, and Hg2+ with phospholipid bilayer vesicles. Colloids Surf 6:365–376
Boadi WY, Urbach J, Brandes JM, Yannai S (1992) In vitro exposure to mercury and cadmium alters term human placental membrane fluidity. Toxicol Appl Pharmacol 116:17–23
Burnell EE, Cullis PR, de Kruijff B (1980) Effects of tumbling and lateral diffusion on phosphatidylcholine model membrane31P-NMR lineshapes. Biochim Biophys Acta 603:63–69
Chávez E, Holguín JA (1988) Mitochondrial calcium release as induced by Hg2+. J Biol Chem 263:3582–3587
Clarkson TW (1994) The toxicology of mercury and its compounds. In: Watras CJ and Huckabee JW (eds) Mercury pollution, integration and synthesis. Lewis Publishers, Boca Raton, pp 631–641
Cullis PR, de Kruijff B (1978) The polymorphic phase behaviour of phosphatidylethanolamines of natural and synthetic origin. Biochim Biophys Acta 513:31–42
Delnomdedieu M, Boudou A, Desmazès JP, Georgescauld D (1989) Interaction of mercury chloride with the primary amine group of model membranes containing phosphatidylserine and phosphatidylethanolamine. Biochim Biophys Acta 986:191–199
Delnomdedieu M, Boudou A, Georgescauld D, Dufourc EJ (1992) Specific interactions of mercury chloride with membranes and other ligands as revealed by mercury-NMR. Chem-Biol Interact 81:243–269
Delnomdedieu M, Allis JW (1993) Interaction of inorganic mercury salts with model and red cell membranes: importance of lipid binding sites. Chem-Biol Interact 88:71–87
Dennis EA, Plückhthun A (1984) Phosphorus-31 NMR of phospholipids in micelles. In: Gorenstein DG (ed) Phosphorus-31 NMR: principles and applications. Academic Press, London, pp 423–446
Dufourc EJ, Mayer C, Stohrer J, Althoff G, Kothe G (1992) Dynamics of phosphate head group in biomembranes. Biophys J 61:42–57
Farrell RE, Germida JJ, Ming Huang PM (1993) Effects of chemical speciation in growth media on the toxicity of mercury(II). Appl Environm Microbiol 59:1507–1514
Foulkes EC, Bergman D (1993) Inorganic mercury absorption in mature and immature rat jejunum: transcellular and intercellular pathways in vivo and in everted sacs. Toxicol Appl Pharmacol 120:89–95
Gutknecht J (1981) Inorganic mercury (Hg2+) transport through lipid bilayer membranes. J Membr Biol 61:61–66
Kinter WB, Pritchard JB (1977) Altered permeability of cell membranes. In: Handbook of Physiology (section 9): Reactions to environmental agents. Williams and Wilkins, London, pp 563–573
Petersheim M, Sun J (1989) On the coordination of La3+ by phosphatidylserine. Biophys J 55:631–636
Rajan S, Kang SY, Gutowsky HS, Oldfield E (1981) Phosphorus nuclear magnetic resonance study of membrane structure. J Biol Chem 256:1160–1166
Ralston GB, Crisp EA (1981) The action of organic mercurials on the erythrocyte membrane. Biochim Biophys Acta 649:98–104
Rance M, Byrd RA (1983) Obtaining high-fidelity spin-1/2 powder spectra in anisotropic media: phase-cycled Hahn echo spectroscopy. J Magn Res 52:221–240
Ribarov SR, Benov LC (1981) Relationship between the hemolytic action of heavy metals and lipid peroxidation. Biochim Biophys Acta 640:721–726
Rothstein A (1976) Cell membrane as site of action of heavy metals. In: Brönner F, Kleinzeller A (ed) Current topics in membrane transport. Academic Press, New York, pp 1–76
Rothstein A (1981) Mercurials and red cell membranes. In: The function of red blood cells: erythrocyte pathobiology. Alan R Liss, New York, pp 105–131
Schanck A, Deleers M (1993)31P-NMR study of the parameters influencing the formation of non-bilayer phases in model membrane. Biochem Biophys Res Common 195:654–658
Schecher WD, McAvoy DC (1991) Mineql. 2.23 program. Environmental Research Software, Cincinnati
Seelig J (1978)31P nuclear magnetic resonance and the head group structure of phospholipids in membranes. Biochim Biophys Acta 515:105–140
Sharma DC, Davis PS, Sharma PK (1981) Studies in search of modifiers of the toxicity of mercurials and speculations on its biochemical mechanism. Biochem Pharmacol 30:3105–3107
Shinada M, Muto H, Takizawa Y (1991) In vitro interaction of selected phospholipid species with mercuric chloride using Fourier transform1H-NMR, Bull Environm Contam Toxicol 47:350–354
Singleton WS, Gray MS, Brown ML, White JL (1965) Chromatographically homogenous lecithin from egg phospholipids. J Am Oil Chem Soc 42:53–56
Stáry J, Kratzer K (1989) Mechanism of the cumulation of mercury species in algaHydrodictyon reticulatum. Toxicol Environm Chem 22:197–202
Tacnet F, Ripoche P, Roux M, Neumann JM (1991)31P-NMR study of pig intestinal brush-border membrane structure: effect of zinc and cadmium ions. Eur Biophys J 19:317–322
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Girault, L., Lemaire, P., Boudou, A. et al. Interactions of inorganic mercury with phospholipid micelles and model membranes. A31P-NMR study. Eur Biophys J 24, 413–421 (1996). https://doi.org/10.1007/BF00576713
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00576713