Abstract
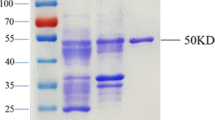
Severe acute respiratory syndrome (SARS), a new disease with symptoms similar to those of atypical pneumonia, raised a global alert in March 2003. Because of its relatively high transmissibility and mortality upon infection, probable SARS patients were quarantined and treated with special and intensive care. Therefore, instant and accurate laboratory confirmation of SARS-associated coronavirus (SARS-CoV) infection has become a worldwide interest. For this need, we purified recombinant proteins including the nucleocapsid (N), envelope (E), membrane (M), and truncated forms of the spike protein (S1–S7) of SARS-CoV inEscherichia coli. The six proteins N, E, M, S2, S5, and S6 were used for Western blotting (WB) to detect various immunoglobulin classes in 90 serum samples from 54 probable SARS patients. The results indicated that N was recognized in most of the sera. In some cases, S6 could be recognized as early as 2 or 3 days after illness onset, while S5 was recognized at a later stage. Furthermore, the result of recombinant-protein-based WB showed a 90% agreement with that of the whole-virus-based immunofluorescence assay. Combining WB with existing RT-PCR, the laboratory confirmation for SARS-CoV infection was greatly enhanced by 24.1%, from 48.1% (RT-PCR alone) to 72.2%. Finally, our results show that IgA antibodies against SARS-CoV can be detected within 1 week after illness onset in a few SARS patients.
Article PDF
Similar content being viewed by others
References
Büttner J. Evaluation of the diagnostic value of laboratory investigations. J Clin Chem Clin Biochem 15:1–12;1977.
CDC SARS Investigative Team, Fleischauer AT. Outbreak of severe acute respiratory syndrome — worldwide, 2003. Mor Mortal Wkly Rep 52:226–228;2003.
Cumulative number of SARS probable cases in Taiwan, SARS Online Information Center, Center for Disease Control, Taiwan [Accessed July 2, 2003]. Available from: URL: http://www.cdc.gov.tw/sarsen.
de Arriba ML, Carvajal A, Pozo J, Rubio P. Mucosal and systemic isotype-specific antibody responses and protection in conventional pigs exposed to virulent or attenuated porcine epidemic diarrhoea virus. Vet Immunol Immunopathol 85:85–97;2002.
Drosten C, Gunther S, Preiser W, van der Werf S, Brodt HR, Becker S, Rabenau H, Panning M, Kolesnikova L, Fouchier RA, et al. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N Engl J Med 348:1967–1976;2003.
Elia G, Fiermonte G, Pratelli A, Martella V, Camero M, Cirone F, Buonavoglia C. Recombinant M protein-based ELISA test for detection of antibodies to canine coronavirus. J Virol Methods 109:139–142;2003.
Krokhin O, Li Y, Andonov A, Feldman H, Flick R, Jones S, Stroeher U, Bastien N, Dasuri KV, Cheng K, et al. Mass spectrometric characterization of proteins from the SARS virus: A preliminary report. Mol Cell Proteomics 2:346–356;2003.
Ksiazek TG, Erdman D, Goldsmith CS, Zaki SR, Peret T, Emery S, Tong S, Urbani C, Comer JA, Lim W, et al. A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med 348:1953–966;2003.
Lee ML, Chen CJ, Su IJ, Chen KT, Yeh CC, King CC, Chang HL, Wu YC, Ho MS, Jiang DD, et al. Severe acute respiratory syndrome-Taiwan, 2003. Morb Mortal Wkly Rep 52:461–466;2003.
Loa CC, Lin TL, Wu CC, Bryan T, Hooper T, Schrader D. Specific mucosal IgA immunity in turkey poults infected with turkey coronavirus. Vet Immunol Immunopathol 88:57–64;2002.
Marra MA, Jones SJ, Astell CR, Holt RA, Brooks-Wilson A, Butterfield YS, Khattra J, Asano JK, Barber SA, Chan SY, et al. The Genome sequence of the SARS-associated coronavirus. Science 300:1399–1404;2003.
Naslund K, Traven M, Larsson B, Silvan A, Linde N. Capture ELISA systems for the detection of bovine coronavirus-specific IgA and IgM antibodies in milk and serum. Vet Microbiol 72:183–206;2002.
Peiris JS, Chu CM, Cheng VC, Chan KS, Hung IF, Poon LL, Law KI, Tang BS, Hon TY, Chan CS, et al. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: A prospective study. Lancet 361:1767–1772;2003.
Peiris JS, Lai ST, Poon LL, Guan Y, Yam LY, Lim W, Nicholls J, Yee WK, Yan WW, Cheung MT, et al. Coronavirus as a possible cause of severe acute respiratory syndrome. Lancet 361:1319–325;2003.
Rota PA, Oberste MS, Monroe SS, Nix WA, Campagnoli R, Icenogle JP, Penaranda S, Bankamp B, Maher K, Chen MH, et al. Characterization of a novel coronavirus associated with severe acute respiratory syndrome. Science 300:1394–1399;2003.
Twu SJ, Chen TJ, Chen CJ, Olsen SJ, Lee LT, Fisk T, Hsu KH, Chang SC, Chen KT, Chiang IH, et al. Control measures for severe Acute respiratory syndrome (SARS) in Taiwan. Emerg Infect Dis 9:718–720;2003.
Use of laboratory methods for SARS diagnosis, WHO 2003 [Accessed July 2, 2003]. Available from: URL: http://www.who.int/csr/sars/labmethods/en/print.html.
van Ginkel FW, Nguyen HH, McGhee JR. Vaccines for mucosal immunity to combat emerging infectious diseases. Emerg Infect Dis 6:123–132;2000.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wu, HS., Hsieh, YC., Su, IJ. et al. Early detection of antibodies against various structural proteins of the SARS-associated coronavirus in SARS patients. J Biomed Sci 11, 117–126 (2004). https://doi.org/10.1007/BF02256554
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02256554