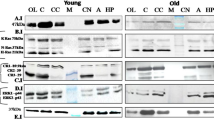
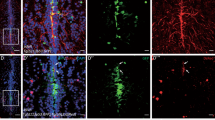
Abstract
Aging, an inevitable complex phenomenon among organisms, exhibit progressive loss of functional anatomy but understanding on molecular mechanisms of aging remains elusive. This report presents analysis of complex network of genes and proteins, their functional cascades and hierarchy, and regulation of regulators like Pax6 (Paired box 6) because symptoms of age-associated changes match with patients having mutation in Pax6. The Pax6 has been observed in the olfactory bulb, amygdala, thalamus, and the cerebellum. It is pro-neurogenic in adult neural progenitors and specifies neuronal subtypes in developing brain and the adult. During aging in brain, the reduction in Pax6-positive cells indicates loss of neurons and affects on adult neuronal stem cells. The alteration in co-localization of Pax6, p53 and SPARC may lead to the loss of plasticity, and p53 mediated cell death pathway during aging. Since the TGF-β, interacts with Pax6 and also gets influenced by Catalase, the Pax6 appears influencing immunological surveillance of brain. The alteration in levels of S100β and genes of oxidative stress management by Pax6 also indicates involvement of Pax6-TGF-β-Catalase axis in aging. The microRNA based regulation is also reflected because the miR335 suppresses the Pax6 expression and acts as anti-oncogenic target in glioma. The implication of Pax6 could also be explored towards possibilities of modifying the rate of aging and reversal of aging clock because the Pax6 interacts with p53 and gets altered during aging.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Arora A, McKay GJ, Simpson DA (2007) Prediction and verification of miRNA expression in human and rat retinas. Invest Ophthalmol Vis Sci 48:3962–3967
Barone E, Cenini G, DiDomenico F, Noel T, Wang C, perluigi M, StClair DK, Butterfield DA (2015) Basal brain oxidative and nitrosative stress levels are finely regulated by interplay between superoxide dismutase2 and p 53. Neurosci Res 93:1728–1739
Bastien-Dionne PO, David LS, Parent A, Saghatelyan A (2010) Role of sensory activity on chemospecific populations of interneurons in the adult olfactory bulb. J Comp Neurol. 518:1847–1861
Bhinge A, Poschmann J, Namboori SC, Tian X, JiaHuiLoh S, Traczyk A, Prabhakar S, Stanton LW (2014) MiR-135b is a direct PAX6 target and specifies human neuroectoderm by inhibiting TGF-β/BMP signalling. EMBO J 33:1271–1283
Bishop KM, Rubenstein JL, O’Leary DD (2002) Distinct actions of Emx1, Emx2 and Pax6 in regulating the specification of areas in the developing neocortex. J Neurosci 22:7627–7638
Bohlen V (2010) Dendritic spine abnormalities in mental retardation. Cell Tissue Res 342:317–323
Brian TW, Poala ST (2003) Theories of aging. J Appl Physiol 95:1706–1716
Brill MS, Snapyan M, Wohlfrom H, Ninkovic J, Jawerka M, Mastick GS, Ashery-Padan R, Saghatelyan A, Berninger B, Gotz M (2008) A dlx2- and pax6-dependent transcriptional code for periglomerular neuron specification in the adult olfactory bulb. J Neurosci 28:6439–6452
Caraci F, Gulisano W, Guida CA, Inpellizzeri AA, Drago F, Puzzo D, Palmeri A (2015) A key role for TGF-β1 in hippocampal synaptic plasticity and memory. Sci Rep 5:1–10
Carrasco GE, Arrizabalago O, Serrano M, Lovell BR, Matheu A (2015) Increased gene dosage of Ink4/Arf and p53 delays age-associated central nervous system functional decline. Aging Cell 14:710–714
Chamberlain CG, Mansfield KJ, Cerra A (2009) Glutathione and catalase suppress TGFβ-induced cataract-related changes in cultured rat lenses and lens epithelial explants. Mol Vis 15:895–905
Cheng Q, Cao H, Chen Z, Ma Z, Wan X, Peng R, Jiang B (2014) PAX6, a novel target of miR-335, inhibits cell proliferation and invasion in glioma cells. Mol Med Rep 10:399–404
Coppe JP, Desprez PY, Krtolica A, Campisi J (2010) The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol 5:99–118
De Bleser PJ, Xu G, Rombouts K, Rogiers V, Geerts A (1999) Glutathione levels discriminate between oxidative stress and transforming growth factor-beta signaling in activated rat hepatic stellate cells. J Biol Chem 274:33881–33887
De Chevigny A, Coré N, Follert P, Gaudin M, Barbry P, Béclin C, Cremer H (2012) miR-7a regulation of Pax6 controls spatial origin of forebrain dopaminergic neurons. Nat Neurosci 15:1120–1126
De Haan G, Van Zant G (1999) Dynamic changes in mouse hematopoietic stem cell numbers during aging. Blood 93:3294–3301
Dellovade TL, Pfaff DW, Schwanzel-Fukuda M (1998) Olfactory bulb development is altered in small-eye (Sey) mice. J Comp Neuro 402:402–418
Dodds C (2006) Physiology of ageing. Anaesthea Intense Care Med 7:456–458
Engelkamp D, Rashbass P, Seawrigth A, Van Heyningen V (1999) Role of Pax6 in development of the cerebellar system. Development 126:3585–3596
Franceschi C, Capri M, Monti D, Giunta S, Olivieri F, Sevini F, Panourgia MP, Incidia L, Celani L, Scurti M, Cevenini E, Castellani GC, Salvioli S (2007) Inflammaging and anti-inflammaging: a systemic perspective on aging and longevity emerged from studies in humans. Mech Ageing Develop 128:92–105
Gage FH (2002) Neurogenesis in the adult brain. J Neurosci 22:612–613
Glaser T, Jepeal L, Edwards JG, Young SR, Favor J, Maas RL (1994) Pax6 gene dosage effect in a family with congenital cataracts, aniridia, anophthalmia and central-nervous-system defects. Nat Genet 7:463–471
Gotz M, Stoykova A, Gruss P (1998) Pax6 controls radial glia differentiation in the cerebral cortex. Neuron 5:1031–1044
Haba H, Nomura T, Suto F, Osumi N (2009) Subtype-specific reduction of olfactory bulb interneurons in Pax6 heterozygous mutant mice. Neurosci Res 65:115–121
Hack MA, Saghatelyan A, de Chevigny A, Pfeifer A, Ashery-Padan R, Lledo PM, Gotz M (2005) Neuronal fate determinants of adult olfactory bulb neurogenesis. Nat Neurosci 8:865–872
Harman D (2003) The free radical theory of aging. Antioxid Redox Signal 5:557–561
Harrison D, Astle C, Stone M (1989) Numbers and functions of transplantable primitive immunohematopoietic stem cells. Effects of age. J Immunol 142:3833–3840
Heins N, Malatesta P, Cecconi F, Nakafuku M, Tucker KL, Hack MA, Chapouton P, Barde YA, Gotz M (2002) Glial cells generate neurons: the role of the transcription factor Pax6. Nat Neurosci 4:308–315
Kanungo MS (1975) A model for aging. J Theor Biol 53:253–261
Kawano H, Fukuda T, Kubo K, Horie M, Uyemura K, Takeuchi K, Osumi N, Eto K, Kawamura K (1999) Pax-6 is required for thalamocortical pathway formation in fetal rats. J Comp Neuro 408:147–160
Kohwi M, Osumi N, Rubenstein JLR, Alvarez-Buylla A (2005) Pax6 is required for making specific subpopulations of granule and periglomerular neurons in the olfactory bulb. J Neurosci 25:6997–7003
Kredo-Russo S, Mandelbaum AD, Ness A et al (2012) Pancreas-enriched miRNA refines endocrine cell differentiation. Development 139:3021–3031
Kroll TT, O’Leary DD (2005) Ventralized dorsal telencephalic progenitors in Pax6 mutant mice generate GABA interneurons of a lateral ganglionic eminence fate. Proc Natl Acad Sci 102:7374–7379
Louveau A, Smirnov I, Keyes TJ, Eccles JD, Rouhani SJ, Peske D, Derecki NC, Castle D, Mandell JW, Lee KS, Harris TH, Kipnis J (2015) Structural and functional features of central nervous system lymphatic vessels. Nautre. doi:10.1038/nature14432
Lu T, Pan Y, Kao SY, Li C, Kohane I, Chan J, Yankner BA (2004) Gene regulation and DNA damage in the ageing human brain. Nature 429:883–889
Maekawa M, Takashima N, Arai Y, Nomura T, Inokuchi K, Yuasa S, Osumi N (2005) Pax6 is required for production and maintenance of progenitor cells in postnatal hippocampal neurogenesis. Genes Cells 10:1001–1014
Mastick GS, Davis NM, Andrew GL, Easter SS (1997) Pax6 functions in boundary formation and axon guidance in the embryonic mouse forebrain. Development 124:1985–1997
Ming GL, Song H (2011) Adult neurogenesis in the mammalian brain: significant answers and significant questions. Neuron 70:687–702
Mishra R, Gorlov IP, Chao LY, Singh S, Saunders GF (2002) PAX6, paired domain influences sequence recognition by the homeodomain. J Biol Chem 277:49488–49494
Mishra S, Maurya SK, Srivastava K, Shukla S, Mishra R (2015) Pax6 influence expression patterns of genes involved in neurodegeneration. Annals of Neuroscience 22:26–231
Nacher J, Varea E, Blasco-Ibanez JM, Castillo-Gomez E, Crespo C, Martinez-Guijarro FJ, Mcewen BS (2005) Expression of the transcription factor Pax6 in the adult rat dentate gyrus. J Neurosci Res 81:753–761
Nikoletopoulou V, Plachta N, Allen ND, Pinto L, Gotz M, Barde YA (2007) Neurotrophin receptor-mediated death of misspecified neurons generated from embryonic stem cells lacking Pax6. Cell Stem Cell 1:529–540
Nimmerjahn A, Kirchhoff F, Helmchen F (2005) Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 308:1314–1318
Nomura T, Haba H, Osumi N (2007) Role of a transcription factor Pax6 in the developing vertebrate olfactory system. Dev Growth Differ 49:683–686
Pang WW, Price AE, Sahoo D, Beerman I, Maloney JW, Rossi JD, Schrier LS, Weissman LI (2011) Human bone marrow hematopoietic stem cells are increased in frequency and myeloid-biased with age. Proc Natl Acad Sci 108:20012–20017
Parrish-Aungst S, Shipley MT, Erdelyi F, Szabo G, Puche AC (2007) Quantitative analysis of neuronal diversity in the mouse olfactory bulb. J Comp Neurol 501:825–836
Rodriguez E, Maoz BB, Dorshkind K (2013) Causes, consequences, and reversal of immune system aging. J Clin Invest 123:958–965
Shibata M, Nakao H, Kiyonari H, Abe T, Aizawa S (2011) MicroRNA-9 regulates neurogenesis in mouse telencephalon by targeting multiple transcription factors. J Neurosci 31:3407–3422
Shubham K, Mishra R (2012) Pax6 interacts with SPARC and TGF-β in murine eyes. Mol Vis 18:951–956
Sisodiya SM, Free SL, Williamson KA, Mitchell TN, Willis C, Stevens JM, Kendall BE, Shorvon SD, Hanson IM, Moore AT, Van Heyningen V (2001) PAX6 haploinsufficiency causes cerebral malformation and olfactory dysfunction in humans. Nat Genet 28:214–216
Stoykova A, Gruss P (1994) Roles of Pax genes in developing and adult brain as suggested by expression patterns. J Neurosci 14:1395–1412
Stoykova A, Treichel D, Hallonet M, Gruss P (2000) Pax6 modulates the dorsoventral patterning of the mammalian telencephalon. J Neurosci 20:8042–8050
Streit WJ, Miller KR, Lopes KO, Njie E (2008) Microglial degeneration in the aging brain–bad news for neurons? Front Biosci 13:3423–3438
Tesseur I, Wyss-Coray T (2006) A role for TGF-beta signaling in neurodegeneration: evidence from genetically engineered models. Curr Alzheimer Res 3:505–513
Thomas A, Rando L, Chang HY (2012) Aging, rejuvenation, and epigenetic reprogramming: resetting the aging clock. Cell 148:46–57
Tichauer JE, Flores B, Soler B, Eugenín-von Bernhardi L, Ramírez G, Von Bernhardi R (2014) Age-dependent changes on TGFbeta1 Smad3 pathway modify the pattern of microglial cell activation. Brain Behav Immun 37:187–196
Tole S, Remedios R, Saha B, Stoykova A (2005) Selective requirement of pax6, but not Emx2, in the specification and development of several nuclei of the amygdaloid complex. J Neurosci 25:2753–2760
Tripathi R, Mishra R (2010) Interaction of Pax6 with SPARC and p53 in brain of mice indicates Smad3 dependent auto-regulation. J Mol Neurosci 41:397–403
Tripathi R (2012) Studies on expression and interaction of Pax6 with p 53 and SPARC in brain of aging mice, Ph.D. thesis, Banaras Hindu University, Varanasi
Tripathi R, Mishra R (2012) Aging-associated modulation in the expression of Pax6 in mouse brain. Cell Mol Neurobiol 32:209–218
Tuoc TC, Radyushkin K, Tonchev AB, Piñon MC, Ashery-Padan R, Molnar Z, Davidoff MS, Stoykova A (2009) Selective cortical layering abnormalities and behavioral deficits in cortex-specific Pax6 knock-out mice. J Neurosci 26:8335–8349
Weinert BT, Timiras PS (2003) Theories of aging. J Appl Physiol 95:1706–1716
Yamasaki T, Kawaji K, Ono K, Bito H, Hirano T, Osumi N, Kengaku M (2001) Pax6 regulates granule cell polarization during parallel fiber formation in the developing cerebellum. Development 128:3133–3144
Yasuda T, Kajimoto Y, Fujitani Y, Watada H, Yamamoto S, Watarai T, Umayahara Y, Matsuhisa M, Gorogawa S, Kuwayama Y, Tano Y, Yamasaki Y, Hori M (2002) PAX6 mutation as a genetic factor common to aniridia and glucose intolerance. Diabetes 51:224–230
Yousef H, Condoy MJ, Morgenthalar A, Schlesinger C, Buqaj L, Paliwal P, Greer C, Conboy IM, Schaffer D (2015) Systemic attenuation of TGF-beta pathway by single drug simultaneously rejuvenates hippocampal neurogenesis and myogenesis in the same old mammal. Oncotarget 6:11959–11978
Zechel S, Werner S, Unsicker K, Halbach O (2010) Expression and functions of fibroblast growth factor 2 (FGF-2) in hippocampal formation. Neuroscientist 4:357–373
Acknowledgments
Authors gratefully acknowledge financial support from the ICMR (54/2/CFP/GER/2011-NCD-II).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media Singapore
About this chapter
Cite this chapter
Maurya, S.K., Tripathi, R., Mishra, S., Mishra, R. (2017). Expression and Regulation of Pax6 in Brain of Aging Mice. In: Rath, P., Sharma, R., Prasad, S. (eds) Topics in Biomedical Gerontology. Springer, Singapore. https://doi.org/10.1007/978-981-10-2155-8_15
Download citation
DOI: https://doi.org/10.1007/978-981-10-2155-8_15
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-2154-1
Online ISBN: 978-981-10-2155-8
eBook Packages: MedicineMedicine (R0)