Summary
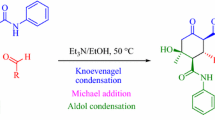
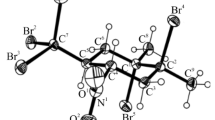
Base catalyzedMichael addition of 5-nitropentan-2-one ethylene ketal (1) and cyclohex-2-enone (2), subsequent deprotection, and intramolecular aldol condensation yields the 8-methyl-5-nitro-1-octalone isomers (5a,b). The structure, relative configuration, and conformation of5a and5b were elucidated utilizing the results of1H and13C NMR investigations
Zusammenfassung
BasenkatalysierteMichael-Addition von 5-Nitropentan-2-on-ethylenketal (1) und Cyclohex-2-enon (2), anschließende Entfernung der Schutzgruppe und darauffolgende Aldolkondensation liefert isomer 8-Methyl-5-nitro-1-octalone (5a,b). Struktur, relative Konfiguration und Konformation von5a und5b wurden mittels1H- und13C-NMR-Spektroskopie aufgeklärt.
Similar content being viewed by others
References
Cook CE, Whichard LP, Wall ME, Egley GH, Coggon P, Luhan PA, McPhail AT (1972) J Am Chem Soc94: 6198
Pepperman AB, Connick WJ, Vail SL, Worsham AD, Pavlista AD, Moreland DE (1982) Weed Sci 561
Kádas I, Árvai G, Töke L, Tóth G, Szöllösy Á, Bihari M (1994) Tetrahedron50: 2895
Tóth G, Halász J, Kádas I, Árvai G, Mikló K, Haeßner R (1995) Magn Reson Chem33: 184
Dewar MJS, Zoebisch EG, Healy EF, Stewart JJP (1985) J Am Chem Soc107: 3902
Haasnoot CAG, de Leeuw FAAM, Altona C (1980) Tetrahedron36: 2783
Subramanian S, Bax A (1987) J Magn Reson71: 325
Kinns M, Sanders JKM (1984) J Magn Reson56: 518
Bax A, Subramanian S (1986) J Magn Reson67: 565
ApSimon JW, Craid WG, Demarco PV, Mathieson DV, Saunders L, Whalley WB (1967) Tetrahedron23: 1967
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Halász, J., Tóth, G., Kádas, I. et al. Synthesis and stereochemistry of 8-methyl-5-nitro-1-octalones. Monatsh Chem 126, 1011–1019 (1995). https://doi.org/10.1007/BF00811021
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00811021