Abstract
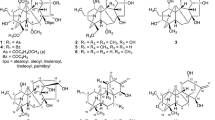
The bioactivity-guided fractionation of the methylene chloride extract of the sclerotium ofPoria cocos led to the isolation of (S)-(+)-turmerone (1), ergosterol peroxide (2), polyporenic acid C (3), dehydropachymic acid (4), pachymic acid (5), and tumulosic acid (6). Compounds4-6 exhibited moderate cytotoxicities, with IC50 values of 20.5, 29.1, and 10.4 αM, respectively, against a human colon carcinoma cell line. However,3-6 not only showed inhibitory activities as potent as etoposide used as a positive control on DNA topoisomerase II (36.1, 36.2, 43.9 and 66.7% inhibition at a concentration of 20 μM, respectively), but also inhibition of DNA topoisomerase I (55.8, 60.7, 43.5, and 83.3% inhibition at a concentration of 100 μM, respectively).
Similar content being viewed by others
References
Buckingham, J., Dictionary of Natural Products, Chapman & Hall, Volume 5, R-Z, pp. 5944–5945 (1994).
Fuganti, C., Serra, S., and Dulio, A., Bakers Yeast mediated enantioselective synthesis of the bisabolane sesquiterpenes curcumene, turmerone, dehydrocurcumene and nuciferal.J. Chem. Soc., Perkin Trans., 1, 279–282 (1999).
Fukuda, M., Nishio, K., Kanzawa, F., Ogasawara, H., Ishida, T., Arioka, H., Bojamowski, K., Oka, M., and Saijo, N., Synergism between cisplatin and topoisomerase I inhibitors, NB-506 and SN-38, in human small cell lung cancer cells.Cancer Res., 56, 789–793 (1996).
Giner, E. M., Manez, S., Recio, M. C., Giner, R. M., Miguel, C. N., and Rios, J. L.,In vivo studies on the anti-inflammatory activity of pachymic and dehydrotumulosic acids.Planta Med., 66, 221–227 (2000).
Hattori, T., Hayashi, K., Nagao, T., Furuta, K., Ito, M., and Suzuki, Y., Studies on antinephritic effects of plant components (3): Effect of pachyman, a main component ofPoria cocos Wolf on original-type anti-GBM nephritis in rats and its mechanisms.Jpn. J. Pharmacol., 59, 89–96 (1992).
Kaminaga, T., Yasukawa, K., Takido, M., Tai, T., and Nunoura, Y., Inhibitory effect ofPoria cocos on 12-O-tetradecanoylphorbol-13-acetate-induced ear edema and tumor promotion in mouse skin.Phytother. Res., 10, 581–584 (1996a).
Kaminaga, T., Yasukawa, K., Kanno, H., Tai, T., Nunoura, Y., and Takido, M., Inhibitory effects of lanostane-type triterpene acids, the components of Poria cocos, on tumor promotion by 12-0-tetradecanoylphorbol-13-acetate in two-stage carcinogenesis in mouse skin.Oncology, 53, 382–385 (1996b).
Kawagishi, H., Li, H., Tanno, O., Inoue, S., Ikeda, S., OhnishiKameyama, M., and Nagata, T., A lanostane-type triterpene from a mushroom Daedalea dickinsii.Phytochemistry, 46, 959–961 (1997).
Kim, D. S., Baek, N. I., Oh, S. R., Jung, K. Y., Lee, I. S., Kim, J. H., and Lee, H. K., Anticomplementary activity of ergosterol peroxide fromNaematoloma fasciculare and reassignment of NMR data.Arch. Pharm. Res., 20, 201–205 (1997).
Liu, L. F., DNA topoisomerase poisons as antitumor drugs.Annu. Rev. Biochem., 58, 351–375 (1989).
Nam, K. S., Jo, Y. S., Kim, Y. H., Hyun, J. W., and Kim, H. W., Cytotoxic activities of acetoxyscirpenediol and ergosterol peroxide from Paecilomyces tenuipes.Life Sci., 69, 229–237 (2001).
Nukaya, H., Yamashiro, H., Fukazawa, H., Ishida, H., and Tsuji, K., Isolation of inhibitors of TPA-induced mouse ear edema from Hoelen, Poria cocos.Chem. Pharm. Bull., 44, 847–849 (1996).
Roth, G. N., Chandra, A., and Nair, M. G., Novel bioactivities of Curcuma longa constituents.J. Nat. Prod., 61, 542–545 (1998).
Rubinstein, L. V., Shoemaker, R. H., Paul, K. D., Simon, R. M., Tosini, S., Skehan, P., Scudiero, D. A., Monks, A., and Boyd, M. R., Comparison of in vitro anticancer-drug-screening data generated with a tetrazolium assay versus a protein assay against a diverse panel of human tumor cell lines.J. Nat. Cancer Inst., 82, 1113–1118 (1990).
Tai, T., Akita, Y., Kinoshita, K., Koyama, K., Takahachi, K., and Watanaba, K., Anti-emetic principles of Poria cocos.Planta Med., 61, 527–530 (1995a).
Tai, T., Akahori, A., and Shingu, T., Triterpenoids from Poria cocos.Phytochemistry, 30, 2796–2797 (1991).
Tai, T., Akahori, A., and Shingu, T., A lanostane triterpenoid from Poria cocos.Phytochemistry, 31, 2548–2549 (1992).
Tai, T., Akahori, A., and Shingu, Triterpenes of Poria cocos.Phytochemistry, 32, 1239–1244 (1993).
Tai, T., On the Constituents ofPoria cocos WOLF. Dissertation, Kobe Gakuin University, Kobe, pp. 74, (1996).
Tai, T., Shingu, T., and Kikuchi, T., Tezuka, Y., and Akahori, A., Isolation of lanostane type triterpene acids having an acetoxyl group from sclerotia of Poria cocos.Phytochemistry, 40, 225–231 (1995b).
Tai, T., Shingu, T., Kikuchi, T., Tezuka, Y., and Akahori, A., Triterpenes from the surface layer of Poria cocos.Phytochemistry, 39, 1165–1169 (1995c).
Ukiya, M., Akihisa, T., Tokuda, H., Hirano, M., Oshikubo, M., Nobukuni, Y., Kimura, Y., Tai, T., Kondo, S., and Nishino, H., Inhibition of tumor-promoting effects by poricoic acids G and H and other lanostane-type triterpones and cytotoxic activity of poricoic acids A and G from Poria cocos.J. Nat. Prod., 65, 462–465 (2002).
Yasukawa, T., Kaminaga, T., Kitanaka, H., Tai, T., Nunoura, Y., Natori, S., and Takido, M., 3Beta-p-hydroxybenzoyldehydrotumulosic acid fromPoria cocos, and its anti-inflammatory effect.Phytochemistry, 48, 1357–1360 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, G., Xu, ML., Lee, CS. et al. Cytotoxicity and dna topoisomerases inhibitory activity of constituents from the sclerotium ofPoria cocos . Arch Pharm Res 27, 829–833 (2004). https://doi.org/10.1007/BF02980174
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02980174