Abstract
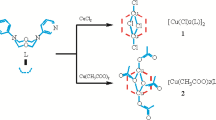
The title compound, Cu(C10H24N4)(ClO4)2, crystallizes in the triclinic system with unit cell dimensionsa= 8.744,b= 8.022,c= 8.677 Å, α = 118.7, β = 56.9, γ = 113.5 °,Z= 1, space group P¯1. The structure was solved by Fourier methods and refined by least-squares techniques using the 1545 nonzero three-dimensional counter-diffraction intensity data (CuKα) to give a conventionalR factor of 0.056. The coordination sphere of the copper ion is defined by a planar arrangement of the four nitrogen donors in the macrocyclic ligand with oxygen atoms from the perchlorate groups lying above and below this plane. The resulting tetragonally distorted octahedron of donor atoms gives interatomic distances Cu−N = 2.02(4) Å and 2.02(3) Å, and Cu−N = 2.57(4) Å.
Similar content being viewed by others
References
Alexander, L. E. and Smith, G. S. (1962)Acta Cryst. 15, 983–1004.
Almlof, J., Lundgren, J. O. and Olovsson, I. (1971)Acta Cryst. B27, 898–904.
Barefield, E. K. (1972)Inorg. Chem. 11, 2273–2274.
Bauer, R. A., Robinson, W. R. and Margerum, D. W. (1973)J. Chem. Soc. Chem. Comm. 289–290.
Bonnet, P. J. J. and Jeannin, Y. (1970)Acta Cryst. B26, 318–326.
Bosnich, R., Poon, C. K. and Tobe, M. L. (1965a)Inorg. Chem. 4, 1102–1108.
Bosnich, R., Mason, R., Pauling, P. J., Robertson, G. B. and Tobe, M. L. (1965b)Chem. Comm. 97–98.
Brown, B. W. and Lingafelter, E. C. (1964)Acta Cryst. 17, 254–259.
Busch, D. H., Farmery, K., Goedken, V., Katovic, V., Melnyk, A. C., Sperati, C. R., and Tokel, N. (1971)Advances in Chemistry Series 100, 44–78.
Curtis, N. F., Swann, D. A. and Waters, T. N. (1973a)J. Chem. Soc. Dalton 1408–1413.
Curtis, N. F., Swann, D. A. and Waters, T. N. (1973b)J. Chem. Soc. Dalton 1963–1974.
Dickens, B. (1969)Acta Cryst. B25, 1875–1882.
Fumas, T. C.Single Crystal Orienter Instruction Manual (General Electric Company, Milwaukee, 1957).
Hathaway, B. J. and Underhill, A. E. (1961)J. Chem. Soc. 3091–3096.
Hathaway, B. J., Procter, I. M., Tomlinson, A. A. G., Brown, D. S., Lee, J. D. and Melsom, B. G. A. (1967)Chem. Commun. 369–371.
Hughes, E. W. (1941)J. Amer. Chem. Soc. 63, 1737–1752.
Hussain, M. S., Joesten, M. D. and Lenhert, P. G. (1970)Inorg. Chem. 9, 162–168.
Komiyama, Y. and Lingafelter, E. C. (1964)Acta Cryst. 17, 1145–1148.
Larson, A. C. (1967)Acta Cryst. 23, 664–665.
Lewis, D. L., Hatfield, W. E. and Hodgson, D. J. (1972)Inorg. Chem. 11, 2216–2221.
Lewis, D. L. and Hodgson, D. J. (1974a)Inorg. Chem. 13, 143–147.
Lewis, D. L., Hatfield, W. E. and Hodgson, D. J. (1974b)Inorg. Chem. 13, 147–152.
Lide, D. R. (1962)Tetrahedron 17, 125–134.
Luukkonen, E., Pajunen, A. and Lehronen, M. (1970)Suom. Kemistilehti B.43, 160–165.
Meyer, M. H., Singh, P., Hatfield, W. E. and Hodgson, D. J. (1972)Acta Cryst. B28, 1607–1613.
Nave, C. and Truter, M. R. (1974)J. Chem. Soc. Dalton 2351–2354.
Pajunen, A. (1967)Suom. Kemistilehti B. 40, 32–34.
Pajunen, A., Smolander, K. and Belinskij, I. (1972a)Suom. Kemistilehti B. 45, 317–322.
Pajunen, A. and Lehtonen, M. (1972b)Suom. Kemistilehti B. 45, 43–46.
Poon, C. K. and Tobe, M. L. (1968)J. Chem. Soc. A 1549–1555.
Poon, C. K. (1971)Inorg. Chim. Acta 5, 322–324.
Rosenthal, M. R. (1973)J. Chem. Ed. 50, 331–335.
Sekizaki, M., Marumo, F., Yamasaki, K. and Saito, Y. (1910)Bull. Chem. Soc. Japan 44, 1731–1734.
Tang, S. C., Koch, S., Weinstein, G. N., Lane, R. W., and Holm, R. H. (1973)Inorg. Chem. 12, 2589–2595.
Whimp, P. O., Bailey, M. F. and Curtis, N. F. (1970)J. Chem. Soc. A 1956–1963.
Wright, D. A. and Quinn, J. D. (1974)Acta Cryst. B30, 2132–2138.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tasker, P.A., Sklar, L. Crystal and molecular structure of di(perchlorato)(1,4,8,11-tetraazacyclotetradecane)copper (II). Cu(cyclam) (ClO4)2 . Journal of Crystal and Molecular Structure 5, 329–344 (1975). https://doi.org/10.1007/BF01270621
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01270621