Summary
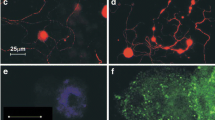
The proposal that the bizarre behavioral changes which occur during rabies infection are due to selective infection of limbic system neurons was further studied in skunks (a species important in naturally occurring disease). A detailed immunohistochemical study of brains of skunks experimentally infected with either Challenge virus standard (CVS) or street rabies virus revealed only trace amounts of viral antigen in many limbic system neurons and marked differences in viral distribution between street and CVS virus. These data were collected during early stage rabies when behavioral changes occur. Areas which contained heavy accumulations of street rabies virus but low amounts of CVS rabies virus were the neuronal perikarya and processes of the dorsal motor nucleus of the vagus, midbrain raphe, hypoglossal and red nuclei. In contrast, large accumulations of CVS virus were found in the Purkinje cells of the cerebellum, the habenular nuclei and in pyramidal cells throughout the cerebral cortex, while corresponding areas in all street virus-infected skunks contained minimal antigen. These findings were very consistent for animals of the same experimental group and between skunks inoculated both intramuscularly and intranasally with skunk street virus. Skunks inoculated intramuscularly with CVS rabies virus failed to develop rabies. Since, in this model, street virus infection generally produces furious rabies and CVS infection results in dumb rabies, we speculate that the behavioral changes which occur in these two different clinical syndromes are due to the heavy and specific accumulation of virus in different regions of the CNS. These results show that regions other than those of the limbic system may also be involved in the pathogenesis of behavior changes in rabid animals.
Similar content being viewed by others
References
Baer GM (1975) Pathogenesis to the central nervous system. In: Baer GM (ed) The natural history of rabies. Academic Press, New York, pp 185–197
Bourgon AR, Charlton KM (1987) The demonstration of rabies antigen in paraffin-embedded tissues using the peroxidase antiperoxidase method: a comparative study. Can J Vet Res 51: 117–120
Charlton KM (1989) The Pathogenesis of Rabies. In: Campbell JB, Charlton KM (eds) Rabies, 1st edn. Kluwer, New York, pp 101–150
Charlton KM, Casey GA, Campbell JB (1984) Experimental rabies in skunks: effects of immunosuppression induced by cyclophosphamide. Can J Comp Med 48: 72–77
Charlton KM, Webster WA, Casey GA, Rupprecht CE (1988) Skunk rabies. Rev Infect Dis 10: S626-S628
Dietzschold D, wiktor TJ, Trojanowski JQ, Macfarlan RI, Wunner WH, Torres-Anjel MJ, Koprowski H (1985) Differences in cell-to-cell spread of pathogenic and apathogenic rabies virus in vivo and in vitro. J Virol 56: 12–18
Feiden W, Feiden U, Gerhard L, Reinhardt V, Wandeler A (1985) Rabies encephalitis: immunohistochemical investigations. Clin Neuropathol 4: 156–164
Feiden W, Kaiser E, Gerhard L, Dahme E, Gylstorff B, Wandeler A, Ehrensberger E (1988) Immunohistochemical staining of rabies virus antigen with monoclonal and polyclonal antibodies in paraffin tissue sections. J Vet Med [B] 35: 247–255
Fekadu M, Chandler FW, Harrison AK (1982) Pathogenesis of rabies in dogs inoculated with an Ethiopian rabies virus strain. Immunofluorescence, histologic and ultrastructural studies of the central nervous system. Arch Virol 71: 109–126
Gourmelon P, Briet D, Court L, Tsiang H (1986) Electrophysiological and sleep alterations in experimental mouse rabies. Brain Res 398: 128–140
Hemachuda T, Phanuphak P, Sriwanthana B, Manutsathit S, Phanthumchinda K, Siripransomsup W, Ukachoke C, Rasameechan S, Kaoroptham S (1988) Immunologic study of human encephalitic and paralytic rabies. Am J Med 84: 673–677
Hsu SM, Raine L (1981) Protein A, avidin and biotin in immunohistochemistry. J Histochem Cytochem 29: 1349–1353
Iwasaki Y, Gerhard W, Clark HF (1977) Role of host immune response in the development of either encephalitic or paralytic disease after experimental rabies infection in mice. Infect Immun 18: 220–225
Jackson AC (1991) Biological basis of rabies virus neurovirulence in mice: comparative pathogenesis study using the immunoperoxidase technique. J Virol 65: 57–540
Jackson AC, Reimer DL (1989) Pathogenesis of experimental rabies in mice: an immunohistochemical study. Acta Neuropathol 78: 159–165
Johnson RT (1965) Experimental rabies. Studies of cellular vulnerability and pathogenesis using flourescent antibody staining. J Neuropathol Exp Neurol 24: 662–674
Siegel A, Brutus M (1990) Neural substrates of aggression and rage in the cat. Prog Psychobiol Physiol Psychol 14: 135–233
Sriwanthana B, Hemachudha T, Griffin DE, Manutsathit S, Tweardy D, Phanuphak P (1989) Lymphocyte subsets in human encephalitic and paralytic rabies. Acta Neurol Scand 80: 287–289
Tirawatnpong S, Hemachudha T, Manutsathit S, Shuangshoti S, Phanthumchinda K, Phanuphak P (1989) Regional distribution of rabies viral antigen in central nervous system of human encephalitic and paralytic rabies. J Neurol Sci 92: 91–99
Tsiang H, Guillon JC (1981) Presence of specific antigens in neuronal cells infected with fixed and street rabies virus strains. Acta Neuropathol (Berl) 55: 263–267
Valzelli L (1984) Reflections on experimental and human pathology of aggression. Prog Neuropsychopharmacol Biol Psychiatry 8: 311–325
Wunner WH, Larson JK, Dietzschold B, Smith CL (1988) The molecular biology of rabies viruses. Rev Infect Dis 10: S771-S784
Yamamoto T, Ueki S (1977) Characteristics in aggressive behavior induced by midbrain raphe lesions in rats. Physiol Behav 19: 105–110
Author information
Authors and Affiliations
Additional information
Supported by an MRC fellowship (NLS)
Rights and permissions
About this article
Cite this article
Smart, N.L., Charlton, K.M. The distribution of challenge virus standard rabies virus versus skunk street rabies virus in the brains of experimentally infected rabid skunks. Acta Neuropathol 84, 501–508 (1992). https://doi.org/10.1007/BF00304469
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00304469