Summary
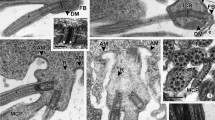
Transmission electron microscopy was used to study the development of the flagellar base and the flagellar necklace during spermatogenesis in a moth (Ephestia kuehniella Z.). Until mid-pachytene, two basal body pairs without flagella occur per cell. The basal bodies, which contain a cartwheel complex, give rise to four flagella in late prophase I. The cartwheel complex appears to be involved in the nucleation of the central pair of axonemal microtubules. In spermatids, there is one basal body; this is attached to a flagellum. At this stage, the nine microtubular triplets of the basal body do not terminate at the same proximal level. The juxtanuclear triplets are shifted distally relative to the triplets distant from the nuclear envelope. Transition fibrils and a flagellar necklace are formed at the onset of axoneme elongation. The flagellar necklace includes Y-shaped elements that connect the flagellar membrane and the axonemal doublets. In spindle-containing spermatocytes, the flagellar necklace is no longer detectable. During spermatid differentiation, the transition fibrils move distally along the axoneme and a prominent middle piece appears. Our observations and those in the literature indicate certain trends in sperm structure. In sperms with a short middle piece, we expect the presence of a flagellar necklace. The distal movement of the transition fibrils or equivalent structures is prevented by the presence of radial linkers between the flagellar membrane and the axonemal doublets. On the other hand, the absence of a flagellar necklace at the initiation of spermiogenesis enables the formation of a long middle piece. Thus, in spermatozoa possessing an extended middle piece, a flagellar necklace may be missing.
Similar content being viewed by others
References
Afzelius BA, Franzén Å (1971) The spermatozoon of the jellyfish Nausithoë. J Ultrastruct Res 37:186–199
Alkon DL (1983) Sensory function of cilia. J Submicrosc Cytol 15:173–176
Andrews D, Nelson DL (1979) Biochemical studies of the excitable membrane of Paramecium tetraurelia. II. Phospholipids of ciliary and other membranes. Biochim Biophys Acta 550:174–187
Azevedo C (1981) The fine structure of the spermatozoon of Patella lusitanica (Gastropoda: Prosobranchia), with special reference to acrosome formation. J Submicrosc Cytol 13:47–56
Azevedo C, Lobo-Da-Cunha A, Oliveira E (1985) Ultrastructure of the spermatozoon in Gibbula umbilicalis (Gastropoda, Prosobranchia), with special reference to acrosomal formation. J Submicrosc Cytol 17:609–614
Bardele CF (1981) Functional and phylogenetic aspects of the ciliary membrane: A comparative freeze-fracture study. Biosystems 14:403–421
Bergstrom BH, Henley C (1973) Flagellar necklaces: Freeze-etch observations. J Ultrastruct Res 42:551–553
Bergstrom BH, Henley C, Costello DP (1973) Particulate flagellar and ciliary necklaces revealed by the use of freeze-etch. Cytobios 7:51–60
Billard R, Fléchon J-E (1969) Spermatogonies et spermatocytes flagellés chez poecilia reticulata (Téléostéens Cyprinodonti-formes). Ann Biol Anim Biochim Biophys 9:281–286
Boisvieux-Ulrich E, Sandoz D, Chailley B (1977) A freeze-fracture and thin section study of the ciliary necklace in quail oviduct. Biol Cell 30:245–252
Burton PR (1981) Polymorphic assemblies of tubulin. In: Dowben RM, Shay JW (eds) Cell and Muscle Motility, vol 1. Plenum, New York, pp 289–333
Cavalier-Smith T (1974) Basal body and flagellar development during the vegetative cell cycle and the sexual cycle of Chlamydomonas reinhardii. J Cell Sci 16:529–556
Dirksen ER, Gilula NB, Davidson L, Schooley C, Satir B, Satir P (1971) New aspects of cilia structure. Anat Rec 169:464
Dustin P (1984) Microtubules. Springer, Berlin Heidelberg New York
Dute R, Kung C (1978) Ultrastructure of the proximal region of somatic cilia in Paramecium tetraurelia. J Cell Biol 78:451–464
Eddy EM (1970) Cytochemical observations on the chromatoid body of the male germ cells. Biol Reprod 2:114–128
Fain-Maurel M-A (1966) Acquisitions récentes sur les spermato-genèses atypiques. Ann Biol 5:513–564
Fawcett DW (1970) A comparative view of sperm ultrastructure. Biol Reprod 2:90–127
Fawcett DW, Eddy EM, Phillips DM (1970) Observations on the fine structure and relationships of the chromatoid body in mammalian spermatogenesis. Biol Reprod 2:129–153
Ferraguti M, Lanzavecchia G (1971) Morphogenetic effects of microtubules. I. Spermiogenesis in Annelida Tubificidae. J Submicrosc Cytol 3:121–137
Flower NE (1971) Particles within membranes: A freeze-etch view. J Cell Sci 9:435–441
Friedländer M, Wahrman J (1966) Giant centrioles in neuropteran meiosis. J Cell Sci 1: 129–144
Friedländer M, Wahrman J (1970) The spindle as a basal body distributor. A study in the meiosis of the male silkworm moth, Bombyx mori. J Cell Sci 7:65–89
Friedländer M, Wahrman J (1971) The number of centrioles in insect sperm: A study in two kinds of differentiating silkworm spcrmatids. J Morphol 134:384–398
Fritz-Niggli H, Suda T (1972) Bildung und Bedeutung der Zentriolen: Eine Studie und Neuinterpretation der Meiose von Drosophila. Cytobiologie 5:12–41
Gall JG (1961) Centriole replication. A study of spermatogenesis in the snail Viviparus. J Biophys Biochem Cytol 10:163–193
Gibbons IR (1981) Cilia and flagella of eukaryotes. J Cell Biol 91:107s-124s
Gilula NB, Satir P (1972) The ciliary necklace. A ciliary membrane specialization. J Cell Biol 53:494–509
Glaser RW (1917) cited in: Lokwood APM (1961) “Ringer” solutions and some notes on the physiological basis of their ionic composition. Comp Biochem Physiol 2:241–289
Hinsch GW, Clark WH (1973) Comparative fine structure of Cnidaria spermatozoa. Biol Reprod 8:62–73
Holm PB, Rasmussen SW (1980) Chromosome pairing, recombination nodules and chiasma formation in diploid Bombyx males. Carlsberg Res Commun 45:483–548
Jespersen Å(1971) Fine structure of the spermatozoon of the Australian lungfish Neoceratodus forsteri (Krefft). J Ultrastruct Res 37:178–185
Kalnins VT, Porter KR (1969) Centriole replication during ciliogenesis in the chick tracheal epithelium. Z Zellforsch 100:1–30
Phillips DM (1974) Spermiogenesis. Academic Press, New York
Pickett-Heaps JD (1969) The evolution of the mitotic apparatus: An attempt at comparative ultrastructural cytology in dividing plant cells. Cytobios 3:257–280
Pitelka DR (1974) Basal bodies and root structures. In: MA Sleigh (ed) Cilia and Flagella. Academic Press, London, pp 437–469
Rieder CL, Borisy GG (1982) The centrosome cycle in PtK2 cells: Asymmetric distribution and structural changes in the pericentriolar material. Biol Cell 44:117–132
Röhlich P (1975) The sensory cilium of retinal rods is analogous to the transitional zone of motile cilia. Cell Tissue Res 161:421–4130
Sakai A, Shigenaga M (1966) Fine structure of centrioles of the grasshoppers, Acrida turrita Linné and Atractomorpha bedeli Bolivar. Cytologia 32:105–110
Satir P, Satir B (1964) A model for ninefold symmetry in α keratin and cilia. J Theor Biol 7:123–128
Satir B, Sale WS, Satir P (1976) Membrane renewal after dibucaine deciliation of Tetrahymena. Exp Cell Res 97:83–91
Satir P, Wais-Steider J, Lebduska S, Nasr A, Avolio J (1981) The mechanochemical cycle of the dynein arm. Cell Motil 1:303–327
Silberglied RE, Shepherd JG, Dickinson JL (1984) Eunuchs: The role of apyrene sperm in Lepidoptera? Am Nat 123:255–265
Sleigh M (1974) Cilia and Flagella. Academic Press, London
Thibout E (1980) Evolution and role of apyrene sperm cells of Lepidopterans: Their activation and denaturation in the leek moth, Acrolepiopsis assectella (Hypomeutoidea). In: Clarke WH, Adams TS (eds) Adv Invertebr Reprod. Elsevier, Amsterdam, pp 231–242
Thompson GA, Bambery RJ, Nozawa Y (1972) Environmentally produced alterations of the tetrahymanol: Phospholipid ratio in Tetrahymena pyriformis membranes. Biochim Biophys Acta 260:630–638
Tokuyasu KT (1974) Dynamics of spermiogenesis in Drosophila melanogaster. III. Relation between axoneme and mitochondrial derivatives. Exp Cell Res 84:239–250
Troyer D, Cameron ML (1980) Spermiogenesis in lumbricid earthworms revisited. I. Function and fate of centrioles, fusion of organelles and organelle movements. Biol Cell 37:273–286
Vorobjev IA, Chentsov YuS (1983) The dynamics of reconstitution of microtubules around the cell center after cooling. Eur J Cell Biol 30:149–153
Warner FD (1981) Structure-function relationships in cilia and flagella. In: Harris JR (ed) Electron Microscopy of Proteins, vol 1. Academic Press, London, pp 301–344
Wheatley DN (1982) The centriole, a central enigma of cell biology. Elsevier, Amsterdam
Wolf K (1987) Cytology of Lepidoptera. I. The nuclear area in secondary oocytes of Ephestia kuehniella contains remnants of the first division. Eur J Cell Biol 43:223–229
Wolf K, Traut W (1987) Ultrastructure of distal flagellar swellings in spermatocytes and spermatids of Ephestia kuehniella (Pyralidae, Lepidoptera) Cell Tissue Res 250:421–424
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wolf, K.W., Kyburg, J. The restructuring of the flagellar base and the flagellar necklace during spermatogenesis of Ephestia kuehniella Z. (Pyralidae, Lepidoptera). Cell Tissue Res. 256, 77–86 (1989). https://doi.org/10.1007/BF00224720
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00224720