Abstract
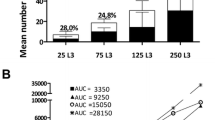
We carried out a comparative study of the dynamics of the sporocyst population and the growth rate of the host habitat (digestive and genital gland) within theSchistosoma mansoni/Biomphalaria glabrata system during monomiracidial infections. The volume of the digestive gland increased sharply during the first 2 weeks of infection and then decreased at 2–4 weeks post-infection during the exponential growth phase of the sporocyst infrapopulation. The effects of the parasite on the development of the host's genital gland resulted in a slowing of the growth of the ovotestis. These interactions, related to the demographic processes involved in the intramolluscal development of the parasite population, were interpreted in relation to the spatial and energy constraints imposed by the developmental needs of both the parasite and the host.
Similar content being viewed by others
References
Bayne CJ, Loker ES (1987) Survival within the snail host. In: Rollinson D, Simpson AJG (eds) The biology of schistosomes: from genes to latrines. Academic Press London, pp 321–346
Becker W (1980) Metabolic interrelationships of parasitic trematodes and molluscs, especiallySchistosoma mansoni inBiomphalaria glabrata. Z Parasitenkd 63:101–111
Carter OS, Bogitsh BJ (1975) Histologic and cytochemical observations of the effects ofSchistosoma mansoni onBiomphalaria glabrata. Histol Cytochem 266:380–393
Cheng TC, Snyder RW (1963) Studies on host-parasite relationships between larval trematodes and their hosts: IV. A histochemical determination of glucose and its role in the metabolism of molluscan host and parasite. Trans Am Microsc Soc 82:342–346
Cheng TC, James HA (1960) The histopathology ofCrepidostomum sp. infection in the second intermediate host,Sphaerium striatinum. Proc Helminthol Soc Wash 27:67–68
Cheng TC, Lee FO (1971) Glucose levels in the molluscBiomphalaria glabrata infected withSchistosoma mansoni. J Invertebr Pathol 18:395–399
Christie JD, Foster WB, Stauber LA (1974) The effect of parasitism and starvation on carbohydrate reserves ofBiomphalaria glabrata. J Invertebr Pathol 23:55–62
Coustau C, Renaud F, Delay B, Robbins I, Mathieu M (1991) Mechanisms involved in parasitic castration: in vitro effects of the trematodeProsorhynchus squamatus on the gametogenesis and the nutrient storage metabolism of the marine bivalve molluscMytilus edulis. Exp Parasitol 73:36–43
Crews AE, Yoshino TP (1989)Schistosoma mansoni: effects of infection on reproduction and gonadal growth inBiomphalaria glabrata. Exp Parasitol 68:326–334
De Jong-Brink M, Elsaadany MM, Boer HH (1988a)Trichobilharzia ocellata: interference with endocrine control of female reproduction ofLymnaea stagnalis. Exp Parasitol 65:91–100
De Jong-Brink M, Elssadany MM, Boer HH (1988b) Schistosomin, an antagonist of calfluxin. Exp Parasitol 65:109–118
Fryer SE, Oswald RC, Probert AJ, Runham NW (1990) The effect ofSchistosoma haematobium infection on the growth and fecundity of three sympatric species of bulinid snails. J Parasitol 76:557–563
Hurd H (1990) Physiological and behavioural interactions between parasites and invertebrate hosts. Adv Parasitol 29:271–318
James BL (1965) The effects of parasitism by larval Digenea on the digestive gland of the intertidal prosobranch,Littorina saxatilis (Olivi) subsp-tenebrosa (Montagu). Parasitology 55:93–115
Joosse J, Van Elk R (1986)Trichobilharzia ocellata: physiological characterization of giant growth, glycogen depletion, and absence or reproductive activity in the intermediate snail host,Lymnaea stagnalis. Exp Parasitol 62:1–13
Jourdane J, Théron A, Combes C (1980) Demonstration of several sporocyst generations as a normal patterns of reproduction ofSchistosoma mansoni. Acta Trop (Basel) 37:177–182
Looker DL, Etges FJ (1979) Effect ofSchistosoma mansoni infection on fecundity and pervitelline fluid composition inBiomphalaria glabrata. J Parasitol 65:880–885
Martoja R, Martoja-Pierson M (1967) Initiation aux techniques de l'histologie animale. Masson et Cie, Paris, p 344
McClelland G, Bourns TKR (1969) Effect ofTrichobilharzia ocellata on growth, reproduction and survival ofLymnaea stagnalis. Exp Parasitol 24:137–146
Meier M, Meier-Brook C (1981)Schistosoma mansoni: effect on growth, fertility, and development of distal male organs inBiomphalaria glabrata exposed to miracidia at different ages. Z Parasitenkd 66:121–131
Meuleman EA (1972) Host-parasite relationships between the freshwater pulmonateBiomphalaria pfeifferi and the trematodeSchistosoma mansoni. Neth J Zool 22:355–427
Minchella DJ, Loverde PT (1981) A cost of increased early reproductive effort in the snailBiomphalaria glabrata. Am Naturalist 118:876–881
Mohamed K, Ishak MM (1981) Growth rate and changes in tissue carbohydrates during schistosome infection of the snailBiomphalaria alexandrina. Hydrobiologia 76:17–21
Nassi H (1990) Le complexeBiomphalaria glabrata-Digènes en Guadeloupe. Contribution à l'étude de la castration des mollusques par les digènes larvaires. PhD Thesis, University of Perpignan
Pan CT (1965) Studies on the host-parasite relationships betweenSchistosoma mansoni andAustralorbis glabratus. Am J Trop Med Hyg 14:931–976
Schallig HDFH, Sassen JM, Hordijk PL, De Jong-Brink M (1991)Trichobilharzia ocellata: influence of infection on the fecundity of its intermediate snail hostLymnaea stagnalis and cercarial induction of the release of schistosomin, a snail neuropeptide antagonizing female gonadotropic hormones. Parasitology 102:85–91
Schwanbek A, Becker W, Rupprecht H (1986) Quantification of parasite development in the host-parasite systemBiomphalaria glabrata andSchistosoma mansoni. Z Parasitenkd 72:365–373
Schwartz CF, Carter OS (1982) Effect ofSchistosoma mansoni on glycogen synthase and phosphorylase fromBiomphalaria glabrata (Mollusca). J Parasitol 68:236–242
Théron A (1981) Dynamics of larval population ofSchistosoma mansoni inBiomphalaria glabrata: II. Chronobiology of the intramolluscan larval development during the shedding period. Ann Trop Med Parasitol 75:547–554
Théron A, Moné H, Gérard C (1992) Spatial and energy compromise between host and parasite: theBiomphalaria-glabrata Schistosoma mansoni system. Int J Parasitol 22:91–94
Thompson SN, Mejia-Scales V (1989) Effects ofSchistosoma mansoni on the nutrition of its intermediate host,Biomphalaria glabrata. J Parasitol 75:329–332
Thornhill SA, Jones JT, Kusel JR (1986) Increased oviposition and growth in immatureBiomphalaria glabrata after exposure toSchistosoma mansoni. Parasitology 93:443–450
Zischke JA, Zischke DP (1965) The effect ofEchinostoma revolutum larval infection on the growth and reproduction of the snail hostStagnicola palustris. Am Zool 5:707–708
Author information
Authors and Affiliations
Additional information
Financially supported by the UNDP World Bank/WHO Special Program for Research and Training in Tropical Diseases and by the CNRS (Sciences de la Vie)
Rights and permissions
About this article
Cite this article
Théron, A., Gérard, C. & Moné, H. Early enhanced growth of the digestive gland ofBiomphalaria glabrata infected withSchistosoma mansoni: side effect or parasite manipulation?. Parasitol Res 78, 445–450 (1992). https://doi.org/10.1007/BF00931703
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00931703