Summary
The morphology of the little skate (Raja erinacea) and spiny dogfish shark (Squalus acanthias) nephron has been investigated in sexually mature females by 1) gross observations of the kidney surfaces, 2) vascular injections, 3) scanning electron microscopy, 4) light microscopy.
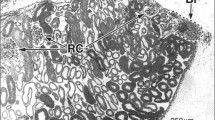
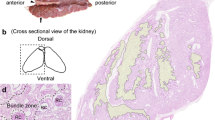
In the little skate, each nephron is highly complex and begins at the urinary pole of the renal corpuscle, which is located between a thin, dorsalbundle zone and a thicker, ventralsinus zone. The nephron loops back and forth, repeatedly entering and exiting each zone. In thebundle zone, segments from each nephron form a bundle of 5 tubules (tubular bundle) which are arranged in a countercurrent loop fashion. A peritubular sheath composed of closely packed, squamous cells wraps the 5 nephron segments of the tubular bundle together and separates each bundle from the next. In thesnus zone the tubules from many nephrons mix freely with each other in large blood sinuses.
In the spiny dogfish, the nephron displays a complex pattern similar to that of the skate. Renal corpuscles are adjacent to abundle zone composed of tubular bundles, each wrapped by a peritubular sheath in a cell-rich connective tissue matrix. However, thebundle zone is not limited to the dorsal region of the shark kidney but extends ventrally along deep interlobular septa. Thesinus zone of the shark is like that of the skate except that it is not limited to the ventral regions of the kidney.
Similar content being viewed by others
Refences
Baglioni S (1906) Einige Daten zur Kenntnis der quantitativen Zusammensetzung verschiedener Körperflüssigkeiten von Seetieren. Beitr Chem Physiol Pathol 9:50–66
Bargmann W (1937) Untersuchungen über Histologie und Histophysiologie der Fischniere. II. Selachier. Z Zellforsch 26:764–788
Bargmann W, von Hehn G (1971) Über das Nephron der Elasmobranchier. Z Zellforsch 114:1–21
Borcea J (1904) Quelques considérations sur l'appareil urinaire des Elasmobranches. Bull Soc Zool France 29:143–148
Borcea K (1906) Recherches sur le système uro-génital des Elasmobranches. Arch Zool Expér Gén Sér 44:199–484
Borghese E (1966) Studies on the nephrons of an elasmobranch fishScyliorhinus stellaris (L.). Z Zellforsch 72:88–99
Boylan JW (1972) A model for passive urea reabsorption in the elasmobranch kidney. Comp Biochem Physiol 42:27–30
Bulger RE (1965) The fine structure of the aglomerular nephron of the toadfish,Opsanus tau. Am J Anat 117:171–192
Bulger RE, Trump BF (1968) Renal morphology of the English sole (Parophrys vetulus). Am J Anat 123:195–226
Creaser CW (1927) The skate, a laboratory manual. Macmillan, New York
Daniel JF (1934) The elasmobranch fishes. Univ of California Press, Berkeley
Davis LE, Schmidt-Nielsen B, Stolte H (1976) Anatomy and ultrastructure of the excretory system of the lizard,Sceloporus cyanogenys. J Morphol 149:279–326
Deetjen P, Antkowiak D, Boylan JW (1970) The nephron of the skateRaja erinacea. Bull Mt Desert Is Biol Lab 10:5–7
Fange R (1963) Structure and function of the excretory organs of myxinoids. In: Brodal A, Fange R (eds) The biology of Myxine. Universitets forlaget, Oslo, p 516–519
Forster RP (1975) Structure and function of aglomerular kidneys. In: Wessing A (ed) Excretion. Fischer, Stuttgart, p 232–247
Forster RP, Goldstein L, Rosen JK (1972) Intrarenal control of urea reabsorption by renal tubules of the marine elasmobranchSqualus acanthias. Comp Biochem Physiol 42A:3–12
Ghouse MA, Bahram P, Boylan JW (1968) The anatomy, microanatomy and ultrastructure of the kidney of the dogfish,Squalus acanthias. Bull Mt Desert Is Biol Lab 8:22–29
Haller B (1902) Über die Urniere vonAcanthias vulgaris ein Beitrag zur Kenntnis sekundärer Metamerie. Gegenbaurs Morphol Jahrb 29:283–316
Heath-Eves MJ, McMillan DB (1974) The morphology of the kidney of the atlantic hagfishMyxine glutinosa (L.). Am J Anat 139:309–334
Hickman CP, Trump BF (1969) The kidney. In: Hoar WS, Randall DJ (eds) Fish physiology. Vol 1 Academic Press, New York, p 91–239
Karnovsky MJ (1965) A formaldehyde-glutaraldehyde fixative of high osmolarity for use in electron microscopy. J Cell Biol 27:137A-138A
Karnovsky MJ (1971) Use of ferrocyanide-reduced osmium tetroxide in electron microscopy. Am Soc Cell Biol, Proc 11th Annual Meeting, New Orleans, p 146
Kempton RT (1937) The dimensions of the renal tubules ofNecturus maculosus. J Morphol 61:51–58
Kempton RT (1939) The morphology of the dogfish renal tubule. Bull Mt Desert Is Biol Lab 42:28–34
Kempton RT (1940) The site of acidification of urine within the renal tubule of the dogfish. Bull Mt Desert Is Biol Lab 42:34–35
Kempton RT (1943) Studies on the elasmobranch kidney. I. The structure of the renal tubule of the spiny dogfish (Squalus acanthias). J Morphol 73:247–263
Kempton RT (1953) Studies on the elasmobranch kidney. II. Reabsorption of urea by the smooth dogfish,Mustelus canis. Biol Bull 104:45–56
Kempton RT (1956) The problem of the “special segment” of the elasmobranch kidney tubule. Year Book Am Phil Soc 210–212
Kempton RT (1957) The elasmobranch kidney tubule. Anat Rec 128:575
Kempton RT (1962) Studies on the elasmobranch kidney. III. The kidney of the lesser rayNarcine brasiliensis. J Morphol 111:217–225
Kempton RT (1964) Some anatomical features of the elasmobranch kidney. Biol Bull 127:377
Kinter WB (1975) Structure and function of renal tubules isolated from fish kidneys. In: Wessing A (ed) Excretion. Fischer Stuttgart, p 221–231
Kozlik F (1939) Zum Bau des Nierenkanälchens der Selachier. Z Anat 109:624–648
Krukenberg CFW (1888) La rétention de l'urée chez les Sélaciens. Ann Mus Hist Nat 3. Mém 1–43
Kühn K, Stolte H, Reale E (1975) The fine structure of the kidney of the hagfish (Myxine glutinosa L.) Cell Tissue Res 164:201–213
Lacy ER, Reale E (1981) The brush border segment of the nephron in an elasmobranch. Thin section and freeze fracture observations. Verh Anat Ges 75:611–612
Lacy ER, Reale E (1985a) The elasmobranch kidney. II. Sequence and structure of the nephrons. Anat Embryol 173 (in press)
Lacy ER, Reale E (1985b) The elasmobranch kidney. III. Fine structure of the peritubular sheath. Anat Embryol 173 (in press)
Lacy ER, Reale E, Schlusselberg D, Smith W, Woodward D (1985) A renal countercurrent system in marine elasmobranch fish: A computer-assisted reconstruction. Science 227:1351–1354
Lacy ER, Schmidt-Nielsen B, Swenson E, Maren T (1975) The urinary bladder of little skateRaja erinacea. Bull Mt Desert Is Biol Lab 15:56–58
Malick LE, Wilson B, Stetson D (1975) Modified thiocarbohydrazide procedure for scanning electron microscopy: Routine use for normal, pathological or experimental tissues. Stain Technol 50:265–269
Marshall EK (1933) The secretion of urea in the frog. J Cell Comp Physiol 2:349–353
Marshall EK (1934) The comparative physiology of the kidney in relation to theories of renal secretion. Physiol Rev 14:133–159
Nash J (1931) The number and size of glomeruli in the kidneys of fishes with observations on the morphology. Am J Anat 47:425–445
Ogawa M, Hirano T (1982) Studies on the nephron of a fresh water stingray,Potamotrygon magdalenae. Zool Mag 91:101–105
Romer AS, Parsons TS (1977) The vertebrate body. Saunders, Philadelphia London Toronto
Sakai T, Kawahara K (1983) The structure of the kidney of Japanese newts,Triturus (Cynops) pyrrhogaster. Anat Embryol 166:31–52
Schmidt-Nielsen B, Ullrich KJ, Rumrich G, Long NS (1966) Micropuncture study of urea movements across the renal tubules ofSqualus acanthias. Bull Mt Desert Is Biol Lab 6:35
Schmidt-Nielsen K (1979) Animal physiology: Adaptation and environment. 2nd ed. Cambridge University Press, Cambridge
Schroeder W (1890) Über die Harnstoffbildung der Haifische. Hoppe-Seylers Z Physiol Chem Phys 14:576–598
Smith HW (1931a) The absorption and excretion of water and salts by the elasmobranch fish. I. Fresh water elasmobranches. Am J Physiol 98:279–295
Smith HW (1931b) The absorption and excretion of water and salts by the elasmobranch. II. Marine elasmobranches. Am J Physiol 98:296–310
Smith HW (1936) The retention and physiological role of urea in elasmobranchii. Biol Rev 11: 49–82
Staedeler G, Frerichs FT (1858) Über das Vorkommen von Harnstoff, Taurin und Scyllit in der Organen der Plagiostomen. J Prakt Chem 73:48–55
Stolte H Eisenbach GM, Antkowiak D, Boylan JW (1971) Renal collecting duct function in the little skate,Raja erinacea. Bull Mt Desert Is Biol Lab 11:91–93
Stolte H, Galaske RG, Eisenbach GM, Lechene C, Schmidt-Nielsen B, Boylan JW (1977) Renal tubule ion transport and collecting duct function in the elasmobranch little skate,Raja erinacea. J Exp Zool 199:403–410
Thurau K, Acquisto P (1969) Localization of the diluting segment in the dogfish nephron: A micropuncture study. Bull Mt Desert Is Biol Lab 9:60–63
Torrey TW (1965) Morphogenesis of the vertebrate kidney. In: DeHann RL, Ursprung H (eds) Organogenesis, Holt Rinehart Winston, New York, p 559–579
Weichert CK (1959) Elements of chordate anatomy. McGraw-Hill, New York Toronto London
Youson JH (1970) The opisthonephric kidney of the sea lamprey of the Great Lakes,Petromyzon marinus L. II. Neck and proximal segments of the tubular nephron. Am J Anat 127:233–258
Youson JH (1971) The opisthonephric kidney of the sea lamprey of the Great Lakes,Petromyzon marinus L. IV. Intermediate, distal and collecting segments of the adult. Am J Anat 130:281–304
Zarnik B (1910) Verglelchende Studien über den Bau der Niere von Echidna und der Reptilienniere. Jena Z Naturwiss 45:113–224
Author information
Authors and Affiliations
Additional information
Supported in part by National Institutes of Health AM 06345 and by Deutsche Forschungsgemeinschaft (Sonderforschungsbereich 146)
Alexander von Humboldt Fellow
Rights and permissions
About this article
Cite this article
Lacy, E.R., Reale, E. The elasmobranch kidney. Anat Embryol 173, 23–34 (1985). https://doi.org/10.1007/BF00707301
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00707301