Summary
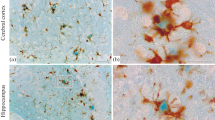
The immunohistochemical localization of the proteinase inhibitor region of amyloid protein precursors (APPI) in the postmortem human neocortex was studied using a polyclonal antibody raised against a purified recombinant human APPI derivative produced by COS-1 cells. APPI-like immunoreactivity (APPI-LI) was found diffusely in the human neocortex. APPI-LI appeared as irregularly shaped granular structures. The size of the APPI-LI structures was 1–4 μm in diameter. APPI-LI usually formed a cluster of 10- to 20-μm diameter in the cortical gray matter and 20- to 40-μm diameter in the subcortical white matter. Double staining for APPI and glial fibrillary acidic protein indicated that APPI-LI in the white matter and molecular layer was localized exclusively in the fibrillary astrocytes. In contrast, APPI-LI was found in neurons as well as in the fibrillary astrocytes in layers II through to VI. Under fluorescence microscopy, APPI-LI in both neurons and fibrillary astrocytes were found in close association with lipofuscin. The present observations indicate that APPI is localized in neurons and astrocytes in the human neocortex and that APPI may be associated with lipofuscin or lysosome in the human neocortex.
Similar content being viewed by others
References
Arai H, Lee VM-Y, Otvos L Jr, Greenberg BD, Lowery DE, Sharma SK, Schmidt ML, Trojanowski JW (1990) Defined neurofilament, τ, and amyloid precursor protein epitopes distinguish Alzheimer from non-Alzheimer senile plaques. Proc Natl Acad Sci USA 87:2249–2253
Bahmanyar S, Higgins GA, Goldgaber D, Lewis DA, Morrison JH, Wilson MC, Shankar SK, Gajdusek DC (1987) Localization of amyloid β protein messenger RNA in brains from patients with Alzheimer's disease. Science 237:77–80
Bancher C, Grundke-Iqbal I, Iqbal K, Kim KS, Wisniewski HM (1989) Immunoreactivity of neuronal lipofuscin with monoclonal antibodies to the amyloid beta-protein. Neurobiol Aging 10:125–132
Beach TG, Tago H, Nagai T, Kimura H, McGeer PL, McGeer EG (1987) Perfusion-fixation of the human brain for immunohistochemistry: comparison with immersion fixation. J Neurosci Methods 19:183–192
Benowitz LI, Rodriguez W, Paskevich P, Mufson EJ, Schenk D, Neve RL (1989) The amyloid precursor protein is concentrated in neuronal lysosomes in normal and Alzheimer disease subjects. Exp Neurol 106:237–250
Bourne GH (1973) Lipofuscin. Prog Brain Res 40:187–201
Brunk U, Ericsson JLE (1972) Electron microscopical studies on rat brain neurons. Localization of acid phosphatase and mode of formation of lipofuscin bodies. J Ultrastruct Res 38:1–15
Cataldo AM, Nixon RA (1990) Enzymatically active lysosomal proteases are associated with amyloid deposits in Alzheimer brain. Proc Natl Acad Sci USA 87:3861–3865
Cataldo AM, Thayer CY, Bird ED, Wheelock TR, Nixon RA (1990) Lysosomal proteinase antigens are prominently localized within senile plaques of Alzheimer's disease: evidence for a neuronal origin. Brain 513:181–192
Cohen ML, Golde TE, Usiak MF, Younkin LH, Younkin SG (1988) In situ hybridization of nucleus basalis neurons shows increased β-amyloid mRNA in Alzheimer disease. Proc Natl Acad Sci USA 85:1227–1231
Cole GM, Masliah E, Huynh TV, DeTeresa R, Terry RD, Okuda C, Saitoh T (1989) An antiserum against amyloid β-protein precursor detects a unique peptide in Alzheimer brain. Neurosci Lett 100:340–346
Cole GM, Masliah E, Shelton ER, Chan HW, Terry RD, Saito T (1991) Accumulation of amyloid precursor fragment in Alzheimer plaques. Neurobiol Aging 12:85–92
Currie J, Barcikowska M, Wegiel J, Morys J, Wisniewski HM (1989) Antiserum raised against a subsequence of Alzheimer amyloid precursor protein selectively labels a subset of microglial cells. J Neuropathol Exp Neurol 48:328
Glenner GG, Wong CW (1984) Alzheimer's disease: initial report of the purification and characterization of a novel cerebrovascular amyloid protein. Biochem Biophys Res Commun 120:885–890
Goedert M (1987) Neuronal localization of amyloid beta protein precursor mRNA in normal human brain and in Alzheimer's disease. EMBO J 6:3627–3632
Higgins GA, Lewis DA, Bahamnyar S, Goldgaber D, Gajdusek DC, Young WG, Morrison JH, Wilson MC (1988) Differential regulation of amyloid-β-protein mRNA expression within hippocampal neuronal subpopulation in Alzheimer disease. Proc Natl Acad Sci USA 85:1297–1301
Joachim CL, Mori H, Selkoe DJ (1989) Amyloid β-protein deposition in tissues other than brain in Alzheimer's disease. Nature 341:226–230
Johnson SA, Pasinetti GM, May PC, Ponte PA, Cordell B, Finch CE (1988) Selective reduction of mRNA for the beta-amyloid precursor protein that lacks a Kunitz-type protease inhibitor motif in cortex from Alzheimer brains. Exp Neurol 102:264–268
Kang J, Lemaire HG, Unterbeck A, Salbaum JM, Masters CL, Grzeschik KH, Multhaup G, Beyreuther K, Muller-Hill B (1987) The precursor of Alzheimer's disease A4 protein resembles a cell-surface receptor. Nature 325:733–736
Kitaguchi N, Takahashi Y, Tokushima Y, Shiojiri S, Ito H (1988) Novel precursor of Alzheimer's amyloid protein shows protease inhibitory activity. Nature 331:530–532
Kitaguchi N, Tokushima Y, Oishi K, Takahashi Y, Shiojiri S, Nakamura S, Tanaka S, Kodaira R, Ito H (1990) Determination of amyloid protein precursors harboring active form of proteinase inhibitor domains in cerebrospinal fluid of Alzheimer's disease patients by trypsin-antibody sandwich ELISA. Biochem Biophys Res Commun 166:1453–1459
Kitaguchi N, Takahashi Y, Oishi K, Shiojiri S, Tokushima Y, Utsunomiya T, Ito H (1990) Enzyme specificity of proteinase inbibitor region in amyloid precursor protein of Alzheimer's disease: different properties compared with protease nexin I. Biochim Biophys Acta 1038:105–113
Kitaguchi N, Tokushima Y, Nakamura S, Urakami K, Tanaka S, Shimizu Y, Yamamoto S (1991) Amyloid protein precursors: their expressions in the brain and concentration in cerebrospinal fluid. In: Proceedings of the Eighth Workshop on Neurotransmitters and Diseases, Tokyo, June 1, Excerpta Medica, Amsterdam, pp 38–57
Lewis DA, Higgins GA, Young WG, Goldgaber D, Gajdusek DC, Wilson MC, Morrison JH (1988) Distribution of precusor amyloid-β-protein messenger RNA in human cerebral cortex: relationship to neurofibrillary tangles and neuritic plaques. Proc Natl Acad Sci USA 85:1691–1695
Masters CL, Simms G, Weinman NA, Multhaup G, McDonald BL, Beyreuther K (1985) Amyloid plaque core protein in Alzheimer's disease and Down's syndrome. Proc Natl Acad Sci USA 82:4245–4249
Nakamura S, Kawamata T, Akiguchi I, Kameyama M, Nakamura N, Wakata Y, Kimura H (1990) Expression of monoamine oxidase B activity in astrocytes of senile plaques. Acta Neuropathol 80:419–425
Ottersdorf T, Lawrence CF, Schenk DB, Liederburg I, Johnson-Wood KL, Beattie EC, Ward PJ, Blacher RW, Dovey HF, Sinha S (1989) The secreted form of the Alzheimer's amyloid precursor protein with the Kunitz domain is protease nexin-II. Nature 341:144–147
Palmert MR, Golde TE, Cohen ML, Kovacs DM, Tanzi RE, Gusella JF, Usiak MF, Younkin LH, Younkin SG (1988) Amyloid protein precursor messenger RNAs: differential expression in Alzheimer's disease. Science 241:1080–1084
Palmert MR, Usiak M, Mayeux R, Raskind M, Tourtellotte WW, Younkin SG (1990) Soluble derivatives of the amyloid protein precursor in cerebrospinal fluid: alterations in normal aging and in Alzheimer's disease. Neurology 40:1028–1034
Ponte P, Gonzalez-DeWhitt P, Schilling J, Miller J, Hsu D, Greenberg B, Davis K, Wallace W, Lieberburg I, Fuller F, Cordell (1988) A new A4 amyloid mRNA contains a domain homologous to serine protease inhibitors. Nature 331:525–527
Shivers BD, Hillbich C, Multhaup G, Salbaum M, Beyreuther K, Seeburg PH (1988) Alzheimer's disease amyloidogenic glycoprotein: expression pattern in rat brain suggests a role in cell contact. EMBO J 7:1365–1370
Siakotos AN, Watanabe I, Saito A, Fleischer S (1970) Procedures for the isolation of two distinct lipopigments from human brains: lipofuscin and ceroid. Biochem Med 4:361–375
Takahashi H, Kurashima C, Utsuyama M, Hirokawa K (1990) Immunohistological study of senile brains by using a monoclonal antibody recognizing β amyloid precursor protein: significance of granular deposits in relation with senile plaques. Acta Neuropathol 80: 260–265
Tanaka S, Nakamura S, Ueda K, Kameyama M, Shiojiri S, Takahashi Y, Kitaguchi N, Ito H (1988) Three types of amyloid precursor mRNA in human brain: their differential expressions in Alzheimer's disease. Biochem. Biophys Res Commun 157:472–479
Tanaka S, Shiojiri S, Takahashi Y, Kitaguchi N, Ito H, Kameyama M, Kimura J, Nakamura S, Ueda K (1989) Tissue-specific expression of three types of β-protein precursor mRNA: enhancement of protease inhibitor-harboring types in Alzheimer's disease brain. Biochem Biophys Res Commun 165:1406–1414
Tanzi RE, McClatchey AI, Lamperti ED, Villa-Komaroff L, Gusella JF, Neve RL (1988) Protease inhibitor domain encoded by an amyloid protein precursor mRNA associated with Alzheimer's disease. Nature 331:528–530
Tate-Ostroff B, Majocha RE, Marotta CA (1989) Identification of cellular and extracellular sites of amyloid precursor protein extracytoplasmic domain in normal and Alzheimer disease brains. Proc Natl Acad Sci USA 86:745–749
Van Nostrand WE, Wagner SL, Suzuki M, Choi BH, Farrow JS, Geddes JW, Cotman CW, Cunningham DD (1989) Protease nexin-II, a potent antichymotrypsin shows identity to amyloid β-protein precursor. Nature 341:546–549
Wisniewski KE, Maslinska D (1989) Immunoreactivity of ceroid lipofuscin storage pigment in Batten disease with monoclonal antibodies to the amyloid β-protein. N Engl J Med 320:256–257
Zimmermann K, Herget T, Salbaum JM, Schubert W, Hilbich C, Cramer M, Masters CL, Multhaup G, Kang J, Lemaire H-G, Beyreuther K, Starzinski-Powitz A (1988) Localization of the putative precursor of Alzheimer's disease-specific amyloid at nuclear envelopes of adult human muscle. EMBO J 7:367–372
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nakamura, S., Suenaga, T., Akiguchi, I. et al. Immunohistochemical localization of the proteinase inhibitor region of amyloid precursor proteins in the neocortex of Alzheimer's disease and aged controls. Acta Neuropathol 84, 244–249 (1992). https://doi.org/10.1007/BF00227816
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00227816