Summary
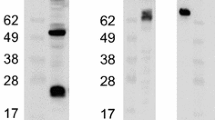
The bovine herpesvirus 4 (BHV-4) isolate LVR-140 growing in MDBK cell cultures was investigated by indirect immunofluorescence (IF), using bovine antisera and 15 monoclonal antibodies (MAb). The cytopathogenic effect was discernible between 20 and 30 hours post inoculation (p.i.) and developed slowly as has been reported for other BHV-4 strains. According to the protein specificity as revealed by radioimmunoprecipitation and the intracellular localisation of reacting antigens as detected by IF, the MAb could be assigned to one of three groups. The initial antigen expression was first detected at 8 hours p.i. and was limited to a proportion of the infected cells only, despite the high multiplicity of infection used; infectious cell-associated progency virus was first recognized at 12 hours p.i.
Similar content being viewed by others
References
Castrucci G, Frigeri F, Cilli V, Donelli G, Ferrari M, Chicchini U, Bordoni E (1986) A study of a herpesvirus isolated from dairy cattle with a history of reproductive disorders. Comp Immun Microbiol Infect Dis 9: 13–21
Ehlers B, Buhk H-J, Ludwig H (1985) Analysis of bovine cytomegalovirus genome structure: cloning and mapping of the monomeric polyrepetitive DNA unit, and comparison of European and American strains. J Gen Virol 66: 55–68
Fleckenstein B, Desrosiers RC (1982) Herpesvirus saimiri and herpesvirus ateles. In: Roizman B (ed) The Herpesviruses, vol 1. Plenum, New York, pp 253–332
Friedli K, Metzler AE (1987) Reactivity of monoclonal antibodies to proteins of a neurotropic bovine herpesvirus 1 (BHV-1) strain and to proteins of representative BHV-1 strains. Arch Virol 94: 109–122
Gibbs EPJ, Rweyemamu MM (1977) Bovine herpesviruses. Part II. Bovine herpesviruses 2 and 3. Vet Bull 47: 411–425
Honess RW, O'Hare P, Young D (1982) Comparison of thymidine kinase activities induced in cells productively infected with herpesvirus saimiri and herpes simplex virus. J Gen Virol 58: 237–249
Honess RW (1984) Herpes simplex and “the herpes complex”. Diverse observations and a unifying hypothesis. J Gen Virol 65: 2077–2107
Kit S, Kit M, Ichimura H, Crandell R, McConnel S (1986) Induction of thymidine kinase activity by viruses with group B DNA genomes: bovine cytomegalovirus (bovine herpesvirus 4). Virus Res 4: 197–212
Ludwig H (1983) Bovine herpesviruses. In: Roizman B (ed) The herpesviruses, vol 2. Plenum, New York, pp 135–214
Ludwig H (1984) Herpesviruses of bovidae: The characterization, grouping and role of different types, including latent viruses. In: Wittmann G, Gaskell RM, Rziha H-J (eds) Latent herpesvirus infections in veterinary medicine. Martinus Nijhoff, Boston, pp 171–189
Metzler AE, Matile H, Gassmann U, Engels M, Wyler R (1985) European isolates of bovine herpesvirus 1: a comparison of restriction endonuclease sites, polypeptides, and reactivity with monoclonal antibodies. Arch Virol 85: 57–69
Metzler AE, Wyler R (1986) Praevalenz des Bovinen Herpesvirus 4 in der schweizerischen Rinderpopulation und mögliche serologische Kreuzreaktion mit dem Bovinen Herpesvirus 1 (IBR/IPV-Virus). Schweiz Arch Tierheilk 128: 459–467
Modrow S, Wolf H (1983) Herpesvirus saimiri-induced proteins in lytically infected cells. I. Time-ordered synthesis. J Gen Virol 64: 37–46
Mohanty SB (1978) Bovine respiratory viruses. Adv Vet Sci Comp Med 22: 83–109
Osorio FA, Reed DE, Rock DL (1982) Experimental infection of rabbits with bovine herpesvirus 4: acute and persistent infection. Vet Microbiol 7: 503–513
Osorio FA, Reed DE (1983) Experimental inoculation of cattle with bovine herpesvirus 4: evidence for lymphoid-associated persistent infection. Am J Vet Res 44: 975–980
Osorio FA, Reed DE, Van der Maaten MJ, Metz CA (1985) Comparison of the herpesviruses of cattle by DNA restriction endonuclease analysis and serologic analysis. Am J Vet Res 46: 2104–2109
Osorio FA, Rock DL, Reed DE (1985) Studies on the pathogenesis of a bovine cytomegalo-like virus in an experimental host. J Gen Virol 66: 1941–1951
Potgieter LND, Maré CJ (1974) Assay and antigenic interrelationships of the recently isolated bovine herpesviruses DN 599, FTC, and V 11. Arch Ges Virusforsch 46: 238–247
Randall RE, Newman C, Honess RW (1984) Isolation and characterization of monoclonal antibodies to structural and nonstructural herpesvirus saimiri proteins. J Virol 52: 872–883
Randall RE, Newman C, Honess RW (1985) Asynchronous expression of the immediate-early protein of herpesvirus saimiri in populations of productively infected cells. J Gen Virol 66: 2199–2213
Rossiter PB, Mushi EZ, Plowright W (1978) Antibody response in cattle and rabbits to early antigens of malignant catarrhal fever virus in cultured cells. Res Vet Sci 25: 207–210
Rweyemamu MM, Loretu K (1973) Isolation of nonsyncytia forming herpes viruses from cattle in Tanzania. J Comp Pathol 83: 377–386
Storz J, Ehlers B, Todd WJ, Ludwig H (1984) Bovine cytomegaloviruses. Identification and differential properties. J Gen Virol 65: 697–706
Todd WJ, Storz J (1983) Morphogenesis of a cytomegalovirus from an American bison affected with malignant catharrhal fever. J Gen Virol 64: 1025–1030
Truman D, Ludwig H, Storz J (1986) Bovines Herpesvirus Typ4 (BHV-4). Untersuchungen zur Biologie und Verbreitung in Rinderbeständen und bei Besamungsbullen. J Vet Med B33: 485–501
Weiland F, Keil GM, Reddehase MJ, Koszinowski UH (1986) Studies on the morphogenesis of murine cytomegalovirus. Intervirology 26: 192–201
Wellemans G, Antoine H, Broes A, Charlier G, Van Opdenbosch E (1983) Isolement d'un virus herpès chez des bovins atteints de métrite post-partum. Ann Méd Vét 127: 481–482
Wellemans G, Van Opdenbosch E, Antoine H (1985) Some aspects of BHV-4 infection in cattle. In: Pastoret PP, Thiry E, Saliki J (eds) Immunity to herpesvirus infections of domestic animals. ECSC-EEC-EAEC, Brussels, Luxembourg, pp 165–172
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Augsburger, H.R., Metzler, A.E. In vitro growth characteristics of bovine herpesvirus 4 (BHV-4) as revealed by indirect immunofluorescence assay with monoclonal antibodies and polyvalent antisera. Archives of Virology 104, 309–321 (1989). https://doi.org/10.1007/BF01315552
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01315552