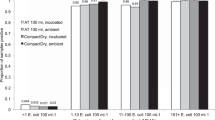
Abstract
Using biochemical and molecular methods, this research determined whether or not the H2S test did correctly identify sewage-contaminated waters by being the first to use culturing and molecular methods to identify the types and numbers of fecal indicator organisms, pathogens, and other microbes present in sewage samples with positive H2S test results. For the culture-based method, samples were analyzed for the presence of fecal bacteria by spread plating the sewage sample onto differential and selective media for Aeromonas spp., Escherichia coli, sulfite-reducing clostridia, H2S-producing bacteria, and Salmonella/Shigella spp. The isolates were then: (1) tested to determine whether they were H2S-producing organisms and (2) identified to the genus and species level using biochemical methods. The molecular method used to characterize the microbial populations of select samples was terminal restriction fragment length polymorphisms. These experiments on sewage provided evidence that positive H2S tests consistently contained fecal bacteria and pathogens. There were strong relationships of agreement between the organisms identified by both methods tested. This study is an important advance in microbial water quality detection since it is focused on the evaluation of a novel, low-cost, water microbiology test that has the potential to provide millions of people worldwide access to water quality detection technology. Of prime consideration in evaluating water quality tests is the determination of the test’s accuracy and specificity, and this article is a fundamental step in providing that information.




Similar content being viewed by others
References
Anwar MS, Chaudhry NA, Tayyab M (1999) Bacteriological quality of drinking water in Punjab: evaluation of H2S strip test. J Pak Med Assoc 49(10):237–241
Blackwood CB, Hudleston D, Zak DR, Buyer JS (2007) Interpreting ecological diversity indices applied to terminal restriction fragment length polymorphism data: insights from simulated microbial communities. Appl Environ Microbiol 73:5276–5283
Castillo G, Duarte R, Ruiz Z, Mauricio MT, Honorato B, Mercado R, Coloma V, Lorca V, Martins MT, Dutka BK (1994) Evaluation of disinfected and untreated drinking water supplies in Chile by the H2S paper strip test. Water Research 28:1765–1770
Committee on Indicators for Waterborne Pathogens (2004) Indicators for Waterborne Pathogens. National Academies Press, Washington
Genthe B, Franck M (1999) A tool for assessing microbial quality in small community water supplies: an H2S strip test. WRC Report 1. Water Research Commission, Division of Water, Environment and Forestry Technology, CSIR, Stellenbosch, South Africa
Gupta SK, Sheikh MA, Islam MS, Rahman KS, Jahan N, Rahman MM, Hoekstra RM, Johnston R, Ram PK, Luby S (2008) Usefulness of the hydrogen sulfide test for assessment of water quality in Bangladesh. J Appl Microbiol 2:388–395
Johnson MC, Devine AA, Ellis JC, Grunden AM, Fellner V (2009) Effects of antibiotics and oil on microbial profiles and fermentation in mixed cultures of ruminal microorganisms. J Dairy Sci 92(9):4467–4480
Kaspar P, Guillen I, Revelli D, Meza T, Velazquez G, Mino de Kaspar H, Pozolli L, Nunez C, Zoulek G (1992) Evaluation of a simple screening test for the quality of drinking water systems. Trop Med Parasitol 43(2):124–127
Kent AD, Smith DJ, Benson BJ, Triplett EW (2003) Web-based phylogenetic assignment tool for analysis of terminal restriction fragment length polymorphism profiles of microbial communities. Appl Environ Microbiol 69:6768–6776
Kim SH, Marsh TL (2004) The analysis of microbial communities with terminal restriction fragment length polymorphism (T-RFLP). In: Kowalchuk GA, de Bruijn FJ, Head IM, Akkermans AD, van Elsas JD (eds) Molecular microbial ecology manual, 2nd edn. Springer, New York, pp 789–808
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33:159–174
Liu WT, Marsh TL, Cheng H, Forney LJ (1997) Characterization of microbial diversity by determining terminal restriction fragment length polymorphisms of genes encoding 16S rRNA. Appl Environ Microbiol 63:4516–4522
Manja KS, Kaul RK (1992) Efficacy of a simple test for bacteriological quality of water. J Ind Assoc Environ Manage 19:18–20
Manja KS, Maurya MS, Rao KM (1982) A simple field test for the detection of faecal pollution in drinking water. Bull World Health Organ 60(5):797–801
Morales SE, Mouser PJ, Ward N, Hudman SP, Gotelli NJ, Ross DS, Lewis TA (2006) Comparison of bacterial communities in New England Sphagnum bogs using terminal restriction fragment length polymorphism (T-RFLP). Microb Ecol 52:34–44
Pant H, Sarfaraz S, Iyengar L (2002) Evaluation of l-cystine-amended H2S test for the detection of faecal pollution in potable water: comparison with standard multiple tube fermentation method. World Journal of Microbiology and Biotechnology 182:321–324
Pathak SP, Gopal K (2005) Efficiency of modified H2S test for detection of faecal contamination in water. Environ Monit Assess 108(1):59–65
Pidiyar VJ, Jangid K, Patole MS, Shouche YS (2004) Studies on cultured and uncultured microbiota of wild Culex quinquefasciatus mosquito midgut based on 16S ribosomal RNA gene analysis. Am J Trop Med Hyg 70(6):597–603
Ratto A, Dutka BJ, Vega C, Lopez C, El-Shaarawi A (1989) Potable water safety assessed by coliphage and bacterial tests. Water Research 23:253–255
Rijal GK, Fujioka RS, Ziel CA (2001) Synergistic effect of solar radiation and solar heating to disinfect drinking water sources. Water Sci Technol 43(12):155–162
Roser DJ, Ashbolt N, Ho G, Mathew K, Nair J, Ryken-Rapp D, Toze S (2005) Hydrogen sulphide production tests and the detection of groundwater faecal contamination by septic seepage. Water Sci Technol 51(10):291–300
Sobsey MD, Pfaender FK (2002) Evaluation of the H2S method for the detection of fecal contamination of drinking-water. WHO/SDE/WSH/02.08, World Health Organization, Geneva
Tewari S, Ramteke PW, Garg SK (2003) Evaluation of simple microbial tests for detection of fecal coliforms directly at 45.5°C. Environ Monit Assess 85:191–198
UNICEF/WHO (2009) Diarrhoea: why children are still dying and what can be done. UNICEF/WHO
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73(16):5261–5267
Widdel F (1988) Microbiology and ecology of sulfate- and sulfur-reducing bacteria. In: Zehnder AJB (ed) Biology of anaerobic organisms. Wiley-Liss, New York, pp 469–586
World Health Organization (2006). Guidelines for drinking-water quality: incorporating 1st and 2nd addenda, vol 1. Recommendations. http://www.who.int/entity/water_sanitation_health/dwq/fulltext.pdf
Acknowledgments
This work was supported by a Gillings Innovation Labs award from the UNC Gillings School of Global Public Health. Lanakila ‘Ku’ McMahan was supported by graduate fellowships from NSF and the US EPA. This article was developed under a STAR Fellowship Assistance Agreement No. FP-916907001 awarded by the U.S. Environmental Protection Agency (EPA). It has not been formally reviewed by EPA. The views expressed in this article are solely those of Lanakila McMahan, and EPA does not endorse any products or commercial services mentioned in this article. We also thank Hach Company and In Silico© for their generous support of this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McMahan, L., Devine, A.A., Grunden, A.M. et al. Validation of the H2S method to detect bacteria of fecal origin by cultured and molecular methods. Appl Microbiol Biotechnol 92, 1287–1295 (2011). https://doi.org/10.1007/s00253-011-3520-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-011-3520-z