Abstract
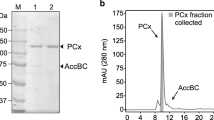
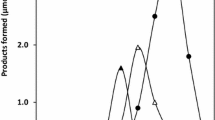
The NADP+-specific glutamate dehydrogenase (GDH) fromEscherichia coli strain D5H3G7, an enzyme that catalyzes the interconversion of α-ketoglutarate andl-glutamate, has been shown to be phosphorylated in vitro in an ATP-dependent enzymatic reaction. The phosphorylated protein is extremely acid labile and is unstable at high pH. Treatment of GDH with diethyl pyrocarbonate (DEP), a histidine-modifying reagent, blocked the incorporation of32P from [γ-32P]ATP. GDH catalytic activity was also inhibited by DEP treatment. Hydroxylamine, a reagent hydrolyzing phosphoramidates, catalyzed the removal of phosphate from phosphorylated GDH, suggesting that GDH may be phosphorylated at a histidine residue(s). A total enzymatic hydrolysis of phosphorylated GDH, which was electroeluted from a native polyacrylamide gel, was analyzed by a Dowex 1-8X anion exchange chromatography. The presence of32P-labeled 3-phosphohistidine, characterized and identified from this hydrolysate, demonstrates that a histidine residue(s) is the site of phosphorylation.
Similar content being viewed by others
Literature Cited
Anderson B, Weigel N, Kundig W, Roseman S (1971) Sugar transport III: purification and properties of phosphocarrier protein (HPR) of the phosphoenol pyruvate dependent phosphotransferase system ofEscherichia coli. J Biol Chem 246:7023–7033
Block RJ, Durrum EL, Zweig G (1958) A manual of paper chromatography and paper electrophoresis, 2nd edn. New York: Academic Press, p 168
Bridger WA (1974) Succinyl-CoA synthetase. In: Boyder PD (ed) The enzymes, 3rd edn, Vol 10. New York: Academic Press, pp 581–606
DeLuca M, Ebner DE, Hultquist DE, Kreil G, Peter JB, Moyer RW, Boyer PD (1963) The isolation and identification of phosphohistine from mitochondrial protein. Biochem Zeit 338:512–525
DiSabato G, Jencks WP (1961) Mechanism and catalysis of reactions of acyl phosphate I. nucleophilic reactions. J Am Chem Soc 83:4393–4399
Gabriel O (1971) Locating enzymes on gels. Methods Enzymol 22:578–604
Hokin LE, Sastry PS, Galsworthy PR, Yoda A (1965) Evidence that a phosphorylated intermediate in a brain transport adenosine triphosphatase is an acyl phosphate. Proc Natl Acad Sci USA 54:177–184
Jameel S, El-Glu T, McFadden BA (1985) Modification of the active site of isocitrate lyase from watermelon cotyledons. Arch Biochem 236:72–81
Kahn FR, McFadden BA (1982) Isocitrate lyase from flax: terminal residues composition, active site, and catalysis. Plant Physiol 70:943–948
Lin H-PP, Reeves HC (1991) Phosphorylation ofEscherichia coli NADP+-specific glutamate dehydrogenase. Curr Microbiol 22:181–184
Lin H-PP, Reeves HC (1991) Purification and characterization of NADP+-specific glutamate dehydrogenase fromEscherichia coli. Curr Microbiol 22:371–376
Oganesuan AS, Gevorky ZhS, Babauan MA (1989) Mechanism or regulation of the activity of glutamate dehydrogenase. Biol Zh Arm 42:156–158
Rathlev T, Rosenberg T (1956) Non-enzymic formation and rupture of phosphorous to nitrogen linkages is phosphoramidate derivatives. Arch Biochem Biophys 65:319–339
Robertson EF, Hoyt JC, Reeves HC (1988) Evidence of histidine phosphorylation in isocitrate lyase fromEscherichia coli. J Biol Chem 263:2477–2482
Sheridan RC, McCullough JF, Wakefield ZT (1972) Phosphoramidic acid and its salts. Inorg Synth 13:23–26
Stewart GR, Mann AF, Fentem PA (1980) Enzymes of glutamate formation: glutamate dehydrogenase, glutamine synthetase and glutamate sythase. In: Miflin BJ (ed) The biochemistry plants, Vol 5. London: Academic Press, pp 271–327
Yasykova MYu, Vener AV, Muronetz VI, Nagradova NK (1988) Phosphorylation of lactate dehydrogenase by ATP. Biochem Intern 17:133–139
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lin, HP.P., Reeves, H.C. Properties of phosphorylated NADP+-specific glutamate dehydrogenase fromEscherichia coli . Current Microbiology 24, 73–79 (1992). https://doi.org/10.1007/BF01570901
Issue Date:
DOI: https://doi.org/10.1007/BF01570901