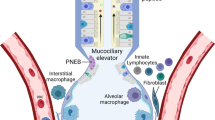
Abstract
Infection of the respiratory system is a frequent cause of morbidity and mortality world-wide [1]. Respiratory tract infection, including croup, tracheobronchitis, bronchiolitis and pneumonia, are significant clinical problems. The increasing number of multidrug-resistant microbes has made the treatment of these infections much more difficult [1]. To further improve therapies for respiratory infection, we need to learn more about the host defence in the lung.
Chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Standiford TJ, Huffnagle GB (1997) Cytokines in host defense against pneumonia. J Invest Med 45: 335–345
Baggiolini M, Dewald B, Moser B (1997) Human chemokines: an update. Annu Rev Immunol 15: 675–705
Baggiolini M (1998) Chemokines and leukocyte traffic. Nature 392: 565–568
Simon RH, Paine III R (1995) Participation of pulmonary alveolar epithelial cells in lung inflammation. J Lab Clin Med 126: 108–118
Standiford TJ, Strieter RM, Greenberger MJ, Kunkel SL (1996) Expression and regulation of chemokines in acute bacterial pneumonia. Biol Signals 5: 203–208
Baggiolini M, Dewald B, Moser B (1994) Interleukin-8 and related chemotactic cytokines-CXC and CC chemokines. Adv Immunol 55: 97–179
Wolpe SD, Cerami A (1989) Macrophage inflammatory proteins 1 and 2: members of a novel superfamily of cytokines. FASEB J 3: 2565–2573
McRitchie DI, Isowa N, Edelson JD et al (2000) Production of tumour necrosis factor alpha by primary cultured rat alveolar epithelial cells. Cytokine 12: 644–654
Standiford T, Kunkel SL, Basha MA et al (1990) Interleukin-8 gene expression by a pulmonary epithelial cell line. A model for cytokine networks in the lung. J Clin Invest 86: 1945–1953
Xavier AM, Isowa N, Cai L, et al (1999) Tumor necrosis factor-alpha mediates lipopolysaccharide-induced macrophage inflammatory protein-2 release from alveolar epithelial cells. Autoregulation in host defense. Am J Respir Cell Mol Biol 21: 510–520
Standiford TJ, Kunkel SL, Phan SH et al (1991) Alveolar macrophage-derived cytokines induce monocyte chemoattractant protein-1 expression from human pulmonary type II-like epithelial cells. J Biol Chem 266: 9912–9918
Paine III R, Rolfe MW, Standiford TJ et al (1993) MCP-1 expression by rat type II alveolar epithelial cells in primary culture. J Immunol 150: 4561–4570
VanOtteren GM, Strieter RM, Kunkel SL et al (1995) Compartmentalized expression of RANTES in a murine model of endotoxemia. J Immunol 154: 1900–1908
Liu M, Slutsky AS (1997) Anti-inflammatory therapies: application of molecular biology techniques in intensive care medicine. Inten Care Med 23: 718–731
Fisher CJ, Jr., Dhainaut JF, Opal SM et al (1994) Recombinant human interleukin 1 receptor antagonist in the treatment of patients with sepsis syndrome. Results from a randomized, double-blind, placebo-controlled trial. Phase III rhIL-lra Sepsis Syndrome Study Group. JAMA 271: 1836–1843
Fisher CJ, Jr., Slotman GJ, Opal SM et al (1994) Initial evaluation of human recombinant interleukin-1 receptor antagonist in the treatment of sepsis syndrome: a randomized, open-label, placebo-controlled multicenter trial. The IL- IRA Sepsis Syndrome Study Group. Crit Care Med 22: 12–21
Standiford TJ, Kunkel SL, Greenberger MJ et al (1996) Expression and regulation of chemokines in bacterial pneumonia. J Leukoc Biol 59: 24–28
Baggiolini M, Moser B (1997) Blocking chemokine receptors. J Exp Med 186: 1189–1191
Bazzoni F, Beutler B (1996) The tumor necrosis factor ligand and receptor families. New Eng J Med 334: 1717–1725
Peppel K, Crawford D, Beutler B (1991) A tumor necrosis factor ( TNF) receptor-IgG heavy chain chimeric protein as a bivalent antagonist of TNF activity. J Exp Med 174: 1483–1489
Larner AC, Finbloom DS (1995) Protein tyrosine phosphorylation as a mechanism which regulates cytokine activation of early response genes. Biochim Biophys Acta 1266: 278–287
Lee JC, Laydon JT, McDonnell PC et al (1994) A protein kinase involved in the regulation of inflammatory cytokine biosynthesis. Nature 372: 739–745
Beyaert R, Cuenda A, Vanden Berghe W et al (1996) The p38/RK mitogen-activated protein kinase pathway regulates interleukin-6 synthesis response to tumor necrosis factor. EMBO J 15: 1914–1923
Collart MA, Baeuerle P, Vassalli P (1990) Regulation of tumor necrosis factor alpha transcription in macrophages: involvement of four kappa B-like motifs and of constitutive and inducible forms of NF-kappa B. Mol Cell Biol 10: 1498–506
Han J, Brown T, Beutler B (1990) Endotoxin-responsive sequences control cachectin/TNF biosynthesis at the translational level. J Exp Med 171: 465–475
Shaw G, Kamen R (1986) A conserved AU sequence from the 3’ untranslated region of GM-CSF mRNA mediates selective mRNA degradation. Cell 46: 659–667
Baggiolini M, Walz A, Kunkel SL (1989) Neutrophil-activating peptide-1/interleukin 8, a novel cytokine that activates neutrophils. J Clin Invest 84: 1045–1049
Carre PC, Mortenson RL, King TE, Jr. et al (1991) Increased expression of the interleukin-8 gene by alveolar macrophages in idiopathic pulmonary fibrosis. A potential mechanism for the recruitment and activation of neutrophils in lung fibrosis. J Clin Invest 88: 1802–1810
Miller EJ, Cohen AB, Nagao S et al (1992) Elevated levels of NAP-1/interleukin-8 are present in the airspaces of patients with the adult respiratory distress syndrome and are associated with increased mortality. Am Rev Respir Dis 146: 427–432
Broaddus VC, Hebert CA, Vitangcol RV et al (1992) Interleukin-8 is a major neutrophil chemotactic factor in pleural liquid of patients with empyema. Am Rev Respir Dis 146: 825–830
Sekido N, Mukaida N, Harada A et al (1993) Prevention of lung reperfusion injury in rabbits by a monoclonal antibody against interleukin-8. Nature 365: 654–657
Pechkovsky DV, Zissel G, Ziegenhagen MW et al (2000) Effect of proinflammatory cytokines on interleukin-8 mRNA expression and protein production by isolated human alveolar epithelial cells type II in primary culture. Eur Cytokine Netw 11: 618–625
Wolpe SD, Davatelis G, Sherry B et al (1988) Macrophages secrete a novel heparin-binding protein with inflammatory and neutrophil chemokinetic properties. J Exp Med 167: 570–581
Shanley TP, Schmal H, Friedl HP et al (1995) Role of macrophage inflammatory protein-1 alpha (MIP-1 alpha) in acute lung injury in rats. J Immunol 154: 4793–4802
Feng L, Xia Y, Yoshimura T, Wilson CB (1995) Modulation of neutrophil influx in glomerulo-nephritis in the rat with anti-macrophage inflammatory protein-2 (MIP-2) antibody. J Clin Invest 95: 1009–1017
Driscoll KE, Hassenbein DG, Howard BW et al (1995) Cloning, expression, and functional characterization of rat MIP-2: a neutrophil chemoattractant and epithelial cell mitogen. J Leukoc Biol 58: 359–364
Gupta S, Feng L, Yoshimura T et al (1996) Intra-alveolar macrophage-inflammatory peptide 2 induces rapid neutrophil localization in the lung. Am J Respir Cell Mol Biol 15: 656–663
Zhang H, Kim YK, Govindarajan A et al (1999) Effect of adrenoreceptors on endotoxin-induced cytokines and lipid peroxidation in lung explants. Am J Respir Crit Care Med 160: 1703–1710
Foley R, Driscoll K, Wan Y et al(1996) Adenoviral gene transfer of macrophage inflammatory protein-2 in rat lung. Am J Pathol 149: 1395–1403
Greenberger MJ, Strieter RM, Kunkel SL et al (1996) Neutralization of macrophage inflammatory protein-2 attenuates neutrophil recruitment and bacterial clearance in murine Klebsiella pneumonia. J Infect Dis 173: 159–165
Lakshminarayanan V, Drab-Weiss EA, Roebuck KA (1998) H2O2 and tumor necrosis factor-alpha induce differential binding of the redox-responsive transcription factors AP-1 and NF-kappaB to the interleukin-8 promoter in endothelial and epithelial cells. J Biol Chem 273: 32670–32678
Brasier AR, Jamaluddin M, Casola A et al (1998) A promoter recruitment mechanism for tumor necrosis factor-alpha-induced interleukin-8 transcription in type II pulmonary epithelial cells. Dependence on nuclear abundance of Rel A, NF-kappaB 1, and c-Rel transcription factors. J Biol Chem 273: 3551–3561
Fiedler MA, Wernke-Dollries K, Stark JM (1998) Inhibition of TNF-alpha-induced NF-kappaB activation and IL-8 release in A549 cells with the proteasome inhibitor MG-132. Am J Respir Cell Mol Biol 19: 259–268
Standiford TJ, Kunkel SL, Basha MA et al (1990) Interleukin-8 gene expression by a pulmonary epithelial cell line. A model for cytokine networks in the lung. J Clin Invest 86: 1945–1953
Bittleman DB, Casale TB (1995) Interleukin-8 mediates interleukin-1 alpha-induced neutrophil transcellular migration. Am J Respir Cell Mol Biol 13: 323–329
Patel JA, Jiang Z, Nakajima N, Kunimoto M (1998) Autocrine regulation of interleukin-8 by interleukin-lalpha in respiratory syncytial virus-infected pulmonary epithelial cells in vitro. Immunology 95: 501–506
Schwarz YA, Amin RS, Stark JM et al (1999) Interleukin-1 receptor antagonist inhibits interleukin-8 expression in A549 respiratory epithelial cells infected in vitro with a replication-deficient recombinant adenovirus vector. Am J Respir Cell Mol Biol 21:388–394
Mori N, Oishi K, Sar B et al (1999) Essential role of transcription factor nuclear factor-kappaB in regulation of interleukin-8 gene expression by nitrite reductase from Pseudomonas aeruginosa in respiratory epithelial cells. Infect Immun 67: 3872–3878
Simeonova PP, Luster MI (1996) Asbestos induction of nuclear transcription factors and interleukin-8 gene regulation. Am J Respir Cell Mol Biol 15: 787–795
Mastronarde JG, Monick MM, Mukaida N et al (1998) Activator protein-1 is the preferred transcription factor for cooperative interaction with nuclear factor-kappaB in respiratory syncytial virus-induced interleukin-8 gene expression in airway epithelium. J Infect Dis 177: 1275–1281
Kito H, Chen EL, Wang X et al (2000) Role of mitogen-activated protein kinases in pulmonary endothelial cells exposed to cyclic strain. J Appl Physiol 89: 2391–2400
Quinn D, Tager A, Joseph PM et al (1999) Stretch-induced mitogen-activated protein kinase activation and interleukin-8 production in type II alveolar cells. Chest 116: 89S - 90S
Roberts ML, Cowsert LM (1998) Interleukin-1. beta and reactive oxygen species mediate activation of c-Jun NH2-terminal kinases, in human epithelial cells, by two independent pathways. Biochem Biophys Res Commun 251: 166–172
Monick M, Staber J, Thomas K, Hunninghake G (2001) Respiratory syncytial virus infection results in activation of multiple protein kinase C isoforms leading to activation of mitogen-activated protein kinase. J Immunol 166: 2681–2687
Chen W, Monick MM, Carter AB, Hunninghake GW (2000) Activation of ERK2 by respiratory syncytial virus in A549 cells is linked to the production of interleukin 8. Exp Lung Res 26: 13–26
Wondering RS, Ghaffar A, Mayer EP (1996) Lipopolysaccharide-induced suppression of phagocytosis: Effects on the phagocytic machinery. Immunopharmacol Immunotoxicol 18: 267–289
Shinji H, Kaiho S, Nakano T, Yoshida T (1991) Reorganization of microfilaments in macrophages after LPS stimulation. Exp Cell Res 193: 127–133
Doherty DE, Downey GP, Schwab III B, Elson E, Worthen GS (1994) Lipopolysaccharide-induced monocyte retention in the lung: role of monocyte stiffness, actin assembly, and CD18-dependent adherence. J Immunol 153: 241–255
Howard TH, Wang D, Berkow RL (1990) Lipopolysaccharide modulates chemotactic peptide-induced actin polymerization in neutrophils. J Leukoc Biol 47: 13–24
Isowa N, Xavier AM, Dziak E et al (1999) LPS-induced depolymerization of cytoskeleton and its role in TNF-alpha production by rat pneumocytes. Am J Physiol 277: L606 - L615
Isowa N, Liu M (2001) Role of LPS-induced microfilament depolymerization in MIP-2 production from rat pneumocytes. Am J Physiol Lung Cell Mol Physiol 280: L762–770
Shinji H, Akagawa KS, Yoshida T (1993) Cytochalasin D inhibits lipopolysaccharide-induced tumor necrosis factor production in macrophages. J Leukoc Biol 54: 336–342
Bubb MR, Senderowicz AM, Sausville EA et al (1994) Jasplakinolide, a cytotoxic natural product, induces actin polymerization and competitively inhibits the binding of phalloidin to F-actin. J Biol Chem 269: 14869–14871
Mourgeon E, Isowa N, Keshavjee S et al (2000) Mechanical stretch stimulates macrophage inflammatory protein-2 secretion from fetal rat lung cells. Am J Physiol Lung Cell Mol Physiol 279: L699–706
Bloom GS, Goldstein LS (1998) Cruising along microtubule highways: how membranes move through the secretory pathway. J Cell Biol 140: 1277–1280
Valentijn KM, Gumkowski FD, Jamieson JD (1999) The subapical actin cytoskeleton regulates secretion and membrane retrieval in pancreatic acinar cells. J Cell Sci 112: 81–96
Cole NB, Lippincott-Schwartz J (1995) Organization of organdies and membrane traffic by microtubules. Curr Opin Cell Biol 7: 55–64
Saraste J, Kuismanen E (1992) Pathways of protein sorting and membrane traffic between the rough endoplasmic reticulum and the Golgi complex. Semin Cell Biol 3: 343–55.
Lippincott-Schwartz J (1998) Cytoskeletal proteins and Golgi dynamics. Curr Opin Cell Biol 10: 52–59
Muesch A, Hartmann E, Rohde K et al (1990) A novel pathway for secretory proteins? Trends Biochem Sci 15: 86–88
Isowa N, Keshavjee SH, Liu M (2000) Role of microtubules in LPS-induced macrophage inflammatory protein-2 production from rat pneumocytes. Am J Physiol:In press.
Allen JN, Moore SA, Liao Z, Wewers MD (1997) Changes in mononuclear phagocyte microtubules after endotoxin stimulation. I. Changes in microtubule stability. Am J Respir Cell Mol Biol 16: 119–126
Cole NB, Ellenberg J, Song Jet al (1998) Retrograde transport of Golgi-localized proteins to the ER. J Cell Biol 140: 1–15
Lippincott-Schwartz J, Donaldson JG, Schweizer A et al (1990) Microtubule-dependent retrograde transport of proteins into the ER in the presence of brefeldin A suggests an ER recycling pathway. Cell 60: 821–836
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2002 Springer-Verlag Italia, Milano
About this paper
Cite this paper
Liu, M. (2002). Alveolar Epithelium in Host Defence: Cytokine Production. In: Baue, A.E., Berlot, G., Gullo, A., Vincent, JL. (eds) Sepsis and Organ Dysfunction. Springer, Milano. https://doi.org/10.1007/978-88-470-2213-3_2
Download citation
DOI: https://doi.org/10.1007/978-88-470-2213-3_2
Publisher Name: Springer, Milano
Print ISBN: 978-88-470-0178-7
Online ISBN: 978-88-470-2213-3
eBook Packages: Springer Book Archive