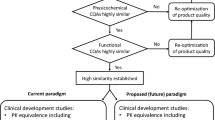
Abstract
Biological medicines have had a profound impact on the health of patients suffering from many debilitating and life threatening diseases. They have been shown to provide “dramatically reduced disability for patients with inflammatory diseases, such as rheumatoid arthritis, extended the lives of patients with many cancers and also provide lifesaving replacement proteins for patients with rare diseases” (US Food and Drug Administration, http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm048341.htm; Krishnan et al., Ann Rheum Dis 71:213–218, 2012; Cox, Biologics 4:299–313, 2010). Biosimilars are copies of biological medicines and in order to obtain approval they are required to undergo head to head similarity exercises [CMC, nonclinical and clinical (as~needed)] against their reference product already marketed in the region/country of interest or countries with stringent regulatory requirements.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Notes
- 1.
Biological Price Competition and Innovation Act of 2009 (BPCIA) allowed for the approval of biosimilar products to the previously licensed innovator biologics in the US: https://www.fda.gov/downloads/drugs/ucm216146.pdf
- 2.
Totality of Evidence requirements are detailed in the FDA guidance tiltled “Scientific Considerations in Demonstrating Biosimilarity to a Reference Product” https://www.fda.gov/downloads/drugs/guidances/ucm291128.pdf
References
Chow S-C, Liu J-p. Statistical assessment of biosimilar products. J Biopharm Stat. 2009;20(1):10–30.
Cox TM. Gaucher disease: clinical profile and therapeutic developments. Biologics. 2010;4:299–313.
Krishnan E, Lingala B, Bruce B, Fries JF. Disability in rheumatoid arthritis in the era of biological treatments. Ann Rheum Dis. 2012;71:213–8.
US Food and Drug Administration. FDA 101: Regulating biological products. US Food and Drug Administration. August 20, 2012. Available at: http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm048341.htm. Accessed 5 Dec 2005.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 American Association of Pharmaceutical Scientists
About this chapter
Cite this chapter
Chance, K. (2018). Design and Implementation of Successful Regulatory Strategies in Biosimilar Development. In: Gutka, H., Yang, H., Kakar, S. (eds) Biosimilars. AAPS Advances in the Pharmaceutical Sciences Series, vol 34. Springer, Cham. https://doi.org/10.1007/978-3-319-99680-6_5
Download citation
DOI: https://doi.org/10.1007/978-3-319-99680-6_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-99679-0
Online ISBN: 978-3-319-99680-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)