Abstract
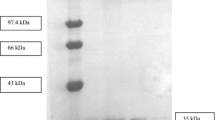
Several strains of Pseudomonas were screened for lipase production using the rhodamine agar diffusion test. On the basis of the diameter of the halo produced, P. fragi CRDA 323 and P. putida ATCC 795 were considered to be good and weak lipase producers respectively. P. fragi, cultured in a 2-1 fermenter, produced a maximal amount of lipase after 3–4 days of incubation at 27°C. The lipase extract of P. fragi was obtained by acidification of culture supernatant at pH 4.0 and partially purified with ammonium sulphate precipitation. The majority of lipase activity (42%) was located in fraction IV, precipitated at 20%–40% of saturation, with a 19-fold enzyme purification. The K m and V max values for the partially purified enzymatic extract (fraction IV) were 0.70 mg/ml and 0.97 × 10−3 U/min respectively. Fraction IV, which showed and optimum activity at pH 8.5, was used for the interesterification of butter fat in a microemulsion free co-surfactant system containing Span 60 (sorbitol monostearate) and Tween 60 (polyoxyethylene sorbitan monostearate) in the ratio 48:52 (v/v). The results showed that the interesterification of butter fat resulted in a considerable decrease in long-chain saturated fatty acids (C12:0, C14:0 and C16:0) with a concomitant increase in C18:0 and C18:1 at the sn-2 position of selected triacylglycerols. In addition, the results demonstrated an increase in the fatty acids (C12:0, C14:0 and 16:0) among the 1 and 3 positions of the triacylglycerol molecules of modified buffer fat accompanied by a decrease in C18:0 and C18:1.
Similar content being viewed by others
References
Abraham G, Murray MA, John VT (1988) Interesterification selectivity in lipase-catalysed reactions of low molecular weight triglycerides. Biotechnol Lett 10:555–558
Alford JA, Pierce DA, Suggs FG (1964) Activity of microbial lipases on natural fats and synthetic triglycerides. J Lipid Res 5:390–394
Badings HT, De Jong C (1983) Capillary gas chromatography of fatty acid methyl esters. A study of conditions for the quantitative analysis of short- and long-chain fatty acids in lipids. J Chromatogr 270:493–506
Benzonana G, Esposito S (1971) On the positional specifities of Candia cylindracea lipase. Biochim Biophys Acta 231:15–22
Bozoglu F, Swaisgood H, Adams D Jr (1984) Isolation and characterization of an extracellular heat-stable lipase produced by Pseudomonas fluorescens MC50. J Agric Food Chem 32:2–6
Christie WW (1987) The analysis of lipids with special reference to milk fat. In: Hamilton RJ (ed) Recent advances in chemistry and technology of fats and oils. Elsevier Applied Science, Amsterdam, pp 57–78
Davis BJ (1964) Disc gel electrophoresis. II. Method and application to human serum proteins. Ann NY Acad Sci 121:404–427
Fletcher PDI, Rees GD, Robinson BH, Freedman RB (1985) Kinetic properties of alfa-chymotroypsin in water-in-oil microemulsion: studies with a variety of substrates and microemulsion systems. Biochim Biophys Acta 832:204–210
Fukumoto J, Iwai M, Tsujisaka Y (1963) Purification and crystallization of a lipase secreted by Aspergillus niger. J Gen Appl Microbiol 9:353–361
Goldberg M, Thomas D, Legoy MD (1990) The control of lipase-catalyzed transesterification and esterification reaction rates: effects of substrate polarity, water activity and water molecules on enzyme activity. Eur J Biochem 190:603–609
Gilbert E, Cornish A, Jones C (1991) Purification and properties of extracellular lipase from Pseudomonas auruginosa EF2, J Gen Microbiol 137:2223–2229
Hartree EP (1972) Determination of protein: a modification of the Lowry method that gives a linear photometric response. Anal Biochem 48:422–427
Hou CT, Johnstone TM (1992) Screening of lipase activities with cultures from the agricultural research service culture collection. J Am Oil Chem Soc 69:1088–1097
Iwai M, Tsujisaka Y (1974) The purification and properties of three kinds of lipases from Rhizopus delemar. Agric Biol Chem 38:241–1247
Kalo P (1986) Quantitative determination of triacylglycerols separated on capillary columns according to acyl carbon number and level of unsaturation. J Chromatogr 368:145–151
Kalo P, Huotari H, Antila M (1989) Pseudomonas fluorescens lipase-catalyzed interesterification of butter fat. Fat Sci Technol 7:276–281
Kermasha S, Metche M (1986) Characterization of seed lipooxygenase of Phaseolus vulgaris cv. Haricot. J Food Sci 50:88–92
Kermasha S, Kubow S, Safari M, Reid A (1993) Determination of positional distribution of selected fatty acids in butter triacylglycerols using high performance liquid chromatography/laser light scattering detection. J Am Oil Chem Soc 70:169–173
Kouker G, Jaeger K (1987) Specific and sensitive plate assay for bacterial lipases. Appl Environ Microbiol 53:211–213
Lee SY, Rhee JS (1993) Production and partial purification of lipase from Pseudomonas putida 3SK. Enzyme Microb Technol 15:617–623
Lineweaver H, Burk D (1934) The determination of enzyme dissociation constants. J Am Chem Soc 56:658–666
Lu JY, Liska BJ (1969) Lipase from Pseudomonas fragi. II. Properties of the enzyme. Appl Microbiol 18:108–113
Macrae AR (1983) Lipase-catalyzed interesterification of oils and fats. J Am Oil Chem Soc 60:291–294
Martinek K, Levashov AV, Khmelnitsky YL (1982) Colloidal solution of water in organic solvents. a microheterogenous medium for enzymatic reactions. Science 218:889–890
Mattson FH, Volpenhein RA (1961) The use of pancreatic lipase for determining the distribution of fatty acids in partial and complete triglycerides. J Lipid Res 2:58–62
Mencher JR, Alford JA (1967) Purification and characterization of the lipase of Pseudomonas fragi. J Gen Microbiol 48:317–328
Myher JJ, Kuksis A (1979) Stereospecific analysis of triacylglycerols via racemic phosphatidylcholines and phospholipase C. Can J Biochem 57:117–124
Nashif SA, Nelson FE (1953) The lipase of Pseudomonas fragi. III. Enzyme action in cream and butter. J Dairy Sci 36:481–488
Nishio T, Chikano T, Kamimura M, (1987) Purification and some properties of lipase produced by Pseudomonas fragi 22.39 B. Agric Biol Chem 51:181–186
Okumura S, Iwai M, Tsujisaka Y (1976) Positional specificities of four kinds of microbial lipases. Agric Biol Chem 40:655–660
Pharmacia (1992) Handbook, 2nd edn. Pharmacia LKB Biotechnology, Uppsala, Sweden
Safari M, Kermasha S, Pabai F (1993) Interesterification of butter fat by lipase from Mucor miehei in mrganic solvent media. Food Biotechnol 7:265–273
Safari M, Kermasha S, Pabai F (1984) Interesterification of butter fat by lipase from Mucor miehei in microemulsion system. J Food Lipids 1:247–263
Sztajer H, Borkowski J, Sobiech K (1991) Purification and some properties of Pseudomonas fluorescens lipase. Biotechnol Appl Biochem 13:67–71
Vadehra D (1974) Staphylococcal lipases. Lipids 9:158–165
Watanabe N, Ota Y, Minoda Y, Yamada K (1977) Isolation and identification of alkaline lipase producing microorganisms, cultural conditions and some properties of crude enzymes. Agric Biol Chem 41:1353–1358
Yamamoto K, Fujiwara N (1988) Purification and some properties of a castor-oil-hydrolysing lipase from Pseudomonas sp. Agric Biol Chem 52:3015–3021
Zaks A, Kilbanov AM (1984) Enzymatic catalysis in organic media at 100°C. Science 224:1249–1251
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pabai, F., Kermasha, S. & Morin, A. Lipase from Pseudomonas fragi CRDA 323: Partial purification, characterization and interesterification of butter fat. Appl Microbiol Biotechnol 43, 42–51 (1995). https://doi.org/10.1007/BF00170621
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00170621