Abstract
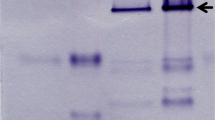
A ferredoxin was purified from the anaerobic protozoon Trichomonas vaginalis. The protein had a molecular weight of 12,000 as assessed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis, gel filtration and amino acid analysis. The protein contained seven 1/2-cystine (cystine plus cysteine) residues and one tyrosine, and lacked tryptophan. Chemical analysis and spectral properties of the ferredoxin indicated the presence of a [2Fe-2S] cluster. The complex optical spectrum of the native ferredoxin had peaks at 310 and 450 nm and shoulders near 415 and 550 nm. The molar absorbance of the protein at 450 nm was 8,000m −1 × cm−1. The EPR spectrum of reduced ferredoxin had two features at g values of 2.02 and 1.94 and revealed axial symmetry. Results of subcellular fractionation studies indicated the ferredoxin to be a major iron-sulfur protein of the cells and to be located in the hydrogenosome. The ability of the ferredoxin to function as an electron carrier was demonstrated by its reduction by pyruvate: ferredoxin oxidoreductase and hydrogenase as detected by EPR spectroscopy and by its stimulation of metronidazole reduction by these enzymes. These observations implicate ferredoxin as an important electron transport component in hydrogenosomes of T. vaginalis.
Article PDF
Similar content being viewed by others
Abbreviations
- P1/2 :
-
half saturation parameter
- SDS-PAGE:
-
sodium dodecyl sulfate-polyacrylamide gel electrophoresis
References
Adams, M.W.W., L.E. Mortenson &J.-S. Chen: Hydrogenase. Biochim. Biophys. Acta 594, 105–176 (1981)
Blum, H. &T. Ohnishi: Electron spin relaxation of the electron paramagnetic resonance spectra of cytochrome c. Biochim. Biophys. Acta 621, 9–18 (1980)
Cammack, R., L. Kerscher &D. Oesterhelt: A stable free radical intermediate in the reaction of 2-oxoacid: ferredoxin oxidoreductases ofHalobacterium halobium. FEBS Lett. 118, 271–273 (1980)
Diamond, L.S.: The establishment of various trichomonads of animals and man in axenic cultures. J. Parasitol. 43, 488–490 (1957)
Dutton, P.L.: Redox potentiometry. Determination of midpoint potentials of oxidation-reduction components of biological electron-transfer systems. Methods Enzymol. 54, 411–435 (1978)
Keresztes-Nagy, S. &E. Margoliash: Preparation and characterization of alfalfa ferredoxin. J. Biol. Chem. 241, 5955–5966 (1966)
Kimura, T. &K. Suzuki: Components of the electron transport system in adrenal steroid hydroxylase. Isolation and properties of non-heme iron protein (adrenodoxin). J. Biol. Chem. 242, 485–491 (1967)
Knoell, H.-E. &J. Knappe:Escherichia coli ferredoxin, an iron-sulfur protein of the adrenodoxin type. Eur. J. Biochem. 50, 245–252 (1974)
Lindmark, D.G. &M. Muller: Hydrogenosome, a cytoplasmic organelle of the anaerobic flagellate,Tritrichomonas foetus, and its role in pyruvate metabolism. J. Biol. Chem. 248, 7724–7728 (1973)
Lindmark, D.G., M. Muller &H. Shio: Hydrogenosomes inTrichomonas vaginalis. J. Parasitol. 61, 552–554 (1975)
Lindmark, D.G. &M. Muller: Antitrichomonad action, mutagenicity, and reduction of metronidazole and other nitroimidazoles. Antimicrob. Agents Chemother. 10, 476–482 (1976)
Lode, E.T., C.L. Murray &J.C. Rabinowitz: Apparent oxidation-reduction potential ofClostridium acidi-urici ferredoxin. Effect of pH, ionic strength, and amino acid replacements. J. Biol. Chem. 251, 1683–1687 (1976)
Mack, S.R. &M. Muller: End products of carbohydrate metabolism in Trichomonas vaginalis. Comp. Biochem. Physiol. 67B, 213–216 (1980)
Marczak, R., T.E. Gorrell &M. Muller: Hydrogenosomal ferredoxin of the anaerobic protozoon,Tritrichomonas foetus. J. Biol. Chem. 258, 12427–12433 (1983)
Moore, S.: On the determination of cystine as cysteic acid. J. Biol. Chem. 238, 235–237 (1963)
Muller, M.: The hydrogenosome. Symp. Soc. Gen. Microbiol. 30, 127–142 (1980)
Ohnishi, T., D. Lloyd, D.G. Lindmark &M. Muller: Respiration ofTritrichomonas foetus. Components detected in hydrogenosomes and in intact cells by electron paramagnetic resonance spectrometry. Mol. Biochem. Parasitol. 2, 39–50 (1980)
Orme-Johnson, W.H. &N.R. Orme-Johnson: Iron-sulfur proteins: The problem of determining cluster type. In: Iron-Sulfur Proteins. Spiro T.G., ed., John Wiley & Sons, New York, pp. 67–96 (1982)
Read, C.P. &A.H. Rothman: Preliminary notes on the metabolism of Trichomonas vaginalis. Am. J. Hyg. 61, 249–260 (1955)
Reeves, R.E., J.D. Guthrie &P. Lobelle-Rich:Entamoeba histolytica: Isolation of ferredoxin. Exp. Parasitol. 49, 83–88 (1980)
Rupp, H., K.K. Rao, D.O. Hall &R. Cammack: Electron spin relaxation of iron-sulphur proteins studied by microwave power saturation. Biochim. Biophys. Acta 537, 255–269 (1978)
Suhara, K., S. Takemori &M. Katagiri: Improved purification of bovine adrenal iron-sulfur protein. Biochim. Biophys. Acta 263, 272–278 (1972)
Tanaka, M., T. Nakashima, H.F. Mower &K.T. Yasunobu: The C- and N-terminal amino acid sequences ofClostridium pasteurianum ferredoxin. Arch. Biochem. Biophys. 105, 570–574 (1964)
Yasunobu, K.T. &M. Tanaka: The types, distribution in nature, structure-function, and evolutionary data of the iron-sulfur proteins. In: Iron-Sulfur Proteins. Vol. II, Molecular Properties. W. Lovenberg ed., Academic Press, New York, pp. 27–130 (1973)
Yoch, D.C. &R.P. Carithers: Bacterial iron-sulfur proteins. Microbiol. Rev. 43, 384–421 (1979)
Author information
Authors and Affiliations
Additional information
Dedicated with admiration and gratitude to Dr.Heinz Holter on the occasion of his 80th birthday
Guest Investigator and Rask-Ørsted Fellow in the Department of Physiology of the Carlsberg Laboratorium in 1965–1966
Rights and permissions
About this article
Cite this article
Gorrell, T.E., Yarlett, N. & Müller, M. Isolation and characterization of Trichomonas vaginalis ferredoxin. Carlsberg Res. Commun. 49, 259–268 (1984). https://doi.org/10.1007/BF02913954
Issue Date:
DOI: https://doi.org/10.1007/BF02913954