Summary
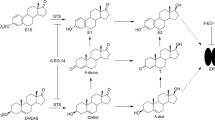
The antitumor activity of the new irreversible aromatase inhibitor 6-methylenandrosta-1,4-diene-3,17-dione (FCE 24304) was studied in rats with 7,12-dimethylbenzanthracene (DMBA)-induced tumors; several endocrine parameters were evaluated in these animals. The compound was given s.c. and p.o. twice daily, 6 days/week, for 4 weeks. The control group showed 13% tumor regressions (0% complete remission, CR; 13% partial remission, PR). FCE 24304 given s.c. induced 44% (22+22) regressions at the dose of 3 mg/kg per day, 70% (40+30) at 10 mg/kg per day, 73% (27+46) at 30 mg/kg per day, and 70% (50+20) at 100 mg/kg per day. FCE 24304 given orally induced 25% (17+8) tumor regressions at 30 mg/kg per day and 50% (17+33) at 100 mg/kg per day. Rats were killed 4 h after the last dose and the aromatase activity of ovarian microsomes (OAA) was evaluated. OAA was reduced by 56% after s.c. treatment with 3 mg/kg per day FCE 24304; complete OAA suppression (≥96%) was obtained starting at 10 mg/kg per day s.c. Oral treatment slightly reduced OAA only at a dose of 100 mg/kg per day (36%). Body weight increased in all the groups s.c. treated with FCE 24304 but not in those treated orally. The weights of the pituitary, adrenals, and uterus were reduced in rats treated s.c. with 10 and 30 mg/kg per day; at 100 mg/kg per day, a decrease in ovarian weight was observed while uterus weight was similar to that of controls. Oral FCE 24304 increased ovarian weight at a dose of 30 mg/kg per day but not at 100 mg/kg per day. Serum prolactin (PRL) and luteinizing hormone (LH) levels did not change. In conclusion, FCE 24304 given s.c. proved highly effective against DMBA-induced tumors in rats but had less activity when given orally. Its intrinsic androgenic activity, higher after s.c. than after oral treatment, could contribute to the antitumor effect in the intact (premenopausal) rat model.
Similar content being viewed by others
References
Brodie AMH (1985) Aromatase inhibition and its pharmacologic implications. Biochem Pharmacol 34: 3213
Brodie AMH, Schwarzel WC, Shaikh AA, Brodie HJ (1977) The effect of an aromatase inhibitor, 4-hydroxy-4-androstene-3,17-dione, on estrogen dependent processes in reproduction and breast cancer. Endocrinology 100: 1684
Brodie AMH, Garrett WM, Hendrickson JR, Morris-Tsai CH, Marcotte PA, Robinson CH (1981) Inactivation of aromatase in vitro by 4-hydroxy-4-androstene-3,17-dione and 4-acetoxy-4-androstene-3,17-dione and sustained effects in vivo. Steroids 38: 693
di Salle E, Giudici D, Ornati G, Briatico G, Lombardi P (1987) Endocrine properties of 6-methylenandrosta-1,4-diene-3,17-dione (FCE 24304), a new irreversible aromatase inhibitor. J Steroid Biochem 28 (Suppl): 146S (Abs D-015)
Geran RI, Greenberg NH, McDonald MM, Schumacher AM, Abbott BJ (1972) Protocol for screening chemical agents and natural products against animal tumors and other biological systems. Cancer Chemother Rep 3: 1
Giudici D, Ornati G, Briatico G, Buzzetti F, Lombardi P, di Salle E (1988) 6-Methylenandrosta-1,4-diene-3,17-dione (FCE 24304): a new irreversible aromatase inhibitor. J Steroid Biochem 30:391
Goss PE, Powles TJ, Dowsett M, Hutchison G, Brodie AMH, Gazet JC, Coombes RC (1986) Treatment of advanced post-menopausal breast cancer with an aromatase inhibitor 4-hydroxyandrostenedione: phase II report. Cancer Res 46: 4823
Harris AL (1985) Aminoglutethimide—a new endocrine therapy in breast cancer. Exp Cell Biol 53: 1
Henderson D, Norbisrath G, Kerb V (1986) 1-Methyl-1,4-androstadiene-3,17-dione (SH 489): characterization of an irreversible inhibitor of estrogen biosynthesis. J Steroid Biochem 24: 303
Johnston JO, Wright CL, Metcalf BW (1984) Biochemical and endocrine properties of a mechanism-based inhibitor of aromatase. Endocrinology 115: 776
Lowry OH, Rosenbrough NJ, Farr AL, Randall LJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193: 265
Rocheforf H, Garcia M (1980) Interactions and actions of androgens on the estrogen receptor. In: Genazzani E, Di Carlo F, Mainwaring WIP (eds) Pharmacological modulation of steroid action. Raven Press, New York, p 75
Santen RJ (1986) Aromatase inhibitors for treatment of breast cancer: current concepts and new perspectives. Breast Cancer Res Treat 7 (Suppl): 23
Santen RJ, Misbin RI (1981) Aminoglutethimide: review of pharmacology and clinical use. Pharmacotherapy 1: 95
Thompson EA, Siiteri PK (1974) Utilization of oxygen and reduced nicotinamide adenine dinucleotide phosphate by human placental microsomes during aromatization of androstenedione. J Biol Chem 249: 5364
Wilkinson JR, Williams JC, Singh D, Goss PE, Easton D, Coombes RC (1986) Response of N-nitrosomethylurea-induced rat mammary tumor to endocrine therapy and comparison with clinical response. Cancer Res 46: 4862
Wing LY, Garrett WM, Brodie AMH (1985) Effects of aromatase inhibitors, aminoglutethimide, and 4-hydroxyandrostenedione on cyclic rats and rats with 7,12-dimethylbenz(a)anthracene-induced mammary tumors. Cancer Res 45: 2425
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zaccheo, T., Giudici, D., Lombardi, P. et al. A new irreversible aromatase inhibitor, 6-methylenandrosta-1,4-diene-3,17-dione (FCE 24304): antitumor activity and endocrine effects in rats with DMBA-induced mammary tumors. Cancer Chemother. Pharmacol. 23, 47–50 (1989). https://doi.org/10.1007/BF00258457
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00258457