Summary
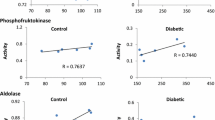
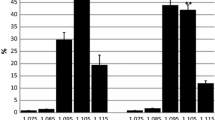
The (Na+-K+)ATPase and (Mg2+)ATPase activities of erythrocyte membranes of Type 1 (insulin-dependent) diabetic patients were found to be significantly reduced compared to matched controls (p < 0.005). On the contrary, erythrocyte Na+ and K+ contents were similar in diabetic patients and in normal subjects. When erythrocyte membranes from diabetic patients were incubated with their own plasma, a significant increase was observed in sodium-potassium ATPase activity (p < 0.005), whereas (Mg2+)ATPase activity was not affected. The plasma stimulatory effect showed saturation kinetics. Maximum average stimulation was 96% (±21.3). A similar stimulation pattern, although more limited in extent (maximum 48.3 % ± 12.2), was found when erythrocyte membranes from normal subjects were incubated with diabetic plasma. Normal plasma exhibited a modest stimulatory effect on erythrocyte (Na+-K+ATPase activity. Similar stimulatory effects by diabetic plasma were observed on a (Na+-K+) ATPase preparation from beef heart. It is proposed that diabetic plasma contains a specific (Na+-K+)ATPase activator in a higher concentration than normal plasma. This may explain why a normal cellular electrolyte content was found in diabetic erythrocytes in spite of a reduced Na+-K+ pump activity. Purification experiments indicate that the plasma activator is a protein with a molecular weight greater than 50,000. Both the (Na+-K+)ATPase activity and the stimulatory effect of diabetic plasma were not influenced by the metabolic control, since they did not correlate significantly with fasting blood glucose and daily insulin dosage. Moreover, no correlation was found with duration of diabetes or age at diagnosis of diabetes. It is suggested that the enzyme defect of erythrocyte membrane and the stimulation of (Na+-K+)ATPase activity by homologous plasma are early biochemical alterations in the course of diabetes mellitus.
Article PDF
Similar content being viewed by others
References
Pell S, D'Alonzo CA (1967) Some aspects of hypertension in diabetes mellitus. J Am Med Assoc 202: 104–110
Knowler WC, Bennett PH, Ballintine EJ (1980) Increased incidence of retinopathy in diabetics with elevated blood pressure. N Engl J Med 303: 645–650
O'Hare JA, Ferriss JB, Twonmey B, O'Sullivan DJ (1983) Poor metabolic control, hypertension and microangiopathy independently increase the transcapillary escape rate of albumin in diabetes. Diabetologia 25: 260–263
Dornan T, Mann JI, Turner R (1982) Factor protecting against retinopathy in insulin-dependent diabetics free of retinopathy for 30 years. Br Med J 285: 1073–1077
Dornan T, Ting A, McPherson CK, Peckar CO, Mann JI, Turner RC, Morns PJ (1982) Genetic susceptibility to the development of retinopathy in insulin-dependent diabetics. Diabetes 31: 226–231
Lauritzen T, Frost-Larsen K, Deckert T, Steno Study Group (1983) Effect of 1 year of near normal blood glucose levels on retinopathy in insulin-dependent diabetics. Lancet 1: 200–204
Viberti GC, Bilous RW, Mackintosh D, Bending JJ, Kenn H (1983) Long-term correction of hyperglycaemia and progression of renal failure in insulin-dependent diabetes. Br Med J 286: 598–602
Helgeland A, Laren P, Perfoss O, Hjermann I, Holme I, LundLarsen PG (1984) Serum glucose levels during long-term observation of treated and untreated men with mild hypertension. Am J Med 76: 802–805
West KM, Erdreich LJ, Stober JA (1980) A detailed study of risks factors for retinopathy and nephropathy in diabetes. Diabetes 29: 501–508
Handrup-Poulsen T, Mortensen SA, Heinertz H, Owerbach D, Johansen K, Sorensen H, Nerup P (1984) DNA sequences flanking the insulin gene on chromosome 11 confer risk of atherosclerosis. Lancet 1:250–252
Garay RP, Dagher G, Pernollet MG, Devynk MA, Meyer P (1980) Inherited defect in Na+, K+-co-transport system in erythrocytes from essential hypertensive patients. Nature 284: 281–283
Woods KL, Beevers DG, West M (1981) Familial abnormality of erythrocyte cation transport in essential hypertension. Br Med J 282:1186–1188
Poston L, Sewell RB, Wilkinson SP, Richardson PJ, Williams R, Clarkson EM, MacGregor GA, DeWardener HE (1981) Evidence for a circulating sodium transport inhibitor in essential hypertension. Br Med J 282: 847–849
Morgan DB, Cumberbatch M, Cohn S, Scott D, Gunasuntharam T, Davidson C, Chapman C (1980) The erythrocyte sodium and potassium in patients treated with digoxin. Br J Clin Pharmacol 10:127–133
Walter U (1981) Red blood cell sodium and potassium after hydrochlorothiazide. Clin Pharmacol Ther 30: 373–377
Kaji DM, Thakkar U, Kahn T (1981) Glucocorticoid-induced alterations in the sodium potassium pump of the human erythrocyte. J Clin Invest 68: 422–430
Raess BU, Vincenzi FF (1980) A semi-automated method for the determination of multiple membrane ATPase activities. J Pharmacol Meth 4: 273–283
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the folin reagent. J Biol Chem 193: 265–275
Fiske CH, Subbarow Y (1925) The colorimetric determination of phosphorus. J Biol Chem 66: 375–400
Finotti P, Palatini P (1981) Canrenone as a partial agonist at the digitalis receptor site of sodium-potassium-activated adenosine triphosphatase. J Pharmacol Exp Ther 217: 784–790
Chan PC, Kindy RJ, Kesner L (1977) Studies on the mechanism of ozone inactivation of erythrocyte membrane (Na+-K+)-activated ATPase. J Biol Chem 252: 8537–8541
Matsui H, Schwartz A (1966) Purification and properties of a highly active ouabain-sensitive Na+-K+-dependent adenosine triphosphatase from cardiac tissue. Biochim Biophys Acta 128: 380–390
Bradley JV (1968) Tests based on method of randomization. In: Distribution-free statistical tests. Prentice Hall, Englewood Cliffs, NJ, pp 96–129
De Wardener HE, MacGregor GA (1982) The natriuretic hormone and essential hypertension. Lancet 1:1450–1454
David-Dufilho M, Devynk MA, Beugras JP, Meyer P (1984) Quantitative changes in cardiac Na+-, K+-adenosine triphosphatase of spontaneously hypertensive rats. J Cardiovasc Pharmacol 6:273–280
Greene DA, Lattimer SA (1984) Action of sorbinil in diabetic peripheral nerve. Diabetes 33: 712–716
Pierce GN, Dhalla NS (1983) Sarcolemmal Na+-K+-ATPase activity in diabetic rat heart. Am J Physiol 243 (3): C241-C247
Baba Y, Motoakik, Kamada T, Setoyama S, Otsuji S (1979) Higher levels of erythrocyte membrane microviscosity in diabetes. Diabetes 28:1138–1140
Bruni A, Palatini P (1982) Biological and pharmacological properties of phospholipids. Prog Med Chem 19: 111–203
Shanahan MF, D'Artel-Ellis J (1984) Orientation of the glucose transporter in the human erythrocyte membrane. J Biol Chem 259:13878–13884
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Finotti, P., Palatini, P. Reduction of erythrocyte (Na+-K+)ATPase activity in Type 1 (insulin-dependent) diabetic subjects and its activation by homologous plasma. Diabetologia 29, 623–628 (1986). https://doi.org/10.1007/BF00869260
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00869260