Abstract
-
1.
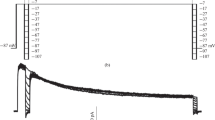
In voltage clamped nodes of Ranvier inactivation of the sodium permeability is slowed by toxin V from the scorpionCentruroides sculpturatus, by sea anemone toxin ATX II or by internally applied KIO3. The slow decay of the Na inward current is markedly accelerated if the test pulse is preceded by a depolarizing conditioning pulse followed by a 10–500 ms pause. This phenomenon was studied in detail, using conditioning pulses of varying amplitude and up to 15 s duration.
-
2.
In nodes treated with toxin V a 20 ms conditioning pulse to positive potentials was sufficient to produce a clear acceleration of the decay of the Na current and a reduction of the inward current remaining at the end of a 50 ms test pulse, i.e. a weakening of the toxin effect. In nodes treated with ATX II or internal KIO3 longer conditioning pulses were required. A similar effect of conditioning pulses on the decaying phase of the Na current was also observed in untreated fibres.
-
3.
To study the phenomenon quantitatively we fitted the decaying phase of the inward Na current with the equationI Na=A exp(-t/τ1)+B exp(-t/τ2)+C The effect of depolarizing conditioning pulses could be described as an increase of A, a decrease of B and C and a reduction of the time constants τ1 and τ1.
-
4.
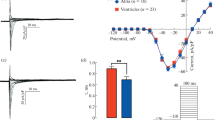
I 50/I peak, the normalised inward current remaining at the end of a 50 ms test pulse, decreased exponentially with increasing duration of the conditioning pulse to a steady-state value. The time constant τ and the steady-state value depended on the potential during the conditioning pulse. For nodes treated with toxin V, τ was 0.24 s at 0 mV and 12° C and half inhibition occurred at −42 mV. The time constant τ was larger for nodes treated with ATX II or internal KIO3. At positive potentials, I50 was reduced to 20% of the control value in toxin V-treated nodes, but only to 70% in KIO3-treated nodes.
-
5.
Recovery from the effect of the conditioning pulse was studied by varying the pause between conditioning pulse and test pulse; recovery was 66–100% complete after 500 ms.
-
6.
The results are interpreted by assuming that a sepolarizing conditioning pulse (a) accelerates inactivation of the sodium permeability and (b) causes dissociation of the toxin-receptor complex or transition into an inactive state. The latter effect occurs in toxin V-treated fibres but not in those treated with ATX II or KIO3.
Similar content being viewed by others
References
Bergman C, Dubois JM, Rojas E, Rathmayer W (1976) Decreased rate of sodium conductance inactivation in the node of Ranvier induced by a polypeptide toxin from sea anemone. Biochim Biophys Acta 455:173–184
Bullock JO, Schauf CL (1979) Immøbilization of intramembrane charge inMyxicola giant axons. J Physiol 286:157–171
Catterall WA (1977) Membrane potential-dependent binding of scorpion toxin to the action potential Na+ ionophore. J Biol Chem 252:8660–8668
Catterall WA (1979) Binding of scorpion toxin to receptor sites associated with sodium channels in frog muscle. J Gen Physiol 74:375–391
Chandler WK, Meves H (1970) Evidence for two types of sodium conductance in axons perfused with sodium fluoride solution. J Physiol 211:653–678
Chiu SY (1977) Inactivation of sodium channels: second order kinetics in myelinated nerve. J Physiol 273:573–596
Corbier A, Dubois JM (1983) Two transient Na currents in the frog node of Ranvier. J Physiol 345:79 P
Frankenhaeuser B (1963) Inactivation of the sodium-carrying mechanism in myelinated nerve fibres ofXenopus laevis. J Physiol 169:445–451
French RJ, Worley JF III, Krueger BK (1984) Voltage-dependent block by saxitoxin of sodium channels incorporated into planar lipid bilayers. Biophys J 45:301–310
Goldman L (1976) Kinetics of channel gating in excitable membranes. Q Rev Biophys 9:491–526
Hahin R, Goldman L (1978) Initial conditions and the kinetics of the sodium conductance inMyxicola giant axons. I. Effects on the time-course of the sodium conductance. J Gen Physiol 72:863–877
Hille B (1977) Local anesthetics: hydrophilic and hydrophobic pathways for the drug-receptor reaction. J Gen Physiol 69:497–515
Hu SL, Meves H, Rubly N, Watt DD (1983) A quantitative study of the action ofCentruroides sculpturatus toxins III and IV on the Na currents of the node of Ranvier. Pflügers Arch 397:90–99
Koppenhöfer E (1967) Die Wirkung von Tetraäthylammoniumchlorid auf die Membranströme Ranvierscher Schnürringe vonXenopus laevis. Pflügers Arch 293:34–55
Meves H, Rubly N (1984) Effect of a depolarizing conditioning pulse on the time course of sodium inactivation in myelinated nerve fibres. Pflügers Arch 400:R 38
Meves H, Rubly N, Watt DD (1982) Effect of toxins isolated from the venom of the scorptionCentruroides sculpturatus on the Na currents of the node of Ranvier. Pflügers Arch 393:56–62
Meves H, Rubly N, Watt DD (1983) Potential-dependent action of a scorpion toxin on the sodium current of the frog myelinated nerve fibre. J Physiol 345:77 P
Mozhayeva GN, Naumov AP (1980) Kinetics of interaction of scorption toxin with sodium channels in the Ranvier node membrane. Neurophysiology 12:409–415
Mozhayeva GN, Naumov AP, Nosyreva ED, Grishin EV (1980) Potential-dependent interaction of toxin from venom of the scorpionButhus eupeus with sodium channels in myelinated fibre. Voltage clamp experiments. Biochim Biophys Acta 597:587–602
Neumcke B, Schwarz W, Stämpfli R (1980) Modification of sodium inactivation in myelinated nerve byAnemonia toxin II and iodate. Analysis of current fluctuations and current relaxations. Biochim Biophys Acta 600:456–466
Nonner W (1969) A new voltage clamp method for Ranvier nodes. Pflügers Arch 309:176–192
Nonner W (1980) Relations between the inactivation of sodium channels and the immobilization of gating charge in frog myelinated nerve. J Physiol 299:573–603
Schwarz JR, Bromm B, Spielmann RP, Weytjens JLF (1983) Development of Na inactivation in motor and sensory myelinated nerve fibres ofRana esculenta. Pflügers Arch 398:126–129
Stämpfli R (1974) Intraaxonal iodate inhibits sodium inactivation. Experientia 30:505–508
Stämpfli R, Hille B (1976) Electrophysiology of the peripheral myelinated nerve. In: Llinas R, Precht W (eds) Frog neurobiology. Springer, Berlin Heidelberg New York, pp 3–32
Ulbricht W, Schmidtmayer J (1981) Modification of sodium channels in myelinated nerve byAnemonia sulcata toxin II/J Physiol (Paris) 77:1103–1111
Vincent JP, Balerna M, Barhanin J, Fosset M, Lazdunski M (1980) Binding of sea anemone toxin to receptor sites associated with gating system of sodium channel in synaptic nerve endingsin vitro. Proc Natl Acad Sci USA 77:1646–1650
Warashina A, Fujita S (1983) Effect of sea anemone toxins on the sodium inactivation process in crayfish axons. J Gen Physiol 81:305–323
Warashina A, Fujita S, Satake M (1981) Potential-dependent effects of sea anemone toxins and scorpion venom on crayfish giant axon. Pflügers Arch 391:273–276
Watt DD, Simard JM, Mancuso PM (1982) Oscillation in resting tension of chick skeletal muscle treated with toxin V from scorpion venom, reduced [calcium]0 and cadium. Comp Biochem Physiol 71 A:375–382
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Meves, H., Rubly, N. & Watt, D.D. Voltage-dependent effect of a scorpion toxin on sodium current inactivation. Pflugers Arch. 402, 24–33 (1984). https://doi.org/10.1007/BF00584827
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00584827