Abstract
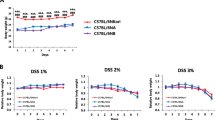
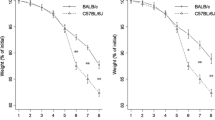
One-week dextran sulfate treatment of conventional (CV) immunodeficient (SCID) mice gave rise to acute colitis in the colon mucosa; germ-free (GF) SCID mice did not exhibit any changes in colon morphology. Dextran sulfate application to CV immunocompetent (BALB/c) mice did induce substantial changes in the colon mucosa (grade4); GF BALB/c mice showed mild changes in the colon morphology (grade1) only. GF SCID mice and CV SCID mice died during the second round of dextran sulfate treatment suffering from chronic colitis; GF BALB/c mice exhibited mild crypt distortion while CV BALB/c mice showed a complete loss of the surface epithelium (grade4), accompanied by T and B lymphocyte infiltration.
Similar content being viewed by others
References
Bosma M.J., Carroll A.M.: The SCID mouse mutant: definition, characterization and potential uses.Ann. Rev. Immunol.9, 323–350 (1991).
Bylund-Fellenius A.C., Lanström E., Axelsson L.D., Midtvedt T.: Experimental colitis is induced by dextran sulfate in normal and germ-free mice.Microb. Ecol. Health Dis.7, 207–215 (1994).
Cebra J.J., Jiang H., Šterzl J., Tlaskalová-Hogenová H.: The role of mucosal microbiota in the development and maintenance of the mucosal immune system, pp. 267–280 in L.O. Ogra, J. Mestecky, M.E. Lamm, W. Strober, J. Bienenstock, J.R. McGhee (Eds).Mucosal Immunology, 2nd edn. Academic Press, New York 1999.
Cooper H.S., Murthy S.N.S., Snak R.S., Sedergran D.S.: Clinicopathologic study of dextran sulfate sodium experimental murine colitis.Lab. Invest.69, 238–249 (1993).
Dieleman L.A., Ridwan B.U., Tennyson G.S., Beagley K.W., Bucy R.P., Elson C.O.: Dextran sodium sulfate-induced colitis occurs in severe combined immunodeficient mice.Gastroenterology107, 1643–1652 (1994).
Duchmann R., Zeitz M.: Crohn's disease, pp. 1055–1080 in L.O. Ogra, J. Mestecky, M.E. Lamm, W. Strober, J. Bienenstock, J.R. McGhee (Eds):Mucosal Immunology, 2nd edn. Academic Press, New York 1999.
Elson C.O., Cong Z., Brandwein S., Weaver C.T., McCabe R.P., Mahler M., Sundberg J.P., Leiter E.H.: Experimental models to study molecular mechanisms underlying intestinal inflammation.Ann. N.Y. Acad. Sci.17, 85–95 (1998).
Elson C.O.: The immunology of inflamatory bowel disease, pp. 97–164 in J.B. Kirsner, R.G. Shorter (Eds).Inflamatory Bowel Disease, 3rd ed. Lea & Febiger, Philadelphia (USA) 1988.
Hans W., Schölmerich J., Gross, V., Falk W.: The role of the resident intestinal flora in acute and chronic dextran sodium sulfate induced colitis in mice.Eur. J. Gastroenterol.-Hepatol.12, 267–273 (2000).
James S.P., Strober W.: Gastrointestinal disease, pp. 455–472 in D.P. Stites, A.I. Terr (Eds):Basic and Clinical Immunology. Victoria Publishing, San Francisco (USA) 1994.
Karp L.C., Targan S.R.: Evidence for an updated hypothesis of disease pathogenesis. pp. 1047–1053 L.O. Ogra, J. Mestecky, M.E. Lamm, W. Strober, J. Bienenstock, J.R. McGhee (Eds):Mucosal Immunology, 2nd edn. Academic Press, New York 1999.
Leach M.W., Davidson N.J., Fort M.M., Powrie F., Rennick D.M.: The role of IL-10 in inflammatory bowel disease of mice and men.Toxicol. Pathol.27, 123–133 (1999).
Louis E., Belaiche J.: Experimental models of inflammatory bowel disease.Acta Gastroenterol Belg.57, 306–309 (1994).
Ni J., Chen S.F., Hollander D.: Effects of dextran sulfate sodium on intestinal epithelial cells and intestinal lymphocytes.Gut39, 234–241 (1996).
Ohkusa T.: Production of experimental ulcerative colitis in hamsters by dextran sulfate sodium and change in intestinal microfloraJapan: J. Gastroenterol.82, 1327–1336 (1985).
Okayasu I., Hatakeyama S., Yamada M., Ohkusa T., Inagi Y., Nakya R.: A novel method in the induction of reliable experimental acute and chronic ulcerative colitis in mice.Gastroenterology98, 694–702 (1990).
Pitcher M.C.L., Beatty E.R., Cummings J.H.: The contribution of sulfate reducing bacteria and 5-aminosalicylic acid to fecal sulfide in patients with ulcerative colitis.Gut46, 694–702 (1997).
Podolsky D.K.: Lessons from genetic models of inflammatory bowel disease.Acta Gastroenterol. Belg.60, 163–165 (1997).
Powrie F., Leach M.W.: Genetic and spontaneous models of inflammatory bowel disease in rodents: evidence for abnormalities in mucosal immune regulation.Ther. Immunol.2, 115–23 (1995).
Shintani N., Nakajima T., Sugiura M., Murakami K., Nakamura K., Mayumi T.: Proliferative effect of dextran sulfate sodium (DSS)-pulsed macrophages on T cells from mice with DSS-induced colitis and inhibition of effect by IgG.Scand. J. Immunol.46, 581–586 (1997).
Simpson E.J., Chapman M.A.S., Dawson J., Berry D., MacDonald I.A., Cole A.In vivo measurement of colonic butyrate metabolism in patients with quiescent ulcerative colitis.Gut46, 73–77 (2000)
Strober W., James S.: Ulcerative colitis, pp. 458–459 in D.P. Stites, A.I. Terr (Eds):Basic and Clinical Immunology. Victoria Publishing, San Francisco (USA) 1994.
Strober W., Ehrhardt R.O.: Chronic intestinal inflammation: an unexpected outcome in cytokine or T well receptor mutant mice.Cell78, 203–295 (1993).
Štěpánková, R., Šinkora J., Hudcovic T., Kozáková H., Tlaskalová-Hogenová H.: Differences in development of lymphocyte subpopulations from gut-associated lymphatic tissue (GALT) of germ-free and conventional rats: effect of aging.Folia Microbiol.43, 531–534 (1998).
Tlaskalová-Hogenová H., Štěpánková R., Tučková L., Farré M.A., Funda D.P., Verdú E.F., Šinkora J., Hudcovic T., Řeháková Z., Cukrowska B., Kozáková H., Prokešová L.: Autoimmunity, immunodeficiency and mucosal infections: chronic intestinal inflammation as a sensitive indicator of immunoregulatory defects in response to normal luminal microflora.Folia Microbiol.43, 545–550 (1998).
Tlaskalová-Hogenová H., Šterzl J., Štěpánková R., Dlabač V., Větvička V., Rossmann P., Mandel L., Rejnek J.: Development of immunological capacity under germ-free and conventional conditions.Ann. N.Y. Acad. Sci.409, 96–113 (1983).
Tlaskalová-Hogenová H.: Gnotobiology as a tool, pp. 1524–1527 in I. Lefkovits (Ed.):Manual of Immunological Methods. Academic Press, New York-London 1997.
Tlakalová-Hogenová H., Štěpánková R., Tučková L., Cukrowska B., Hudcovic T., Bendjelloul F., Malý P., Jirkovská M., Mandys V., Kofroñová O., Verdú E.F., Berčik P., Farré M., Prokešová L., Funda D.P., Kozáková H., Michetti P., Cebra J.: Effector mechanisms in intestinal immunity and inflammation, pp. 74–83 in J. Emmrich, S. Liebe, E.F. Strange (Eds.):Innovative Concepts in IBD. Kluwer Academic Publishers, Dordrecht-Boston-London 1999.
Trinchieri G., Perusia B.: Immune interferon: a pleiotropic lymphokine with multiple effects.Immunol. Today6, 131–136 (1985).
Verdú, E.F., Berčik P., Cukrowska B., Farré M., Bonson R.H., Saraga E., Lum A., Corthesy I., Tlaskalová H.: Oral administration antigens from intestinal flora an aerobic bacteria reduces the severity of experimental acute colitis in BALB/c mice.Clin. Exp. Immunol.120, 46–50 (2000).
Watt, J., Marcus R.: Carrageenan-induced ulceration of the large intestine in the guinea pig.Gut12, 164–171 (1971).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hudcovic, T., Štěpánková, R., Cebra, J. et al. The role of microflora in the development of intestinal inflammation: Acute and chronic colitis induced by dextran sulfate in germ-free and conventionally reared immunocompetent and immunodeficient mice. Folia Microbiol 46, 565–572 (2001). https://doi.org/10.1007/BF02818004
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02818004