Summary
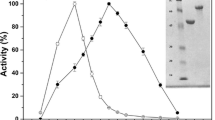
The thermal stability of a purifiedAspergillus niger β-glucosidase (EC 3.2.1.21) has been dramatically increased by intramolecular crosslinking with glutaraldehyde at pH 9.0, followed by borohydride reduction. The stabilization has been shown to be due to the formation of a distinct subpopulation of enzyme molecules, the activity of which has a half-life at 65°C of approximately 40 hours, which is some 80 times as long as that of the native enzyme at this temperature.
Similar content being viewed by others
References
Durand, H., Soucaille, P., and Tiraby, G. (1984)Enzyme Microb. Technol. 6, 175–180.
Hornby, W.E., and Goldstein, L. (1976)Methods Enzymol. 44, 118–134.
Howell, J.A. and Stuck, J.D. (1975)Biotechnol. Bioeng. 17, 873–893.
Hsu, T.A., Gong, G.S., Tsao, G.T. (1980)Biotechnol. Bioeng. 22, 2305.
Lee, K.M., Blaghen, M., Samama, J.-P., and Biellmann, J.-F. (1986)Bioorg. Chem. 14, 202–210.
Pfeuffer, E., Dreher, R.-M., Metzger, H., and Pfeuffer, T. (1985)Proc. Natl. Acad. Sci. USA, 82, 3086–3090.
Quicho, F.A. (1976)Methods Enzymol. 44, 546–558.
Woodward, J., Whaley, K.S., Zachry, G.S., and Wohlpart, D.L. (1981)Biotech. Bioeng. Symp. 11, 619–629.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Baker, J.O., Oh, K.K., Grohmann, K. et al. Thermal stabilization of fungal β-glucosidase through glutaraldehyde crosslinking. Biotechnol Lett 10, 325–330 (1988). https://doi.org/10.1007/BF01026159
Issue Date:
DOI: https://doi.org/10.1007/BF01026159