Abstract
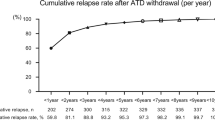
Antithyroid drugs may be proposed as the first-line therapy for hyperthyroidism due to Graves’ disease since some patients undergo prolonged remission after drug withdrawal. On the other hand, some studies, though controversial, indicated that methimazole (MMI) has some immunomodulating activity. We retrospectively analyzed 384 consecutive patients newly diagnosed with Graves’ disease in the years 1990–2002 to ascertain whether long-term therapy with low doses of MMI may prevent relapse of thyrotoxicosis. Two hundred and forty-nine patients were included in our study. The date of reduction of MMI dose to 5 mg/day was considered time 0 for survival analysis. In 121 MMI was discontinued in less than 15 months after time 0 (group D), while in the remaining 128 a daily MMI 2.5–5 mg dose was maintained (group M). One hundred and thirty-five patients were excluded for inadequate response to MMI, relapse of thyrotoxicosis that could be related to an improper withdrawal or reduction of MMI, inadequate or too short follow-up, iodide contamination, steroid or interferon therapy, pregnancy or post-partum. D and M groups did not differ for clinical and hormonal parameters except age, which was lower in D (p=0.019). Age > vs < 35 yr was relevant in survival analysis; therefore patients were divided in 2 groups according to this age cut-off. In younger patients relapse of thyrotoxicosis occurred in 15 patients of group D 2.4–39.6 months (median 19.0) after time 0, and 8 M after 5.9–40.0 (21.3) months, while 14 D and 5 M maintained euthyroidism until the end of the observation after 31.8–95.3 (56.6) months and 30.4–62.1 (46.5) months, respectively. Survival analysis indicated that the risk of relapse was similar in group D and M. In older patients relapse of thyrotoxicosis occurred in 40 patients of group D after 8.2–65.8 (25.4) months and 29 M after 5.8–62.5 (22.4) months, while 52 D and 86 M maintained euthyroidism until the end of the observation, 20.1–168.0 (46.7) months and 24.1–117.4 (53.4) months respectively. Survival analysis indicated that the risk of relapse was increased in group D. Therefore long-term treatment with low doses of MMI seems to prevent relapse in Graves’ disease in patients above 35 yr of age. This should be confirmed in a prospective study.
Similar content being viewed by others
References
Mori T, Sugawa H, Kosugi S, Ueda M, Hai N, Matsuda A. Recent trends in the management of Graves’ hyperthyroidism in Japan: opinion survey results, especially on the combination therapy of antithyroid drug and thyroid hormone. Endocr J 1997, 44: 509–17.
Cooper DS. Antithyroid drugs for the treatment of hyperthyroidism caused by Graves’ disease. Endocrinol Metab Clin North Am 1998, 27: 225–47.
Ljunggren JG, Törring O, Wallin G, et al. Quality of life aspects and costs in treatment of Graves’ hyperthyroidism with antithyroid drugs, surgery, or radioiodine: results from a prospective, randomized study. Thyroid 1998, 8: 653–9.
Escobar-Jiménez F, Férnandez-Soto ML, Luna-López V, Quesada-Charneco M, Glinoer D. Trends in diagnostic and therapeutic criteria in Graves’ disease in the last 10 years. Postgrad Med J 2000, 76: 340–4.
Weetman AP. Controversy in thyroid disease. J R Coll Physicians Lond 2000, 34: 374–80.
Zantut-Wittmann DE, Tambascia MA, da Silva Trevisan MA, et al. Antithyroid drugs inhibit in vivo HLA-DR expression in thyroid follicular cells in Graves’ disease. Thyroid 2001, 11: 575–80.
Montani V, Shong M, Taniguchi SI, et al. Regulation of major histocompatibility class II gene expression in FRTL-5 thyrocytes: opposite effects of interferon and methimazole. Endocrinology 1998, 139: 290–302.
Mozes E, Zinger H, Kohn LD, Singer DS. Spontaneous autoimmune disease in (NZB × NZW)F1 mice is ameliorated by treatment with methimazole. J Clin Immunol 1998, 18: 106–13.
Volpé R. Evidence that the immunosuppressive effects of antithyroid drugs are mediated through actions on the thyroid cell, modulating thyrocyte-immunocyte signaling: a review. Thyroid 1994, 4: 217–23.
Allannic H, Fauchet R, Orgiazzi J, et al. Antithyroid drugs and Graves’ disease: a prospective randomized evaluation of the efficacy of treatment duration. J Clin Endocrinol Metab 1990, 70: 675–9.
Tajiri J, Noguchi S, Morita M, Tamaru M, Marukami N. Antithyroid drug therapy for Graves’ hyperthyroidism: is long-term administration of a small maintenance dose necessary? Endocrinol Jpn 1991, 38: 223–7.
García-Mayor RV, Páramo C, Luna Cano R, Pérez Mendez LF, Galofré JC, Andrade A. Antithyroid drug and Graves’ hyperthyroidism. Significance of treatment duration and TRAb determination on lasting remission. J Endocrinol Invest 1992, 15: 815–20.
Maugendre D, Gatel A, Campion L, et al. Antithyroid drugs and Graves’ disease-prospective randomized assessment of long-term treatment. Clin Endocrinol 1999, 50: 127–32.
Benker G, Reinwein D, Kahaly G, et al. Is there a methimazole dose effect on remission rate in Graves’ disease? Results from a long-term prospective study The European Multicentre Trial Group of the Treatment of Hyperthyroidism with Antithyroid Drugs. Clin Endocrinol (Oxf) 1998, 49: 451–7.
McIver B, Rae P, Beckett G, Wilkinson E, Gold A, Toft A. Lack of effect of thyroxine in patients with Graves’ hyperthyroidism who are treated with an antithyroid drug. N Engl J Med 1996, 334: 220–4.
Lucas A, Salinas I, Rius F, et al. Medical therapy of Graves’ disease: does thyroxine prevent recurrence of hyperthyroidism? J Clin Endocrinol Metab 1997, 82: 2410–3.
Grebe SK, Feek CM, Ford HC, et al. A randomized trial of short-term treatment of Graves’ disease with high-dose carbimazole plus thyroxine versus low-dose carbimazole. Clin Endocrinol 1998, 48: 585–92.
Rittmaster RS, Abbott EC, Douglas R, et al. Effect of methimazole, with or without L-thyroxine, on remission rates in Graves’ disease. J Clin Endocrinol Metab 1998, 83: 814–8.
Pujol P, Osman A, Grabar S, et al.TSH suppression combined with carbimazole for Graves’ disease: effect on remission and relapse rates. Clin Endocrinol (Oxf) 1998, 48: 635–40.
Raber W, Kmen E, Waldhäusl W, Vierhapper H. Medical therapy of Graves’ disease: effect on remission rates of methimazole alone and in combination with triiodothyronine. Eur J Endocrinol 2000, 142: 117–24.
Glinoer D, de Nayer P, Bex M; Belgian Collaborative Study Group on Graves’ Disease. Effects of l-thyroxine administration, TSH-receptor antibodies and smoking on the risk of recurrence in Graves’ hyperthyroidism treated with Antithyroid drugs: a double-blind prospective randomized study. Eur J Endocrinol 2001, 144: 475–83.
Saggiorato E, Arecco F, Mussa A, et al; Piedmont Goiter Study Committee. Goiter prevalence and urinary iodine status in urban and rural/mountain areas of Piedmont region. J Endocrinol Invest 2006, 29: 67–73.
Dayan CM. Interpretation of thyroid function tests. Lancet 2001, 357: 619–24.
Cho BY, Shong MH, Yi KH, Lee HK, Koh CS, Min HK. Evaluation of serum basal thyrotrophin levels and thyrotrophin receptor antibody activities as prognostic markers for discontinuation of antithyroid drug treatment in patients with Graves’ disease. Clin Endocrinol (Oxf) 1992, 36: 585–90.
Perez C, Scrimshaw S, Munoz A. Technique of endemic goitre surveys. Monogr Ser World Health Organ 1960, 44: 369–83.
Carella C, Mazziotti G, Sorvillo F, et al. Serum thyrotropin receptor antibodies concentrations in patients with Graves’ disease before, at the end of methimazole treatment, and after drug withdrawal: evidence that the activity of thyrotropin receptor antibody and/or thyroid response modify during the observation period. Thyroid 2006, 16: 295–302.
Vitti P, Rago T, Chiovato L, et al. Clinical features of patients with Graves’ disease undergoing remission after antithyroid drug treatment. Thyroid 1997, 7: 369–75.
Schott M, Minich WB, Willenberg HS, et al. Relevance of TSH receptor stimulating and blocking autoantibody measurement for the prediction of relapse in Graves’ disease. Horm Metab Res 2005, 37: 741–4.
Quadbeck B, Hoermann R, Hahn S, Roggenbuck U, Mann K, Janssen OE. Binding, stimulating and blocking TSH receptor antibodies to the thyrotropin receptor as predictors of relapse of Graves’ disease after withdrawal of antithyroid treatment. Horm Metab Res 2005, 37: 745–50.
Nedrebo BG, Holm PI, Uhlving S, et al. Predictors of outcome and comparison of different drug regimens for the prevention of relapse in patients with Graves’ disease. Eur J Endocrinol 2002, 147: 583–9.
Yamada T, Aizawa T, Koizumi Y, Komiya I, Ichikawa K, Hashizume K. Age-related therapeutic response to antithyroid drug in patients with hyperthyroid Graves’ disease. J Am Geriatr Soc 1994, 42: 513–6.
Kashiwai T, Hidaka Y, Takano T, et al. Practical treatment with minimum maintenance dose of anti-thyroid drugs for prediction of remission in Graves’ disease. Endocr J 2003, 50: 45–9.
Chiovato L, Barbesin G, Pinchera A. Graves’ disease. In: DeGroot LJ, Jameson JL (eds), Endocrinology, 4th edition. Philadelphia, USA: Saunders Co. 2001, 1422.
Barrio R, López-Capapé M, Martinez-Badás I, Carrillo A, Moreno JC, Alonso M. Graves’ disease in children and adolescents: response to long-term treatment. Acta Paediatr 2005, 94: 1583–9.
Akuzawa M, Murakami M, Yamada M, Satoh T, Shimizu H, Mori M. Preserved activation of thyrotropin receptor antibody to stimulate thyroid function despite long-term treatment in euthyroid patients with Graves’ disease. Eur J Endocrinol 1998, 138: 281–5.
Feldt-Rasmussen U, Schleusener H, Carayon P. Meta-analysis evaluation of the impact of thyrotropin receptor antibodies on long term remission after medical therapy of Graves’ disease. J Clin Endocrinol Metab 1994, 78: 98–102.
Takasu N, Yamashiro K, Komiya I, Ochi Y, Sato Y, Nagata A. Remission of Graves’ hyperthyroidism predicted by smooth decreases of thyroid-stimulating antibody and thyrotropin-binding inhibitor immunoglobulin during antithyroid drug treatment. Thyroid 2000, 10: 891–6.
Yamano Y, Takamatsu J, Sakane S, Hirai K, Kuma K, Ohsawa N. Differences between changes in serum thyrotropin-binding inhibitory antibodies and thyroid-stimulating antibodies in the course of antithyroid drug therapy for Graves’ disease. Thyroid 1999, 9: 769–73.
Giovanella L, Ceriani L, Garancini S. Clinical applications of the 2nd generation assay for anti-TSH receptor antibodies in Graves’ disease. Evaluation in patients with negative 1st generation test Clin Chem Lab Med 2001, 39: 25–8.
Maugendre D, Massart C. Clinical value of a new TSH binding inihibitory activity assay using human TSH receptors in the follow-up of antithyroid drug treated Graves’ disease. Comparison with thyroid stimulating antibody bioassay. Clin Endocrinol 2001, 54: 89–96.
Zimmermann-Belsing T, Nygaard B, Rasmussen AK, Feldt-Rasmussen U. Use of the 2nd generation TRAK human assay did not improve prediction of relapse after antithyroid medical therapy of Graves’ disease. Eur J Endocrinol 2002, 146: 173–7.
Cooper DS. Drug therapy: antithyroid drugs. N Engl J Med 2005, 352: 905–17.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mazza, E., Carlini, M., Flecchia, D. et al. Long-term follow-up of patients with hyperthyroidism due to Graves’ disease treated with methimazole. Comparison of usual treatment schedule with drug discontinuation vs continuous treatment with low methimazole doses: A retrospective study. J Endocrinol Invest 31, 866–872 (2008). https://doi.org/10.1007/BF03346433
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03346433