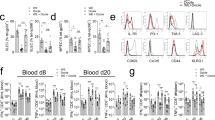
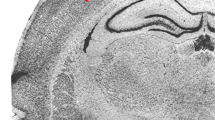
Abstract
The strength of the lymphocytic choriomeningitis virus (LCMV) model rests on the following five foundations. First, the virus in vivo in its natural host, the mouse, or in vitro in cultured cells is non-cytolytic. This quality allows clear separation of effects caused by the virus from those caused by the host immune system. Consequently, the host cell control of viral infection as opposed to how virus interacts with cells to distort their functions without killing them can be decoded. Second, reactions to LCMV infection can encompass a widely diverse range of immune responses (Fig. 1). Usually when immunocompetent adult mice are injected with LCMV, they generate a marked immune response to eliminate the infectious agent. Although their innate responses include the production of interferon (IFN), macrophages and natural killer (NK) cells (Muller et al. 1994; see reviews Buchmeier et al. 1980; Borrow and Oldstone 1997; see chapter by Biron et al., this volume), it is the adoptive immune response — primarily the virus-specific CD8+ CTL response — that is responsible for virus clearance.
Chapter PDF
Similar content being viewed by others
Keywords
- Major Histocompatibility Complex
- Persistent Infection
- Major Histocompatibility Complex Molecule
- Growth Hormone Cell
- Growth Hormone mRNA
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Aebischer T, Moskophidis D, Rohrer UH, Zinkernagel RH, Hengartner R (1991) In vitro selection of lymphocytic choriomeningitis virus escape mutants by cytotoxic T lymphocytes. Proc Natl Acad Sci 88: 11047–11051
Ahmed R, Byrne JA, Oldstone MBA (1984a) Virus specificity of cytotoxic T lymphocytes generated during acute lymphocytic choriomeningitis virus infection: role of the H-2 region in determining cross-reactivity for different lymphocytic choriomeningitis virus strains. J Virol 51:34–41
Ahmed R, Jamieson BD, Porter D (1987) Immune therapy of a persistent and disseminated viral infection. J Virol 61:3920–3929
Ahmed R, Oldstone MBA (1988) Organ-specific selection of viral variants during chronic infection. J Exp Med 167:1719–1724
Ahmed R, Hahn CS, Somasundaram T, Villarete L, Matloubian M, Strauss JH (1991) Molecular basis of organ-specific selection of viral variants during chronic infection. J Virol 65:4242–4247
Ahmed R, Salmi A, Butler LD, Chiller JM, Oldstone MBA (1984b) Selection of genetic variants of lymphocytic choriomeningitis virus in spleens of persistently infected mice: role in suppression of cytotoxic T lymphocyte response and viral persistence. J Exp Med 160:521–540
Ahmed R, Simon R, Matloubian M, Kohleker S, Southern P, Freedman D (1988) Genetic analysis of in vivo-selected viral variants causing chronic infection: importance of mutation in the L RNA segment of lymphocytic choriomeningitis virus. J Virol 62:3301–3308
Althage A, Odermatt B, Moskophidis D, et al. (1992) Immunosuppression by lymphocytic choriomeningitis virus infection: competent effector T and B cells but impaired antigen presentation. Eur J Immunol 22:1803–1812
Anderson J, Byrne JA, Schreiber R, Patterson S, Oldstone MB (1985) Biology of cloned cytotoxic T lymphocytes specific for lymphocytic choriomeningitis virus: clearance of virus and in vitro properties. J Virol 53:552–560
Baenziger J, Hengartner H, Zinkernagel RM, Cole GA (1986) Induction or prevention of immuno-pathological disease by cloned cytotoxic T cell lines specific for lymphocytic choriomeningitis virus. Eur J Immunol 16:1237–1242
Battegay M, Moskophidis D, Rahemtulla A, Hengartner H, Mak TW, Zinkernagel RM (1994) Enhanced establishment of a virus carrier state in adult CD4+ T-cell-deficient mice. J Virol 68:4700
Beijerinck MW (1899) Bemerkung zu dem Aufsatz von Herrn Iwanowsky über die Mosaikkrankheit der Tabakspflanze. Zentralbl. Bakteriol. Parasitenkd. Infektionskr. Hyg Abt I Orig 5:310–311
Berger DP, Homann D, Oldstone MBA (2000) Defining parameters for successful immunocytotherapy of persistent viral infection. Virology 266:257–263
Bertoletti A, Costanzo A, Chisari FV, Levrero M, Artini M, Sette A, Penna A, Giuberti T, Fiaccadori F, Ferrari C (1994a) Cytotoxic T lymphocyte response to a wild-type hepatitis B virus epitope in patients chronically infected by variant viruses carrying substitutions within the epitope. J Exp Med 180:933–943
Bertoletti A, Sette A, Chisari FV, Penna A, Levrero M, De Carli M, Fiaccadori F, Ferrari C (1994b) Natural variants of cytotoxic epitopes are T cell receptor antagonists for anti-viral cytotoxic T cells. Nature 369:407–410
Bjorkman PJ, Saper MA, Samraoui B, Bennett WS, Strominger JL, Wiley DC (1987) Structure of the human class I histocompatibility antigen, HLA-A2. Nature 329:506–511
Blount P, Elder J, Lipkin WI, Southern PJ, Buchmeier MJ, Oldstone MBA (1986) Dissecting the molecular anatomy of the nervous system: Analysis of RNA and protein expression in whole body sections of laboratory animals. Brain Res 382:257–265
Borrow P, Evans CF, Oldstone MBA (1995) Virus-induced immunosuppression: immune system-mediated destruction of virus-infected dendritic cells results in generalized immunosuppression. J Virol 69: 1059–1070
Borrow P, Lewicki H, Wei X, Horwitz MS, Peffer N, Meyers H, Nelson JA, Gairin JE, Hahn BH, Oldstone MBA, Shaw GM (1997) Antiviral pressure exerted by HIV-1-specific cytotoxic T lymphocytes (CTLs) during primary infection demonstrated by rapid selection of CTL escape virus. Nature Medicine 3:205–211
Borrow P, Oldstone MBA (1997) Lymphocytic choriomeningitis virus. In: Nathanson N, et al. (eds) Viral Pathogenesis. Lippincott-Raven Publishers, Philadelphia, pp 593–627
Borrow P, Vedovato V, Buesa-Gomez J, de la Torre J, Oldstone M (1993) The in vivo pathogenicity of different LCMV isolates correlates with binding to putative cellular receptors in vitro and the amino acid sequence of the virion attachment protein GP-1 (abstract). IXth International Congress of Virology, Glasgow, Scotland
Buchmeier MJ, Welsh RM, Dutko FJ, Oldstone MBA (1980) The virology and immunobiology of lymphocytic choriomeningitis virus infection. Adv Immunol 30:275–331
Buesa-Gomez J, Teng MN, Oldstone MBA, de la Torre JC (1996) Variants able to cause growth hormone deficiency syndrome are present within the disease-nil WE strain of lymphocytic choriomeningitis virus. J Virol 70:8988–8992
Bureau JF, Le Goff S, Thomas D, Parlow AF, de la Torre JC, Homann D, Brahic M, Oldstone MBA (2001) Disruption of differentiated functions during viral infection in vivo. V. Mapping of a locus involved in susceptibility of mice to growth hormone deficiency due to persistent lymphocytic choriomeningitis virus infection. Virology (in press)
Butz EA, Bevan MJ (1998) Massive expansion of antigen-specific CD8+ T cells during an acute virus infection. Immunity 8: 167–175
Byrne JA, Ahmed R, Oldstone MBA (1984) Biology of cloned cytotoxic T lymphocytes specific for lymphocytic choriomeningitis virus. I. Generation and recognition of virus strains and H-2b mutants. J Immunol 133:433–439
Cao W, Henry MD, Borrow P, Yamada H, Elder JH, Ravkov EV, Nichol ST, Compans RW, Campbell KP, Oldstone MBA (1998) Identification of alpha-dystroglycan as a receptor for lymphocytic choriomeningitis virus and Lassa fever virus. Science 282:2079–2081
Casali P, Rice GPA, Oldstone MBA (1984) Viruses disrupt functions of human lymphocytes: effects of measles virus and influenza virus in lymphocyte-mediated killing and antibody production. J Exp Med 159:1322–1337
Cole GA, Nathanson N, Prendergast RA (1972) Requirement for Θ-bearing cells in lymphocytic choriomeningitis virus-induced central nervous system disease. Nature 238:335–337
de Campos-Lima PO, Gavioli R, Zhang QJ, Wallace LE, Dolcetti R, Rowe M, Rickinson AB, Masucci MG (1993) HLA-A11 epitope loss isolates of Epstein-Barr virus from a highly A11+ population. Science 260:98–100
de La Torre JC, Mallory M, Brot M, Gold L, Koob G, Oldstone MBA, Masliah E (1996) Viral persistence in neurons alters synaptic plasticity and cognitive functions without destruction of brain cells. Virology 220:508–515
de la Torre JC, Oldstone MBA (1992) Selective disruption of growth hormone transcription machinery by viral infection. Proc Natl Acad Sci USA 89:9939–9943
de la Torre JC, Rail G, Oldstone C, Sanna P, Borrow P, Oldstone MBA (1993) Replication of lymphocytic choriomeningitis virus is restricted in terminally differentiated neurons. J Virol 67:7350–7359
Dockter J, Evans CF, Tishon A, Oldstone MBA (1996) Competitive selection in vivo by a cell for one variant over another: implications for RNA virus quasispecies in vivo. J Virol 70: 1799–1803
Doherty PC, Allan JE (1986) Role of the major histocompatibility complex in targeting effector T cells into a site of virus infection. Eur J Immunol 16:1237–1242
Evans CF, Borrow P, de la Torre JC, Oldstone MBA (1994) Virus-induced immunosuppression: kinetic analysis of the selection of a mutation associated with viral persistence. J Virol 68:7367–7373
Falk K, Rotzschke O, Stevanovic S, Jung G, Rammensee HG (1991) Allele-specific motifs revealed by sequencing of self-peptides eluted from MHC molecules. Nature 351:290–296
Gairin JE, Joly E, Oldstone MB (1991) Persistent infection with lymphocytic choriomeningitis virus enhances expression of MHC class I GP on cultured mouse brain endothelial cells. J Immunol 146:3953–3957
Gairin JE, Mazarguil H, Hudrisier D, Oldstone MBA (1995) Optimal lymphocytic choriomeningitis virus sequences restricted by H-2Db major histocompatibility complex class I molecules and presented to cytotoxic T lymphocytes. J Virol 69:2297–2305
Garcia KC, Degano M, Stanfield RL, Brunmark A, Jackson MR, Peterson PA, Teyton L, Wilson IA (1996) An αβ T cell receptor structure at 2.5Å and its orientation in the TCR-MHC complex. Science 274:209–219
Gilden DH, Cole GA, Monjan AA, Nathanson N (1972a) Immunopathogenesis of acute central nervous system disease produced by lymphocytic choriomeningitis virus. I. Cyclophosphamide-mediated induction of virus-carrier state in adult mice. J Exp Med 135:860–873
Gilden DH, Cole GA, Nathanson N (1972b) Immunopathogenesis of acute central nervous system disease produced by lymphocytic choriomeningitis virus. II. Adoptive immunization of virus carriers. J Exp Med 135:874–889
Gold LH, Brot MD, Polis I, Schroeder R, Tishon A, de la Torre JC, Oldstone MBA, Koob GF (1994) Behavioral effects of persistent lymphocytic choriomeningitis virus infection in mice. Behav Neural Biol 62:100–109
Goulder PJR, Phillips RE, Colbert RA, McAdam S, Ogg G, Nowak MA, Giangrande P, Luzzi G, Morgan B, Edwards A, McMichael AJ, Rowland-Jones S (1997) Late escape from an immuno-dominant cytotoxic T-lymphocyte response associated with progression to AIDS. Nature Medicine 3:312–317
Guidotti LG, Borrow P, Brown A, McClary H, Koch R, Chisari FV (1999) Noncytopathic clearance of lymphocytic choriomeningitis virus from the hepatocyte. J Exp Med 189:1555–1564
Henry MD, Campbell KP, (1999) Dystroglycan inside and out. Curr Opin Cell Biol 11:602–607
Holtzer H, Biehl J, Yeoh G, Meganathan R, Kaji A (1975) Effect of oncogenic virus on muscle differentiation. Proc Natl Acad Sci USA 72:4051–4055
Holtzer H, Pacifici M, Tapscott S, Bennett G, Payette R, Dlugosz A (1982) Lineages in cell differentiation and in cell transformation. In: Revoltella RF (ed) Expression of differentiated functions in cancer cells. Raven Press, New York, pp 169
Homann D, Teyton L, Oldstone MBA (2001) Loss of antiviral CD4+ T cell memory is associated with reduced bcl-2 and telomerase expression. Nature Medicine (in press)
Hotchin JE, Cintis M (1958) Lymphocytic choriomeningitis infection of mice as a model for the study of latent virus infection. Can J Microbiol 4:149–163
Hotchin JE, Seegal R (1977) Virus-induced behavioral alteration of mice. Science 196:671–674
Hudrisier D, Mazarguil H, Laval F, Oldstone MBA, Gairin JE (1996) Binding of viral antigens to major histocompatibility complex class I H-2Db molecules is controlled by dominant negative elements at peptide non-anchor residues: implications for peptide selection and presentation. J Biol Chem 271:17829–17836
Ivanovski DI (1899) Ueber die Mosaikkrankheit der Tabakspflanze. Zentralbl. Bakteriol. Parasitenkd. Infektionskr. Hyg Abt II Orig 5:250–254
Jamieson BD, Somasundaram T, Ahmed R (1991) Abrogation of tolerance to a chronic viral infection. J Immunol 147:3521–3529
Joly E, Mucke L, Oldstone MBA (1991) Viral persistence in neurons explained by lack of major histocompatibility complex class I expression. Science 253:1283–1285
Kagi D, Seiler P, Pavlovic J, Ledermann B, Burki K, Zinkernagel RM, Hengartner H (1995) The roles of perforin-and Fas-dependent cytotoxicity in protection against cytopathic and noncytopathic viruses. Eur J Immunol 25:3256–3262
Klavinskis LS, Geckeler R, Oldstone MB (1989a) Cytotoxic T lymphocyte control of acute lymphocytic choriomeningitis virus infection: interferon gamma, but not tumour necrosis factor alpha, displays antiviral activity in vivo. J Gen Virol 70:3317–3325
Klavinskis LS, Oldstone MBA (1989) Lymphocytic choriomeningitis virus selectively alters differentiated but not housekeeping functions: block in expression of growth hormone gene is at the level of transcriptional initiation. Virology 168:232–235
Klavinskis LS, Whitton JL, Oldstone MBA (1989b) Molecularly engineered vaccine which expresses an immunodominant T cell epitope induces cytotoxic T lymphocytes that confer protection from lethal virus infection. J Virol 63:4311–4316
Klenerman P, Rowland-Jones S, McAdam S, Edwards J, Daenke S, Lalloo D, Koppe B, Rosenbrg W, Boyd D, Edwards A, Giangrande P, Phillips RE, McMichael AJ (1994) Cytotoxic T-cell activity antagonized by naturally occurring HIV-1 gag variants. Nature 369:403–407
Koup RA (1994) Virus escape from CTL recognition. J Exp Med 180:779–782
Lampert PW, Oldstone MBA (1974) Pathology of the choroid plexus in spontaneous immune complex disease and chronic viral infections. Virchows Archiv 363:21–32
Leist TP, Zinkernagel RM (1990) Treatment with anti-tumor necrosis factor alpha does not influence the immune pathological response against lymphocytic choriomeningitis virus. Cytokine 2:29–34
Lewicki HA, Tishon A, Borrow P, Evans C, Gairin JE, Hahn KM, Jewell DA, Wilson IA, Oldstone MBA (1995a) CTL escape viral variants. I. Generation and molecular characterization. Virology 210:29–40
Lewicki HA, von Herrath MG, Evans CF, Whitton JL, Oldstone MBA (1995b) CTL escape viral variants. II. Biologic activity in vivo. Virology 211:443–450
Lipkin WI, Battenberg ELF, Bloom FE, Oldstone MBA (1988) Viral infection of neurons can depress neurotransmitter mRNA levels without histologic injury. Brain Res 451:333–339
Lipkin WI, Villarreal LP, Oldstone MBA (1989) Neurotransmitter abnormalities in Borna disease. Brain Res 475:366–370
Loeffler F, Frosch P (1898) Berichte der Kommission zur Erforschung der Maul und Klauenseuche bei dem Institut für Infektionskrankheiten in Berlin. Zentralbl. Bakteriol. Parasitenkd. Infektionskr. Hyg Abt I Orig 23:371–391
Lundstedt C (1969) Interaction between antigenically different cells: virus induced cytotoxicity by immune lymphocytes in vitro. Acta Pathol Microbiol Scand 75:134–147
Marker O, Volkert M (1973) Studies on cell-mediated immunity to lymphocytic choriomeningitis virus in mice. J Exp Med 137:1511–1525
Matloubian M, Concepcion RJ, Ahmed R (1994) CD4+ T cells are required to sustain CD8+ cytotoxic T-cell responses during chronic viral infection. J Virol 68:8056–8063
Matloubian M, Somasundaram T, Kolhekar SR, Selvakumar R, Ahmed R (1990) Genetic basis of viral persistence: single amino acid change in the viral glycoprotein affects ability of lymphocytic choriomeningitis virus to persist in adult mice. J Exp Med 172: 1043–1048
Matsumura M, Fremont DH, Peterson PA, Wilson IA (1992) Emerging principles for the recognition of peptide antigens hy MHC class I molecules. Science 257:927–934
Medawar PB, Medawar JS (1983) Aristotle to Zeus, a philosophical dictionary of biology. Harvard University Press, Cambridge, Massachusetts, pp 275
Mims CA, Blanden RV (1972) Antiviral action of immune lymphocytes in mice infected with lymphocytic choriomeningitis virus. Infect Immun 6:695–698
Moskophidis D, Zinkernagel RM (1995) Immunobiology of cytotoxic T-cell escape mutants of lymphocytic choriomeningitis virus. J Virol 69:2187–2193
Muller U, Steinhoff U, Reis LFL, et al. (1994) Functional role of type I and type II interferons in antiviral defense. Science 264: 1918–1921
Murali-Krishna K, Altman JD, Suresh M, Sourdive DJ, Zajac AJ, Miller JD, Slansky J, Ahmed R (1998) Counting antigen-specific CD8 T cells: a reevaluation of bystander activation during viral infection. Immunity 8: 177–187
Odermatt B, Eppler M, Leist TP, Hengartner H, Zinkernagel RM (1991) Virus-triggered acquired immunodeficiency by cytotoxic T-cell-dependent destruction of antigen-presenting cells and lymph follicle structure. Proc Natl Acad Sci USA 88:8252–8256
Oldstone MBA (1975) Virus neutralization and virus-induced immune complex disease: virus-antibody union resulting in immunoprotection or immunologic injury — two sides of the same coin. In: Melnick JL (ed) Progress in medical virology, Vol. 19. S. Karger, Basel, pp 84–119
Oldstone MBA (1984) Virus can alter cell function without causing cell pathology: disordered function leads to imbalance of homeostasis and disease. In: Notkins AL, Oldstone MBA (eds) Concepts in viral pathogenesis. Springer, Heidelberg Berlin New York, pp 269–276
Oldstone MBA (1985) An old nemesis in new clothing: viruses playing new tricks by causing cytopathology in the absence of cytolysis. J Infect Dis 152:665–667
Oldstone MBA (1987) Molecular anatomy of viral disease. Neurology 37:453–460
Oldstone MBA (1989) Viral alteration of cell function. Sci Amer 260:42–48
Oldstone MBA (1991) Molecular anatomy of viral persistence. J Virol 65:6381–6386
Oldstone MBA (1993) Viruses and diseases of the twenty-first century. Amer J Pathol 143:1241–1249
Oldstone MBA (1997) How viruses escape from cytotoxic T lymphocytes: molecular parameters and players. Virology 234: 179–185
Oldstone MBA, Ahmed R, Buchmeier MJ, Blount P, Tishon A (1985) Perturbation of differentiated functions during viral infection in vivo. I. Relationship of lymphocytic choriomeningitis virus and host strains to growth hormone deficiency. Virology 142:158–174
Oldstone MBA, Blount P, Southern PJ, Lampert PW (1986) Cytoimmunotherapy for persistent virus infection: unique clearance pattern from the central nervous system. Nature 321:239–243
Oldstone MBA, Dixon FJ (1969) Pathogenesis of chronic disease associated with persistent lymphocytic choriomeningitis viral infection. I. Relationship of antibody production to disease in neonatally infected mice. J Exp Med 129:483–505
Oldstone MBA, Dixon FJ (1972) Disease accompanying in utero viral infection: the role of maternal antibody in tissue injury after transplacental infection with lymphocytic choriomeningitis virus. J Exp Med 135:827–838
Oldstone MBA, Habel K, Dixon FJ (1969) The pathogenesis of cellular injury associated with persistent LCM viral infection. Fed Proc 28:429
Oldstone MBA, Holmstoen J, Welsh RM (1977) Alterations of acetylcholine enzymes in neuroblastoma cells persistently infected with lymphocytic choriomeningitis virus. J Cell Physiol 91:459–472
Oldstone MBA, Rodriguez M, Daughaday WH, Lampert PW (1984a) Viral perturbation of endocrine function: disorder of cell function leading to disturbed homeostasis and disease. Nature 307:278–280
Oldstone MBA, Sinha YN, Blount P, Tishon A, Rodriguez M, von Wedel R, Lampert PW (1982) Virus-induced alterations in homeostasis: alterations in differentiated functions of infected cells in vivo. Science 218: 1125–1127
Oldstone MBA, Southern P, Rodriguez M, Lampert P (1984b) Virus persists in beta cells of islets of Langerhans and is associated with chemical manifestations of diabetes. Science 224:1440–1443
Oldstone MBA, Tishon A, Buchmeier MJ (1983) Virus induced immune complex disease: genetic control of C1q binding complexes in the circulation of mice persistently infected with lymphocytic choriomeningitis virus. J Immunol 130:912–918
Oldstone MBA, Tishon A, Chiller J, Weigle W, Dixon FJ (1973) Effect of chronic viral infection on the immune system. I. Comparison of the immune responsiveness of mice chronically infected with LCM virus with that of noninfected mice. J Immunol 110: 1268–1278
Oldstone MBA, Whitton JL, Lewicki H, Tishon A (1988) Fine dissection of a nine amino acid glycoprotein epitope, a major determinant recognized by lymphocytic choriomeningitis virus specific class I restricted H-2Db cytotoxic T lymphocytes. J Exp Med 168:559–570
Oxenius A, Zinkernagel RM, Hengartner H (1998) Comparison of activation versus induction of unresponsiveness of virus-specific CD4+ and CD8+ T cells upon acute versus persistent viral infection. Immunity 9:449–457
Pewe L, Xue S, Perlman S (1998) Infection with cytotoxic T-lymphocyte escape mutants results in increased mortality and growth retardation in mice infected with a neurotropic coronavirus. J Virol 72:5912–5918
Phillips RE, et al. (1991) Human immunodeficiency virus genetic variation that can escape cytotoxic T cell recognition. Nature 354:453–459
Pircher H, Moskophidis D, Rohrer U, Burki K, Hengartner H, Zinkernagel RM (1990) Viral escape by selection of cytotoxic T cell-resistant virus variants in vivo. Nature 346:629–633
Rambukkana A, Oldstone MBA (2001) Manuscript in preparation.
Rammensee HG, Friede T, Stevanovic S (1995) MHC ligands and peptide motifs: first listing. Immunogenetics 41:178–228
Riviere Y, Ahmed R, Southern PJ, Buchmeier MJ, Dutko FJ, Oldstone MBA (1985a) The S RNA segment of lymphocytic choriomeningitis virus codes for the nucleoprotein and glycoproteins 1 and 2. J Virol 53:966–968
Riviere Y, Ahmed R, Southern PJ, Buchmeier MJ, Oldstone MBA (1985b) Genetic mapping of lymphocytic choriomeningitis virus pathogenicity: virulence in guinea pigs is associated with the L RNA segment. J Virol 55:704–709
Rodriguez M, Buchmeier MJ, Oldstone MBA, Lampert PW (1983) Ultrastructural localization of viral antigens in the CNS of mice persistently infected with lymphocytic choriomeningitis virus (LCMV). Amer J Pathol 110:95–100
Rotzschke O, Falk K, Deres K, Schild H, Norda M, Melzger J, Jung G, Rammensee HG (1990) Isolation and analysis of naturally processed viral peptides as recognized by cytotoxic T cells. Nature 348: 252–254
Rowe WP (1954) Studies on pathogenesis and immunity in lymphocytic choriomeningitis infection of the mouse. Research Report NM 005048.14.01. Naval Medical Research Institute, Bethesda, Maryland
Salvato M, Shimomaye E, Southern P, Oldstone MBA (1988) Virus-lymphocyte interactions. IV. Molecular characterization of LCMV Armstrong (CTL +) and that of its variant, Clone 13 (CTL -). Virology 164:517–522
Schrier RD, Oldstone MBA (1986) Recent clinical isolates of cytomegalovirus suppress human cytomegalovirus-specific human leukocyte antigen-restricted cytotoxic T-lymphocyte activity. J Virol 59: 127–131
Sevilla N, Kunz S, Holz A, Lewicki H, Homann D, Yamada H, Campbell KP, de la Torre JC, Oldstone MBA (2000) Immunosuppression and resultant viral persistence by specific viral targeting of dendritic cells. J Exp Med 192:1249–1260
Smelt SC, Borrow P, Kunz S, Cao W, Tishon A, Lewicki H, Campbell KP, Oldstone MBA (2001) Differences in affinity of binding of lymphocytic choriomeningitis virus strains to the cellular receptor α-dystroglycan correlate with viral tropism and disease kinetics. J Virol 75:448–457
Sutcliffe JG, Foye PE, Erlander MG, Hilbush BS, Bodzin LJ, Durham JT, Hasel KW (2000) TOGA: an automated parsing technology for analyzing expression of nearly all genes. Proc Natl Acad Sci USA 97:1976–1981
Teng NM, Oldstone MBA, de la Torre JC (1996a) Suppression of lymphocytic choriomeningitis virus-induced growth hormone deficiency syndrome by disease-negative virus variants. Virology 223: 113–119
Teng NM, Borrow P, Oldstone MBA, de la Torre JC (1996b) A single amino acid change in the glycoprotein of lymphocytic choriomeningitis virus is associated with the ability to cause growth hormone deficiency syndrome. J Virol 70:8438–8443
Tishon A, Borrow P, Evans C, Oldstone MBA (1993a) Virus induced immunosuppression. 1. Age at infection relates to a selective or generalized defect. Virology 195:397–405
Tishon A, Eddleston M, de la Torre JC, Oldstone MBA (1993b) Cytotoxic T lymphocytes cleanse viral gene products from individually infected neurons and lymphocytes in mice persistently infected with lymphocytic choriomeningitis virus. Virology 197:463–467
Tishon A, Lewicki H, Rail G, von Herrath M, Oldstone MBA (1995) An essential role for type I interferon-γ in terminating persistent viral infection. Virology 212:244–250
Tishon A, Oldstone MBA (1990) Perturbation of differentiated functions during viral infection in vivo. In vivo relationship of host genes and lymphocytic choriomeningitis virus to growth hormone deficiency. Am J Pathol 137:965–969
Tishon A, Salmi A, Ahmed R, Oldstone MBA (1991) Role of viral strains and host genes in determining levels of immune complexes in a model system: implications for HIV infection. AIDS Res & Human Retrovir 7:963–969
Traub E (1936) Persistence of lymphocytic choriomeningitis virus in immune animals and its relation to immunity. J Exp Med 63:847–861
Valsamakis A, Riviere Y, Oldstone MBA (1987) Perturbation of differentiated functions in vivo during persistent viral infection. III. Decreased growth hormone mRNA. Virology 156:214–220
Varga SM, Welsh RM (1998) Detection of a high frequency of virus-specific CD4+ T cells during acute infection with lymphocytic choriomeningitis virus. J Immunol 161:3215–3218
Villarete L, Somasundaram T, Ahmed R (1994) Tissue-mediated selection of viral variants: correlation between glycoprotein mutation and growth in neuronal cells. J Virol 68:7490–7496
von Herrath MG, Berger DP, Homann D, Tishon T, Sette A, Oldstone MBA (2000) Vaccination to treat persistent viral infection. Virology 268:411–419
von Herrath MG, Dockter J, Nerenberg M, Gairin JE, Oldstone MBA (1994) Thymic selection and adaptability of cytotoxic T lymphocyte responses in transgenic mice expressing a viral protein in the thymus. J Exp Med 180:1901–1910
Walsh CM, Matloubian M, Liu CC, Ueda R, Kurahara CG, Christensen JL, Huang MT, Young JD, Ahmed R, Clark WR (1994) Immune function in mice lacking the perforin gene. Proc Natl Acad Sci USA 91:10854–10858
Whitton JL, Oldstone MBA (2001) The immune response to viruses. In: Knipe D, et al. (eds) Fields Virology, 4th edn. Lippincott Williams & Wilkins, Philadelphia (in press)
Yanagi Y, Maekawa R, Cook T, Kanagawa O, Oldstone MBA (1990) Restricted V-segment usage in T-cell receptors from cytotoxic T lymphocytes specific for a major epitope of lymphocytic choriomeningitis virus. J Virol 64:5919–5926
Yotsumoto S, Fujiwara H, Horton JH, Mosby TA, Wang X, Cui Y, Ko MS (1996) Cloning and expression analyses of mouse dystroglycan gene: specific expression in maternal decidua at the peri-implantation stage. Human Mol Genetics 5:1259–1267
Young AC, Zhang W, Sacchettini JC, Nathenson SG (1994) The three-dimensional structure of H-2Db at 2.4 Å resolution: implications for antigen-determinant selection. Cell 76:39–50
Young LH, Klavinskis LS, Oldstone MB, Young JD (1989) In vivo expression of perforin by CD8+ lymphocytes during an acute viral infection. J Exp Med 169:2159–2171
Zinkernagel RM, Doherty PC (1974) Restriction of in vitro T cell-mediated cytotoxicity in lymphocytic choriomeningitis within a syngeneic or semiallogeneic system. Nature 248:701–702
Zinkernagel RM, Welsh RM (1976) H-2 compatibility requirement for virus-specific T cell-mediated effector functions in vivo. I. Specificity of T cells conferring antiviral protection against lymphocytic choriomeningitis virus is associated with H-2K and H-2D. J Immunol 117:1495–1502
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2002 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Oldstone, M.B.A. (2002). Biology and Pathogenesis of Lymphocytic Choriomeningitis Virus Infection. In: Oldstone, M.B.A. (eds) Arenaviruses II. Current Topics in Microbiology and Immunology, vol 263. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-56055-2_6
Download citation
DOI: https://doi.org/10.1007/978-3-642-56055-2_6
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-62724-8
Online ISBN: 978-3-642-56055-2
eBook Packages: Springer Book Archive