Abstract
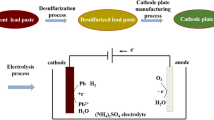
The dissolution of the active materials of lead-acid batteries in fluoboric electrolyte has been studied. The use of redox couples such as Ti3+/Ti4+ is proposed for an efficient and quick dissolution of lead and lead dioxide mixtures. For PbO2 and Pb electrodeposited on platinum electrodes the rate of dissolution in HBF4 (200 g dm−3) containing Ti ions (0.3m) corresponds to a current density of 400 A cm−2 and 160 A cm−2, respectively. Dissolved oxygen has a marked influence on lead dissolution acting as an oxidizer of Ti3+ to Ti4+. It has also been shown that the Fe2+/Fe3+ couple can be used, although with lower benefits. For industrial applications, a concentration of Ti ions of about 0.051m and the use of a counter-current electrolyte flow in the electrolysis cell can advantageously accomplish the leaching process of pastes and slimes in a batteries recycling plant.
Similar content being viewed by others
References
R. D. Pregaman and H. McDonald, U.S. Patent 4 229 271 (1980).
M. Olper and P. Fracchia, Eur. Pat. Appl. 0 313 153 A1 (1989).
M. Maja, P. Spinelli, N. Penazzi, M. V. Ginatta, U. Ginatta and G. Orsello, ‘EFCE Publication 51’, Symposium Series 98, The Institute of Chemical Engineering (1986).
M. Maja, N. Penazzi, M. Baudino and M. V. Ginatta, J. Power Sources 31 (1990) 287.
M. Olper and P. Fracchia, Ital. Patent 1 188 203 (1985).
M. Olper, MABat 91, Warsaw, Poland 5 (dy1991).
C. E. Tucker, U.S. Patent 1 148 062 (1915).
W. C. Smith, U.S. Patent 1 752 356 (1930).
J. H. Calbeck, U.S. Patent 1 911 604 (1933).
A. F. Gaumann, U.S. Patent 4 107 007 (1978).
M. F. Elmore, U.S. Patent 4 118 219 (1978).
U. Ducati, U.S. Patent 4 460 442 (1984).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Maja, M., Bodoardo, S., Serracane, C. et al. Dissolution of pastes in lead-acid battery recycling plants. J Appl Electrochem 23, 819–826 (1993). https://doi.org/10.1007/BF00249955
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00249955