Abstract
Predominantly antibody deficiencies (PADs) are the most frequent forms of primary immunodeficiency diseases (PIDs). Commonly accompanied with complications involving several body systems, immunoglobulin substitution therapy along with prophylactic antibiotics remained the cornerstone of treatment for PADs and related complications. Patients with respiratory complications should be prescribed an appropriate therapy as soon as possible and have to be adhering to more and longer medical therapies. Recent studies identified a gap for screening protocols to monitor respiratory manifestations in patients with PADs. In the present chapter, the pulmonary manifestations of different PADs for each have been discussed. The chapter is mainly focused on X-linked agammaglobulinemia, common variable immunodeficiency, activated PI3K-δ syndrome, LRBA deficiency, CD19 complex deficiencies, CD20 deficiency, other monogenic defects associated with hypogammaglobulinemia, immunoglobulin class switch recombination deficiencies affecting B-cells, transient hypogammaglobulinemia of infancy, and selective IgA deficiency.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
- Bruton-type agammaglobulinemia
- Agammaglobulinemia
- Non-Bruton type
- Activated PI3K-delta syndrome
- Common variable immunodeficiency
- Turcot syndrome
3.1 Introduction
Predominantly antibody deficiencies (PADs) are the most frequent forms of primary immunodeficiency diseases (PIDs). These conditions are resulted from a primary defect in B-cells. Though to a lesser extent, they are caused by a defect in T-cells or other immune cell populations known to contribute to B-cell or plasma cell development and function. Overall, PADs are characterized by a malfunctioned antibody response which is reflected in low or undetectable levels of immunoglobulin(s). As a result, recurrent infection is the most common presentation leading to diagnosis of PADs. It would also remain the cause of most complications during the course of disease. Overall, physicians who are the most likely to encounter patients with PADs are those of infectious disease specialists [1].
Patients with chronic and recurrent respiratory infections are prone to develop severe respiratory conditions such as bronchiectasis and obliterative bronchiolitis. Therefore, patients with respiratory infections need particular attention. They should be prescribed an appropriate therapy as soon as possible and have to be adhering to more and longer medical therapies. Immunoglobulin substitution therapy along with prophylactic antibiotics remained the cornerstone of treatment for PADs and related complications [2]. Generally, immunoglobulin replacement therapy can effectively reduce both incidence and severity of infections [2]. However, immunoglobulin products contain only purified IgG antibodies and lack other antibody isotypes. It is thus expected that pulmonary infections may persist and even flourish under regular immunoglobulin replacement therapy. Thereby, the patient will be more predisposed to chronic lung diseases and related severe sequels such as respiratory failure [3]. Recent studies have identified a gap for screening protocols to monitor respiratory manifestations in patients with PADs [4].
(For further information you may see Aghamohammadi A, Plebani A, Lougaris V, Durandy A, Condino-Neto A, Kanegane H, Hammarström L. Predominantly antibody deficiencies. In: Rezaei N, Aghamohammadi A, Notarangelo LD, editors. Primary immunodeficiency diseases: definition, diagnosis, and management. 2nd ed. p. 183–244.)
3.2 X-Linked Agammaglobulinemia (Bruton’s Disease or BTK Deficiency)
Among the most severe types of PADs is X-linked agammaglobulinemia (XLA), the first PID described by Ogden Bruton in 1952. XLA is the most common monogenic immunodeficiency [5] resulting from mutations on the X chromosome in the gene encoding a tyrosine kinase, the so-called Bruton’s tyrosine kinase (Btk). These mutations are of loss-of-function type, making an arrest in the early stages of B-cell development [5]. XLA is therefore a humoral immunodeficiency characterized by depletion of B-cells and low levels of all immunoglobulins (IgG, IgA, IgM, IgD, and IgE) [6]. While the number of T-cells and NK cells varies within normal range, it is however realized that lack of B-cells might lower the optimal function of T-cells, as reflected in the diminished T-cell memory to specific antigens. For example, patients with XLA show a defect in T-cell memory to N. meningitidis but not influenza [7]. The presentation of this antibody immunodeficiency is expected to occur (a) earlier than other types of PADs, (b) after maternal antibodies waned, and (c) during the first 2 years of life with recurrent and severe sinopulmonary infections [8,9,10,11]. However, there are reports, for example, from China, where the mean age at diagnosis was more than 6 years. Even, there have been reports of late-onset XLA (up to the fourth decade of life) [12]. Overall, the mean age at diagnosis falls within the range 3.2–7.7 years, whereas the mean age at onset of symptoms happens within the range 1.8–4.2 years [13,14,15,16,17, 10, 18]. This reflects the delay in diagnosis ranging from 1.4 to 3.6 years.
In conjunction with its expression on different cells such as B-cells, monocytes, macrophages, granulocytes, dendritic cells, and osteoclasts, BTK serves as a potential contributor to various intracellular functions, essentially B-cell development [19] and differentiation [20], natural killer (NK) cell activation [21], and T-cell memory [22]. While BTK deficiency can leave the body in a state of immunodeficiency (e.g., XLA), its upregulation may lead to autoimmune states, such as rheumatoid arthritis and systemic lupus erythematosus [19], as well as malignant states, notably B-cell malignancies [23], squamous cell carcinoma, and pancreatic cancer [24]. Hundreds of variants have been identified in the gene BTK from patients with XLA [25,26,27,28,29, 18] which may explain phenotypic divergence in XLA [30]. Therefore, data are integrated to investigate genotype-phenotype interactions [31]. Overall, the most common complications of XLA are pulmonary infections and bronchiectasis.
3.2.1 Pulmonary Infections
Generally, infections account for the highest proportion of presenting manifestations of XLA [9, 10, 16, 29]. In particular, a prospective study of 101 individuals with XLA reported the presence of at least one of these three infectious complications: pneumonia, sinusitis, and chronic lung disease with bronchiectasis in 76% of patients with XLA [14]. Moreover, the majority of infections affect the upper and lower respiratory tracts [16, 10, 18, 32] and are of bacterial origin, among which bronchitis, pneumonia, chronic sinusitis, otitis, and conjunctivitis are the most common complications of XLA [33]. Infections occur not only before diagnosis but remain a major cause of complications during the course of disease, immunoglobulin substitution, and death in patients with XLA [32, 10]. The most common infections prior to and after diagnosis include otitis media, pneumonia, and sinusitis [9, 29].
Pneumonia is the most common acute infection associated with XLA [14]. Recurrent pneumonia should be, therefore, regarded as an alarm sign for PIDs including XLA [34, 35]. Before the diagnosis is made, between 60% and 83% of patients with XLA have a history of at least one episode of pneumonia [36], and approximately 30% of patients have a history of hospitalization due to pneumonia [16]. With an adequate immunoglobulin therapy, the rate of pneumonias is reduced by more than 10% [14] and lies within the range of 0.00–0.10 pneumonias per treatment year [13, 37]. However, approximately 50% of patients might experience deterioration in their respiratory status while receiving immunoglobulin therapy [38]. The rate of infectious episodes during immunoglobulin therapy seems to be age-related, since there is a threefold increased risk for infections in adult patients compared with pediatric patients (2.12 vs. 0.74 infections/patient/year) [38]. More important is that pneumonias after initiation of immunoglobulin therapy might be severe as many as half of episodes might require hospitalization for intravenous antibiotic treatment [13]. In multivariate analysis, patients with bronchiectasis had a > 3-fold increased risk of pneumonia during the 5-year immunoglobulin therapy [14]. Its source remained unknown in almost 80% of cases. However, sputum cultures could reveal the following as bacterial pathogens underlying pneumonia in patients with XLA: Haemophilus influenzae, Streptococcus pneumoniae, Staphylococcus aureus, and Mycoplasma pneumonia [13, 33]. Analysis of bronchoalveolar lavage (BAL) in patients with XLA without acute pulmonary function identified bacterial pathogens such as Haemophilus influenzae and Veillonella [39]. Also, bacteria that were observed through protected specimen brush samples included Haemophilus influenzae, β-hemolytic streptococci, and Porphyromonas [39]. Other bacterial pathogens found in association with pneumonia among patients with XLA are Pseudomonas species and M. tuberculosis [16]. Looking at chest X-ray, pneumonia in patients with XLA can cause peribronchial thickening and then segmental atelectasis [13]. Antigen-specific immunological responses might predict the risk of respiratory tract infections in patients with XLA. More precisely, individuals who developed IgG and IgM responses specific to bacteriophage phi-X 174 were less likely to be affected by respiratory tract infections (RTI) compared with those who developed only IgM responses [22]. Although very rare, there are reports of pulmonary infections with fungal (aspergillosis and Pneumocystis jiroveci) [40] and viral pathogens (respiratory syncytial virus) [41,42,43] in patients with XLA even despite regular immunoglobulin substitution therapy. Chronic sinusitis seems to predispose individuals with XLA to pulmonary aspergillosis [40].
3.2.2 Bronchiectasis
There are individuals with impaired pulmonary function, as revealed through pulmonary function test, that show a normal chest X-ray [13]. However, an abnormal chest X-ray clearly warrants that pulmonary function is impaired [13]. Patients with XLA show changes in pulmonary function test (PFT) that favor obstructive lung disease. These include a reduction in forced expiratory volume in the first second (FEV1) as well as a reduction in forced expiratory flow (FEF) [13]. Pulmonary function abnormalities appear not to be age-related [13]. The delay in diagnosis has shown an inverse association with the risk of chronic lung disease. Supporting this, lung function decreases with aging in patients with XLA and is worsened by smoking and bronchiectasis. A longitudinal study of patients with XLA (n = 8) over an average period of 7.6 years calculated an average annual decline of 65 ± 11 mL/year and 58 ± 20 mL/year for FEV1 and FVC, respectively [15]. Immunoglobulin therapy dose demonstrated a negative association with lung function decline. This reflects the protective effect of immunoglobulin therapy [15].
Evidence of chronic lung disease (CLD) before diagnosis is present in nearly one-third of patients with XLA [16]. Factors known to increase the risk of CLD before diagnosis of XLA include higher age at diagnosis and the presence of a lower respiratory tract infection (LRTI) [16]. This may indicate that delayed diagnosis might leave patients facing higher risk of CLD [18]. CLD is observed in 40% of patients with XLA who had a history of LRTI. Approximately, 30% of patients develop CLD later in the course of disease [16]. CLD risk after diagnosis of XLA is predicted by pneumonia and inappropriate immunoglobulin substitution therapy [16]. Study of 201 US patients with XLA identified CLD as the cause of death in 25% of cases [9]. CLD also has been shown to have a negative impact on the life quality of patients with XLA [44].
Keeping in mind the high incidence of pulmonary infections, bronchiectasis is considered a common complication of XLA. A study of Australian adults estimated a twofold incidence of infection with bronchiectasis compared to infection without bronchiectasis among patients with XLA (67% vs. 33%) [45]. In a multicenter study of 199 patients with XLA, the use of high-resolution chest CT imaging indicated a prevalence of bronchiectasis as high as 56% [33]. The prevalence of bronchiectasis increases with age [14] and that turning 18 years old was the most important factor predictive of bronchiectasis [33]. However, other factors associated with an increased risk of bronchiectasis included a history of pneumonia and treatment with IVIG compared with subcutaneous IG [33]. In addition, patients with chronic sinusitis were four times more likely to have bronchiectasis [14]. It is important that almost half of patients with XLA suffer from chronic sinusitis [14], whereas no effect of IgG levels on the risk of bronchiectasis was found [14]. The age range for development of bronchiectasis was 7–45 years [33]. Regardless of age, bronchiectasis lowers lung function as reflected in reduced FEV1 [33]. Bronchiectasis is, therefore, the real cause of diminished life quality among patients with XLA [38]. CXR findings are often evident in the middle and lower lobes of the lung(s) [11]. HRCT appears to be more sensitive than CXR for evaluation of pulmonary abnormalities in patients with primary hypogammaglobulinemia including XLA [46].
3.2.3 Chronic Pleurisy
There is report of recurrent chronic pleurisy presented with thickened pleura and calcification in CT imaging and intraluminal fibrosis, foamy alveolar macrophages, and chronic inflammation in histopathological examination of the pleural tissue [47]. Broad-range bacterial polymerase chain reaction (PCR) identified H. equorum-like bacterium as the underlying pathogen of these abnormalities. Taking high-dose panipenem/betamipron (PAPM/BP) and clarithromycin significantly improved the patients’ situation.
3.2.4 Potential Mechanisms of Respiratory Manifestations in XLA
Both lipopolysaccharide (LPS) and anti-IL-8/IL-8 immune complexes act as stimulus to neutrophils [48]. Upon stimulation, FcγRIIa is recruited from intracellular compartments to the cell surface. FcγRIIa recruitment is accompanied with activation of Btk, which in turn induces the expression of adaptor molecule MyD88. In this manner, Btk act to direct cross talk between FcγRIIa and toll-like receptor 4 (TLR4) [49]. It is followed by engagement of MyD88 adapter-like (Mal)/TIRAP interaction which plays a crucial role in TLR-dependent NF-kappaB (NF-κB) pro-inflammatory responses [50]. Matrix metalloproteinases (MMP) mainly MMP9 induced by NF-κB contribute to tissue remodeling in acute lung injury. As expected, Btk inhibitors could abrogate the expression of both Btk and MyD88 in human neutrophils activated with anti-IL-8/IL-8 immune complexes [48]. Additionally, treatments blocking either Btk or MMP9 diminished the expression of MMP9 in neutrophils from mice exposed to secondhand smoke [51]. Altogether, it is plausible to think of Btk-targeted neutrophil-specific therapy in conditions associated with acute lung injury (ALI)/acute respiratory distress syndrome (ARDS) [48] and chronic obstructive pulmonary disease (COPD) [51].
Ibrutinib is used in targeted therapy of malignancies and autoimmune diseases to inhibit BTK. It has been shown to prevent influenza-induced acute lung injury through reduction of pro-inflammatory mediators [52]. Btk inhibitor RN983 showed much more anti-inflammatory potency than corticosteroid budesonide in mouse lung in form of inhalation [53]. This makes it effective in the treatment of allergic asthma. RNA interference (RNAi) of Btk has been demonstrated to decrease protein levels of Btk and phosphorylated Btk (p-Btk) and diminish the expression and activation of Btk in alveolar macrophages [54]. In a mouse model of cecal ligation and puncture (CLP)-induced sepsis, pretreatment with Btk RNAi was able to prevent epithelial cell apoptosis, pulmonary edema, vascular permeability, the expression of inflammatory cytokines (TNF-α, IL-1β, and IL-6) and signaling (NF-κB), and neutrophil lung infiltration.
On the contrary, there is evidence that the over-suppression of innate immunity by ibrutinib might cause toxic events. Ibrutinib can lead to epithelial cell apoptosis and inflammation that promote pulmonary infections [55] and fibrosis [56]. Even, there is report of invasive aspergillus infection in association with ibrutinib in patients with chronic lymphocytic leukemia [57]. These toxic effects induced by ibrutinib draw a picture of what happens in patients with XLA, where loss-of-function mutations lead to downregulation of Btk.
Study of XLA mice suggest that IgM contributes to alveolar macrophage phagocytosis and thereby confining the fungal infection to limited areas [58]. In this manner, low IgM levels in patients with XLA make them prone to develop more severe infections with fungal pathogens such as Cryptococcus neoformans.
B-cells act as the regulator of neutrophil migration to the site of stimulus. Depletion of B-cells in XLA dictates the rapid migration of neutrophils to the site of stimulus whereby the opportunity of macrophages to attract to the site of stimulus is captured, while macrophage activation is required for engagement of interferon gamma (IFNy)-producing T-cells, which in turn elicit an effective response to bacillus Calmette-Guérin (BCG) vaccination [59]. In this manner, impairment in antituberculosis immunity exists as a result of disrupted IFNy-producing T-cell-macrophage axis in XLA.
TLR signaling pathways as well as TLR effector function are maintained despite loss of Btk function [60]. Neutrophils from patients with XLA upon stimulation with LPS revealed activation of MAPK cascades, production of reactive oxygen species (ROSs), and reduction of neutrophil apoptosis. All these events occurred in a comparable fashion in neutrophils from controls. Particularly, ROSs are required for phagocytosis of pathogens [61].
Impairment in B-cell development causes airway inflammation [62], and therefore allergic symptoms might be present in patients with XLA [63]. Compared with wild-type mice, X-linked immunodeficiency (Xid) mice exhibited an increase in production of cytokines (IL-4, IL-5, IL-10, IL-13, CCL5, and IFNy), lung inflammation, interstitial eosinophilia, and mucus production in response to challenge with cockroach antigen.
3.2.5 Management of Respiratory Manifestations in XLA
Immunoglobulin therapy helps to restore IgG levels to protective levels [13]. Patients on high-dose intravenous immunoglobulin therapy rather than low-dose or intramuscular immunoglobulin therapy are more likely to achieve recommended levels of IgG [64]. Analysis of serial determination of IgG levels in patients with XLA receiving IVIG therapy indicated that a median IgG level of 354 mg/dl predicts that individual immunity against infections is acceptable [65]. The annual incidence of bacterial infections (pneumonia) was estimated to be 0.16 (0.12), 0.05 (0.05), and 0.00 (0.00) when IgG levels are below 500 mg/dL, between 500 and 800 mg/dL, and above 800 mg/dL [37]. Of note, a 5-year prospective study of 101 individuals with XLA identified only one episode of pneumonia in cases with IgG levels >1000 mg/dL [14]. Individuals taking a high dose of IVIG (397 mg/kg) are expected to be infection-free [65].
Lung transplantation (LTx) is the option available for patients with bronchiectasis and end-stage respiratory failure caused by XLA. Even after LTx, regular immunoglobulin substitution is still required for prevention of infections [66]. Although it is promising that the lungs often reach an acceptable or predicted functional level, post-lung transplant infection and respiratory failure remain life-threatening [45]. The authors in [67] reported the long-term outcome of six patients with XLA who underwent LTx. All patients but one died within the first 3 years after LTx. The pulmonary infection (bronchiolitis and pulmonary sepsis) was the cause of death for all but one case who died from progressive multifocal leukoencephalopathy.
3.3 Autosomal Recessive Agammaglobulinemia (μ Heavy Chain Deficiency, λ5 Deficiency, Igα Deficiency, Igβ Deficiency, BLNK Deficiency)
When we have a male child with congenital hypogammaglobulinemia and absent B-cells, the first differential diagnosis to consider is XLA, which is resulted from mutant BTK. When we have a female child with similar clinical and immunological presentation, the first differential diagnosis to consider is autosomal recessive (AR) agammaglobulinemia (ARA). Mutations identified as the cause of autosomal recessive forms of agammaglobulinemia occur in the genes encoding molecules that contribute to the structure and function of pre-B-cell receptor (BCR) or its downstream pathways such as the μ heavy chain (IGHM), B-cell linker adaptor protein (BLNK), the immunoglobulin λ-like polypeptide1 (IGLL1), leucine-rich repeat-containing 8 (LRRC8A), Igα (CD79A), and Igβ (CD79B) [31].
As pre-B-cells are known as precursors of B-cells, the pre-B-cell receptor (BCR) is considered as precursor of B-cell receptor. As a result, pre-BCR is an essential to B-cell development. It is composed of the μ heavy chain (μHC) and the surrogate light chain (SLC). The SLC, in turn, includes invariant Vpre-B and λ5 polypeptides. Also, transmembrane protein Igα in association with Igβ serves as the signal transduction component for the pre-BCR complex. Abnormalities in pre-BCR structure and function hinder the transition from pro-B- to pre-B-cell stage [68], predisposing individuals to immunodeficiency, malignancy, and autoimmunity (for review see [69]).
3.3.1 μ Heavy Chain Deficiency
Yel et al. 1996 [70] were the first group to identify mutations in the IGHM gene as a cause of agammaglobulinemia. They reported seven patients from two families with AR B-cell defects. Patients presented between 1 and 15 months of age. Clinical manifestations included fever, weakness, rashes, chronic enteroviral encephalitis, recurrent infections (pneumonia, bronchopneumonia, and otitis), failure to thrive, gastrointestinal disorders, septic shock (due to Pseudomonas aeruginosa), arthritis, and perirectal abscesses.
Thereafter, more studies have investigated the IGHM as a potential candidate gene for AR B-cell defects [31, 71,72,73,74]. These studies indicate that (a) the IGHM gene is very polymorphic [72], (b) the presence of large deletions rather than point mutations is more likely in patients with agammaglobulinemia [74], and (c) mutant IGHM accounts for about 20–30% of patients who have AR B-cell defects [71] and as well for about 5% of all patients with agammaglobulinemia [74]. Also, it is concluded that patients carrying mutant IGHM experience an earlier onset and more difficult course of agammaglobulinemia compared to patients carrying mutant BTK [71]. Overall, mutant IGHM may cause respiratory manifestations such as recurrent upper respiratory tract infections (viral rhinitis, sinusitis, otitis, pharyngitis, and rhinopharyngitis), recurrent pneumonia, bronchopneumonia, bronchiectasis, and asthma.
3.3.2 λ5 Deficiency
Studies show that despite allelic exclusion of Ig μ heavy chain would remain normal, B-cell development is, however, not completely [75] ablated in mice deficient in λ5 [76]. Minegishi et al. 1998 [77] reported a case with hypogammaglobulinemia, less than 1% of normal number of B-cells, and undetectable levels of CD19. He presented with recurrent otitis at 2 months of age.
3.3.3 Igα Deficiency
Minegishi et al. 1999 [78] published the first case of defect in Igα (CD79a) with agammaglobulinemia. The patient presented with recurrent diarrhea, failure to thrive, bronchitis, and neutropenia. The age at onset of disease was before 1 month of age. The authors demonstrated that defect in the Igα gene results in complete blocking of B-cell development as defect in the IGHM. This study proved that the functional role of Igα as a component of signal transduction molecule is not less than the role that the mu heavy chain plays as a structural component of pre-BCR. The second one was reported by Wang et al. 2002 [79]. Clinical and immunologic features consisted recurrent upper and lower respiratory tract infections, otitis media, weakness, almost complete absence of B-cells but not T-cells, hypogammaglobulinemia, dermatomyositis, and diarrhea.
3.3.4 Igβ Deficiency
Study in Drosophila melanogaster [80] showed that mutant Igβ is a contributing factor to the dissociation of Igα/Igβ, whereby the pre-BCR complex cannot be assembled and B-cell development is blocked. Moreover, deletion of Igβ dictated death in murine developing B-cells including pre-B-cells and immature B-cells [81]. The first patient carrying mutant Igβ (CD79b) was described by Dobbs et al. 2007 [82]. She presented with recurrent bronchitis, persistent cough, pneumonia, and hypogammaglobulinemia. The age at onset of symptoms was about 5 months. Ferrari et al. 2007 [80] were the second group who found mutant Igβ in a patient presented with less than 1% of normal CD19 levels, hypogammaglobulinemia, but normal counts of T and NK cells. His symptoms began to appear at 8 months of age and included recurrent pneumonia, conjunctivitis, otitis media, sinusitis, and bronchitis. At the time of evaluation, he was 20 and demonstrated chronic sinusitis at his CT scan. The third case was recently introduced by Lougaris et al. 2014 [83]. The patient was a female child, whose symptoms started at 14 months of age. She presented with recurrent upper respiratory tract infections, fever, neutropenia, profound hypogammaglobulinemia, and complete absence of B-cells but normal numbers of T and NK cells.
3.3.5 BLNK Deficiency
BLNK is essential for B-cell development and function. Mice deficient in BLNK failed to generate B220 + CD43− precursor B-cells and lacked mature B-cells in the periphery [84]. BLNK deficiency also led to reduce IL-10 production in mice [85]. Mutant BLNK in a patient lacking pre-B-cells or mature B-cells was first reported in 1999 [86]. The patient presented with recurrent otitis, pneumonia, and hypogammaglobulinemia. His symptoms commenced at 8 months of age. Other studies that have found mutant BLNK in association with agammaglobulinemia can be found here [87,88,89]. They confirm recurrent otitis and sinopulmonary infections as the most common initial presentation of BLNK deficiency [88, 89]. However, other clinical manifestations included diarrhea, enteroviral infection, arthritis, dermatitis, and bronchiectasis [89].
3.4 Other Forms of Agammaglobulinemia with Absent B-Cells (TCF3 Deficiency, LRRC8 Deficiency, Thymoma with Immunodeficiency)
3.4.1 TCF3 Deficiency
Studies of mice have shown that the transcription factor 3 (TCF3) gene plays a crucial role in B-cell development [90,91,92]. This gene which is also known as E22 gene encodes transcription factors E12 and E47. Mutations in TCF3 have been reported in both autosomal recessive [93] and autosomal dominant [94] agammaglobulinemia. The authors in [93] described a patient with B-cell acute lymphoblastic leukemia (B-ALL) presented with profound hypogammaglobulinemia, recurrent pneumonia, meningitis, pancytopenia, and splenomegaly. His immunological findings included less than 1 percent normal number of CD19+ B-cells in the periphery in the presence of normal counts of CD3+, CD4+, and CD8+ T-cells. Boisson et al. 2013 [94] identified the same de novo mutation in the TCF3 gene (within the exon that encodes E47) in four patients with an unusual phenotype characterized by almost complete absence of BCR in the presence of increased expression of CD19 on B-cells [95]. All patients presented severe hypogammaglobulinemia and less than 3 percent CD19+ cells in the periphery. Age at diagnosis ranged from 9 months to 4 years. Clinical presentations included pneumococcal meningitis, recurrent otitis, vaccine-associated polio, and arthritis. Other clinical findings were dermatitis and hepatomegaly.
3.4.2 LRRC8 Deficiency
Sawada et al. 2003 [96] are the first group that isolated the leucine-rich repeat-containing 8 (LRRC8) and showed that B-cell development is impaired in mouse model for a truncated expression of LRRC8. They also provided a report describing a girl patient with agammaglobulinemia, absence of B-cells, epicanthic folds, mild hypertelorism, high-arched palate, and lowered ears. The patient carried mutant LRRC8. To our knowledge, no other report of mutant LRRC8 in association with agammaglobulinemia has been published.
3.4.3 Thymoma with Immunodeficiency (Good’s Syndrome)
Good’s syndrome (GS) or thymoma with immunodeficiency refers to the combined conditions of thymoma and immunodeficiency. It is mainly characterized by thymoma, hypogammaglobulinemia, and low number of B-cells in the periphery. However, low number of T-cells and an inverted CD4/CD8 ratio are also common.
Tarr et al. 2001 [97] in the study provided a report of 5 patients with GS presented with infection as well as a review of 46 other patients with GS and infections. Infections have a propensity to affect the respiratory and gastrointestinal tract [98, 99]. The following are infectious complications documented in GS: recurrent sinopulmonary infection, CMV disease, bacteremia, oral or esophageal candidiasis, persistent mucocutaneous candidiasis, chronic diarrhea, urinary tract infections, P. carinii pneumonia, tuberculosis, Kaposi sarcoma, disseminated varicella, candidemia, wound infection with Clostridium perfringens, Mycoplasma arthritis, etc. However, the main clinical findings at presentation appear to be sinopulmonary infections with encapsulated bacteria. When compared to patients with CVID, patients with GS experience a more difficult course of immunodeficiency complicated with opportunistic infections such as P. carinii pneumonia, mycobacterium tuberculosis, and mucocutaneous candidiasis, resembling that which occurs in human immunodeficiency virus (HIV) infection [100]. Therefore, if a patient with thymoma or CVID develops refractory infections, then we should consider GS as a differential diagnosis. Particularly, persistent infection (pneumonia) following thymectomy might warrant that your patient is very likely to have GS [101,102,103,104,105]. Overall, respiratory manifestations in association with GS include recurrent sinopulmonary infections (sinusitis, rhinosinusitis, otitis media, and pneumonia) [106,107,108,109,110,111,112,113,114] with bacterial (especially with encapsulated bacteria including Haemophilus influenzae), fungal (aspergillus and P. carinii), and viral (CMV and HSV) pathogens [115,116,117,118,119,120,121], tracheobronchitis [118], diffuse panbronchiolitis [122,123,124], granulomatous lung disease [125], and pulmonary nodules [123]. As for patients with other forms of agammaglobulinemia, patients with GS remain dependent on immunoglobulin replacement therapy. A recent study has estimated the median survival of 14 years for 47 patients with GS [111].
3.5 Activated PI3K-δ Syndrome
Activated PI3K-δ syndrome (APDS) is a heterogeneous disorder associated with a spectrum of clinical pictures and immunological findings classified under two types [126]. Autosomal dominant gain-of-function mutations (E1021K and C416R) in the gene PIK3CD (PI3K-δ, phosphoinositide 3-kinase δ) can cause activated PI3K-δ syndrome type 1 (APDS1), while activated PI3K-δ syndrome type 2 (APDS2) is caused by an autosomal dominant gain-of-function mutation in the gene PIK3R1 (phosphoinositide-3-kinase regulatory subunit 1), inducing the skipping of exon 11. Both types are characterized with similar immunological findings including low levels of IgG2 and lymphocytes and high levels of IgM and B-cells [127]. Overall, patients with APDS are prone to recurrent respiratory infections, airway disease [127], lymphoproliferation, cytopenias, skin diseases, chronic EBV and CMV viremia, and enteropathy [126]. All these major manifestations tend to decrease over time [126]. Of notable importance is that respiratory infections are the most frequent, initial manifestations of APDS [126].
The prospective European Society for Immunodeficiencies (ESID)-APDS registry has recently reported data on 51 patients with APDS1 and 26 patients with APDS2 [126]. Overall, respiratory infections, particularly pneumonia, otitis media, and sinusitis, were observed in nearly all patients and in most patients occurred before 15 years of age. Looking at CT scans, evidences of bronchiectasis were found in 60% of patients with APDS1 and 27% of patients with APDS2. Mean age at diagnosis of bronchiectasis was 11.2 years. Therapeutic options available to patients with APDS include antibiotics, antifungal agents, immunoglobulin replacement therapy, systemic immunosuppressive therapy, and HSCT [126]. Particularly a cohort study of 36 patients with APDS2 revealed that recurrent upper respiratory tract infections were experienced by all patients [128]. Other common respiratory complications in this population included pneumonia and respiratory tract lymphoid hyperplasia, which were found in about 70% and 50% patients.
3.5.1 Potential Mechanisms of Respiratory Manifestations in APDS
PI3K as lipid kinases play role to maintain normal function of airway. Abnormal PI3K signaling can alter airway function, so that inflammatory responses are aggravated [129]. Such a condition is seen in patients with respiratory diseases especially allergic inflammation and asthma. Airway biopsies from 11 patients with atopic asthma provide evidence of increased activation of the PI3K signaling after exposure to allergen [130].
PI3K-δ is a class I PI3K isoform proven to particularly contribute to both innate and adaptive immune responses [129]. Such contribution is resulted from its interaction with Akt, together triggering the PI3K-δ/Akt signaling pathway. Increased activation of this signaling pathway is thought to mediate airway inflammation as seen in obstructive lung diseases [131, 132] and induced by cigarette smoking [133]. Moreover, PI3K-δ is able to reduce glucocorticoid sensitivity of airways ([131, 132]. This explains why some patients are resistant to glucocorticoids. Supporting this, PI3K-δ inhibitor can mitigate allergic inflammation in asthma models [134]. The effect remained true upon exposure to Aspergillus fumigatus. At least part of this anti-inflammatory effect of PI3K-δ inhibitor appears to be mediated through reducing endoplasmic reticulum stress [135], hypoxia-inducible factor-1α (HIF-1α), and nucleotide-binding domain, leucine-rich-containing family, and pyrin domain-containing-3 (NLRP3) expression in lung tissue [136, 134]. All these factors are involved in generating inflammatory responses upon stimulation [136]. Additionally, PI3K-δ contributes to virus escape possibly by increasing the expression of B7-H1 (PD-L1) [137]. Therefore, it is well-expected that APDS, a condition associated with PI3K-δ upregulation, should be accompanied with bacterial and viral infections as well as different respiratory manifestations.
The current literature also supports the potential of PI3K-δ to be exploited as a target for treatment of respiratory infections and airway diseases [138, 139]. For example, anti-inflammatory effect of erythromycin and dexamethasone seems to be mediated via suppression of PI3K-δ signaling [140]. Animal models of asthma demonstrate that PI3K-δ blockade hampers infiltration of inflammatory cells by decreasing vascular permeability [141]. Moreover, PI3K-δ inhibition can help to regain glucocorticoid sensitivity and, therefore, enhance anti-inflammatory effects of glucocorticoids [131, 132, 142]. However, attention should be paid that over-suppression of this pathway might cause toxicities. As for APDS, PI3K-δ function is abnormally high in malignant states particularly B-cell malignancies [143]. As a result, PI3K-δ inhibitor (idelalisib) can be used to treat these malignancies [144]. However, it might reduce cellular respiration and so be toxic to lung tissues [145].
3.6 LRBA Deficiency
Lipopolysaccharide (LPS)-responsive and beige-like anchor (LRBA) protein deficiency is a rare PID characterized by hypogammaglobulinemia and CD19+ B-cell deficiency and also to a lesser extent by CD4+ T-cell deficiency and NK-cell deficiency [146]. Patients with LRBA deficiency often present with recurrent infections (pneumonia, urinary tract infections, and otitis media), lymphoproliferative disorders (lymphadenopathy, splenomegaly, hepatomegaly, and granuloma), autoimmune disorders (type 1 diabetes, ulcerative colitis, immune thrombocytopenic purpura, autoimmune hemolytic anemia, and Graves’ disease), atopic disorders (food allergy, insect sting allergies, allergic dermatitis, urticaria, and asthma), enteropathy, and failure to thrive [147, 148, 146]. Therefore, LRBA can be appropriately considered as a subgroup of CVID with autoimmune and inflammatory features.
A retrospective cohort of ten patients with LRBA deficiency in Israel highlighted chronic cough as the most common respiratory manifestation in patients with LRBA deficiency [147]. Less frequent respiratory manifestations were dyspnea, perioral cyanosis, and clubbing. Chest X-rays and CT scans in these cases provided evidences of axillary, hilar, and mediastinal lymphadenopathy, consolidation, lobar and sub-lobar atelectasis, bronchiectasis, and interstitial lung disease [147]. Patients with LRBA deficiency demonstrated no serious abnormalities on PFT. They also were likely to have a normal bronchoscopy. Potential findings in bronchoscopy included tracheomalacia and adenoid hypertrophy. The mean age at onset of symptoms was 4.65 [147].
In a longitudinal study of patients with LRBA (n = 17) in Iran [146], respiratory tract infections were the most common first presentation of immunodeficiency, seen in more than 40% of patients. Additionally, there were two patients (11.8%) presented with allergy and asthma. The median age at onset of symptoms and diagnosis were 2.0 and 7.0 years, respectively. With a median follow-up of 14 years, all patients (100%) developed infectious complications, which in order of frequency were pneumonia, sinusitis, and otitis media.
There is a case report of bronchiolitis obliterans organizing pneumonia (BOOP) in a 10-year-old boy who presented with recurrent respiratory infections, anemia, and thrombocytopenia [149]. He showed hypogammaglobulinemia of IgA and IgG, while IgM levels were normal. He was refractory to regular IVIG and all antimicrobial agents, and his respiratory condition deteriorated during these treatments. Diffuse lung disease with peripheral nodules was found at CT scans. PCR of BAL fluid was positive for Pseudomonas aeruginosa, Pneumocystis jiroveci, and CMV. Looking at lung tissue, there were evidences of disseminated infiltration of inflammatory cells and bronchiolitis obliterans organizing pneumonia (BOOP). To establish genetic causality, combined homozygosity mapping and exome sequencing was performed and showed a homozygous mutation (NM_001199282: c.743_744insAAGA: p. Asp248Glufs*2) in the gene LRBA. Each parent carried a copy of this mutation, supporting the autosomal recessive form of disease.
In this manner, different mutations in the gene LRBA are associated with the spectrum of phenotypes [150]. Even two siblings might present with incompatible pictures of LRBA deficiency [151].
Polymorphisms of the gene LRBA known to influence the function of LRBA protein have been associated with risk of pneumoconiosis in coal workers [152].
In conclusion, it is promising that respiratory manifestations of LRBA patients are mostly sensitive to therapy with IVIG, systemic immunosuppression, abatacept (CTLA4-Ig), or hematopoietic stem cell transplantation (HSCT) [147]. Drugs for prophylaxis of recurrent infections include antibiotics, antivirals, and antifungal medicines [146]. Mastoidectomy might be indicated in a severe case with frequent otitis media [146]. Therapeutic options for autoimmune complications vary from individual to individual and include IVIG, systemic immunosuppression, thymectomy, and HSCT [146]. However, respiratory failure remains the leading cause of death in these patients [85].
3.7 CD19 Complex Deficiencies
The CD19 complex is a transmembrane complex comprising CD19, CD21, CD81, and CD225. It can function as a co-receptor for B-cells that promotes B-cell receptor (BCR) signaling. Therefore, it is well-appreciated that defects in the formation of CD19 complex can cause autoimmunity and impairment of humoral immunity [153].
3.7.1 CD19 Deficiency
CD19 includes two extracellular immunoglobulin superfamily (IgSF) constant domains: a transmembrane region and a cytoplasmic region. Its cytoplasmic tail is comprised of nine tyrosine kinase residues that contribute to intracellular signaling. To our knowledge, there have been reported ten cases of CD19 deficiency [154,155,156,157,158,159,160]. The genetic defects responsible for CD19 deficiency include mutations resulting in premature termination codons and also missense mutations leading to an amino acid change. Overall, CD19 deficiency is featured by respiratory manifestations such as recurrent respiratory tract infections (pneumonia, bronchiolitis, and otitis media), wheezing, chronic obstructive pulmonary disease, atopy, and asthma [154]. Hypogammaglobulinemia (IgG and IgM) is also commonly found in patients with CD19 deficiency. Infectious pathogens found in these patients are respiratory syncytial virus, Haemophilus influenzae, and Streptococcus pneumoniae. CXR and CT scans reveal lobar atelectasis, peribronchial thickening, and bronchiectasis. Despite absence of CD19+ B-cells, flow cytometry can be used to confirm presence of CD20+ B-cells. Patients are also likely to have reduced numbers of transitional B-cells, memory B-cells, CD3+ T cells, and CD16+/CD56+ NK cells. Interestingly, studies indicate a delay between Ca2+ influx and stimulation with anti-IgM in B-cells from patients with CD19 deficiency. It should be noted that Ca2+ influx plays a crucial role in BCR signaling.
3.7.2 CD81 Deficiency
CD81 is a 26KDa surface protein also known as TAPA-1 (target of the antiproliferative antibody 1) and Tetraspanin-28 (Tspan-28). CD81 acts as regulator of proliferation of different cells such as glial cells, astrocytes, oocytes, and retinal pigment epithelial cells [161,162,163,164]. It is also required for the expression of CD19 on B-cells and thereby contributes to the activation of B-cells and production of antibodies against T-cell-dependent antigens. Its deficiency interferes with the formation of CD19 complex, resulting in the spectrum of respiratory manifestations related to CD19 deficiency.
The authors in the study [165] presented a patient suffering from recurrent respiratory tract infections during the first 2 years of life. She then developed glomerulonephritis, recurrent thrombocytopenia, and hypogammaglobulinemia of IgG. There was no significant change in the numbers of B, T, and NK cells. However, flow cytometry revealed a reduction in the relative number of transitional B-cells and memory B-cells. Additionally, the patient’s B-cells showed loss of both CD19 and CD81. Genetic analysis revealed an intact CD19 gene but homozygous splice site mutation (c.561+1G>A) in the CD81 gene. While the patient’s family members (her parents and her brother) harboring a single copy of this mutation were immunologically healthy, the patient did not show the expected increase in IgA and IgG levels upon vaccination with tetanus toxoid and pneumococcal antigens. CD81 is thus essential to produce an appropriate antibody response to protein and polysaccharide antigens.
A study of mice links CD9/CD81 double knockout to the spontaneous development of pulmonary emphysema [166]. It is accompanied with an increase in the activity of matrix metalloproteinases MMP-2 and MMP-9 in alveolar macrophages. The role of MMPs is well-appreciated in the degradation of the alveolar matrix as well as in the aggravation of pulmonary inflammatory processes, which both events have been implicated in COPD [167]. It is thus suggested that obstructive lung disease might arise from CD81 deficiency, owing to abnormal activity of MMPs.
Also, allergen-induced airway activity in CD81-deficient mice is decreased compared to wild-type mice [168]. CD81-deficient mice also demonstrated a reduced production of Th2 inflammatory cytokines IL-4, IL-5, and IL-13. It is therefore possible to assume that CD81 plays role in allergen-induced airway hyperactivity by inducing the expression of cytokines.
3.7.3 CD21 Deficiency
CD21 or complement receptor type 2 (CR2) precursor is a 145 KDa protein implicated in EBV entry [169]. It is expressed by mature B-cells and follicular dendritic cells, whereby activation of B-cell responses is enhanced by binding of immune complexes that comprise cleavage products of C3d and antigens [170]. This is beneficial in combating against toxic or infectious agents and on the other hand can cause an imbalance favoring autoimmunity.
The study [171] provides a report of first case of CD21 deficiency. Early childhood respiratory tract infections forced the patient to undergo tonsillectomy at 6 years old. Then, the patient experienced a long remission for about 20 years. At 28 years old, his symptoms including recurrent respiratory tract infections, myalgia, diarrhea, and weight loss began to flare up. The pathogen found in his respiratory secretions was Haemophilus influenzae. The patient also showed hypogammaglobulinemia of IgG1 and IgG4 and was thus considered for immunoglobulin replacement therapy. The patient’s B-cells completely lacked CD21. Genetic analysis revealed two heterozygous mutations: one resulting in the skipping of exon 6 and the other being a premature stop codon mutation occurring in exon 13.
The patient reported in [172] was a 13-year-old boy who presented with myalgia, rigidity, and hypogammaglobulinemia of IgG1, 2, and 4. Flow cytometry revealed absence of CD21. Sequencing of CD21 gene identified two heterozygous mutations: one nonsense mutation occurring in exon 2 and the other being a frameshift mutation in exon 15. Interestingly, the patient remained free of serious infections until writing the paper.
In the study [173], the authors have described two siblings who carried CD21 deficiency. Symptoms began to manifest at 5 and 7 years of age. Clinical manifestations mainly included recurrent otitis media, rhinopharyngitis, bronchitis, and lobar pneumonia.
3.8 Common Variable Immunodeficiency
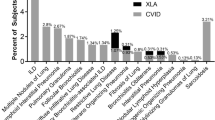
Common variable immunodeficiency (CVID) is the most common symptomatic PID estimated to occur in 1/10,000–1/200,000 depending on the study population. It is mainly characterized by a defective B-cell differentiation resulting in hypogammaglobulinemia of IgA, IgG, and IgM with two or more standard deviations below the normal mean. Also, about half of patients manifest characteristics of malfunctioning T-cells and cytokine/chemokine system including a reduction in CD40 ligand expression and IL-2 production [174]. Therefore, CVID covers a spectrum of disease phenotypes, and the diagnosis should only be made after ruling out other potential causes of hypogammaglobulinemia including other PIDs and immunodeficiency diseases due to infectious agents, malignancies, protein-losing agents, and drugs [175]. CVID involves a complex interaction of different disease processes. However, there are a few monogenetic forms of CVID that are caused by defect in a single gene such as ICOS [176, 177], TACI [178, 179], and CD19 [155]. Clinical presentations mainly include recurrent bacterial infections especially affecting upper and lower respiratory tracts, inflammatory diseases, autoimmune disorders, lymphoproliferative disorders, chronic lung diseases, gastrointestinal disorders, malabsorption, hepatitis, and malignancies (especially lymphomas). Clinical symptoms usually peak in the second or third decade of life. Early-onset disease is, however, common, especially in male patients [180]. Recently a multicenter study of 2212 patients with CVID has reported that the median diagnostic delay is around 4–5 years in most populations [181]. Overall, as much as all patients with CVID experience infectious respiratory diseases [182,183,184] and more than half of patients develop noninfectious respiratory complications as well [185].
3.8.1 Respiratory Tract Infections
The French national study of adults with CVID (n = 252) confirmed recurrent respiratory tract infections, e.g., bronchitis, sinusitis, and pneumonia, as the most common initial presentation [181]. In order they were found in about 69%, 63%, and 58% of patients. Lower respiratory tract infections are also found in 84% of patients [182]. In particular, pneumonia is experienced by about 32–77% of patients [180, 182, 184, 186]. Early-onset CVID in male (before 10 years of age) patients is considered a discrete disease entity characterized by a higher risk of infectious complications including pneumonia, but not noninfectious complications [180]. Encapsulated bacteria such as Streptococcus pneumoniae, Staphylococcus aureus, and Haemophilus influenzae are the main pathogens associated with respiratory tract infections in patients with CVID. In particular, Bordetella pertussis may contribute to the development of respiratory disease in children with CVID. About 13% of patients reveal a severe defect in switched memory B-cells (IgD-CD27+ cell percentage ≤2% of B-cells), which is related to an increased need for antibiotic therapy. Follow-up studies indicate that the incidence of acute pneumonia and otitis will decrease by time [186]. Other infectious respiratory manifestations that are possible but less frequent than the aforementioned ones include mycoplasma pneumonia, cryptococcal lung abscess, and Mycobacterium avium complex pulmonary disease [184].
3.8.2 Chronic Lung Disease
Of note, some patients remain refractory to immunoglobulin therapy and continue to suffer from upper respiratory tract infections [181]. These patients are prone to progress to chronic sinusitis and CLD including bronchiectasis. As expected, longitudinal studies show that the prevalence of chronic sinusitis and CLD will increase by time [186]. Studies estimate the prevalence of CLD to be between 22% and 67.5% in patients with CVID [182,183,184, 186]. In particular, bronchiectasis is found in 37–58% of patients with CVID [181, 182]. Patients who have a positive sputum culture are more likely to demonstrate evidence of bronchiectasis in HRCT [187]. In addition to the pathogens mostly involved in respiratory tract infections including Streptococcus pneumoniae and Haemophilus influenzae, bronchiectasis has been associated with Pseudomonas aeruginosa, Staphylococcus aureus, and Corynebacterium spp. [182]. A severe defect in total B-cells (CD19+ cell percentage ≤1%) was present in about 40% of patients and was associated with a higher risk of bronchiectasis. In addition, patients with CD4+ cell count below 700 cells/μL, patients with a history of pneumonia, and patients with older age were more prone to bronchiectasis [188].
3.8.3 Lymphocytic Interstitial Lung Disease and Follicular Bronchitis/Bronchiolitis
As reviewed in [189], both profiles of lymphocytic interstitial pneumonia (LIP) and follicular bronchitis/bronchiolitis have been detected in lung tissues from patients with CVID. LIP presents “with a diffuse, interstitial infiltrate of lymphocytes, immunoblasts, plasma cells, fibroblasts, and scattered macrophages that sometimes aggregate into granulomas” [189]. Follicular bronchitis/bronchiolitis is characterized by “reactive lymphoid follicles mainly around airways with minimal interstitial disease” [189]. While LIP has been associated with both hypogammaglobulinemia and hypergammaglobulinemia, follicular bronchitis/bronchiolitis does not appear to have been accompanied by any immunoglobulin abnormalities [189]. Clinical symptoms include cough, dyspnea, fever, and pleuritic chest pain.
The first case of LIP in CVID was described by Levinson et al. [190] and that the first case of nodular lymphoid interstitial pneumonia in CVID was documented by Kohler et al. [191]. Duke et al. also found two other cases with pulmonary infiltration in CXR, which were then histologically diagnosed with LIP [192].
3.8.4 Granulomatous Lung Disease
Generally, granulomatous lesions are found in 5.4–10% of patients with CVID [189]. Noninfectious diffuse lung complications which are collectively referred to granulomatous-lymphocytic interstitial lung disease (GLILD) predict worse survival in patients with CVID [185]. Patients with defects in T-cell function, low CD3+ cell count, and low CD8+ cell count are more likely to develop GLILD. The presence of GLILD has been linked to dyspnea, splenomegaly, restrictive pattern of pulmonary function (a low DLCO), consolidation, ground-glass opacity, interstitial infiltrates, and reticular pulmonary opacification. GLILD in CVID has been associated with a higher prevalence of HHV8 infection, leading to an increased risk of lymphoproliferative disorders [193]. It is promising that monoclonal antibodies and immunosuppressive agents can effectively assist with resolution of granulomas and restoration of lung structure [194]. FDG PET-CT imaging can be used to monitor therapeutic response [194].
3.8.5 Pulmonary Function
The majority of patients in spirometry are normal. About 25% of patients show abnormal features favoring an airflow obstruction and to a lesser extent a restrictive pattern [187]. When compared to patients without bronchiectasis, the spirometry test revealed reduced values of FEV1, FEF25–75%, and FVC in patients with bronchiectasis [182].
On the contrary, more than 60% of patients have abnormal MMEF. Therefore, there is a large subgroup of patients who are abnormal in MMEF and normal in spirometry [187].
3.8.6 Imaging Findings
HRCT in patients with CVID may provide evidence of granulomatous lung disease, hilar and mediastinal lymphadenopathy, pulmonary nodules, ground-glass opacity, bronchiectasis, and reduced peribronchiectatic shadowing [187, 188]. In particular, patients with bronchiectasis may demonstrate parenchymal scarring, pleural thickening, and atelectasis [182]. Bronchiectasis in patients with CVID has been shown to occur with the involvement of left lower lobe; lingual, right lower lobe; right middle lobe; right upper lobe; multiple lobes; and both lungs [182]. Patients with LIP demonstrate pulmonary infiltration especially basilar, coarse interstitial-alveolar infiltrations in CXR [189]. Patients with ground-glass appearance are more likely to have a high monocyte count, reduced number of CD19 + IgM-IgD-CD27+ isotype-switched memory B-cells, a history of lung disease, and pulmonary nodule(s) and be younger than those with bronchiectasis without interstitial lung disease [188]. Patients who exhibit evidence of bronchiectasis on their CT scan are more likely to have lower CD4+ T-cell counts [188]. Patients with five or more pulmonary nodules have lower CD8+ T-cell counts [188].
3.8.7 Morbidity and Mortality
Immunoglobulin dose required for prevention of bacterial infections is between 0.2 and 1.2 g/kg/months [195]. A higher dose of replacement immunoglobulin is, however, required to benefit cases with CLD including bronchiectasis [196]. Regular immunoglobulin therapy has shown to be effective in reducing the incidence of acute infections. A study of 50 patients with CVID with pneumonia demonstrated that the prevalence of pneumonia was reduced from 84% to 22% following immunoglobulin therapy [197]. Additionally, immunoglobulin therapy can effectively improve pulmonary function test results and HRCT scores in patients with CVID with CLD [196]. Such improvement does not appear to reach significance in cases with CVID without CLD [196]. Given that the mortality of CVID increases with age, it is not clear whether it can be effective in long term as well as short term. Lung transplant is the option available to patients with respiratory failure. Altogether, acute or chronic respiratory tract infections and associated respiratory failure are a leading cause of morbidity and mortality in patients with CVID [187, 186], especially in cases below 40 years of age [183].
3.9 CD20 Deficiency
It is a humoral immunodeficiency resulting from a homozygous mutation (MS4A1) in a splice junction of the CD20 gene, leading to the production of nonfunctional mRNA species [198]. Despite normal development of antigen-independent B-cells, CD20-deficient mice fail to generate normal antibody response to T-cell-independent antigens. Also, T-cell-dependent humoral immunity is impaired in CD20 deficiency [199]. This is thought to be a secondary effect of the impairment in B-cell function [199].
3.9.1 Potential Mechanisms of Respiratory Manifestations in CD20 Deficiency
CD20 acts as a regulator of B-cell development. It is thus well-understood that chronic administration of anti-CD20 antibodies can lead to B-cell depletion [200]. The effects that CD20 deficiency might have on lung tissues can be tackled in those who received anti-CD20 treatments. As reviewed by [201], anti-CD20 treatments predispose patients to severe and refractory respiratory infections with bacteria, fungi, and viruses, while, anti-CD20 antibodies are being increasingly used to treat autoimmune diseases and non-Hodgkin lymphoma [202]. This has been parallel by increasing the number of cases of pneumocystis pneumonia [203]. This increased susceptibility might be due to suppressive effect of anti-CD20 antibodies on type 2 helper T (Th2) responses, e.g., production of cytokines interleukin (IL)-4, IL-5, and IL-13 [202]. Moreover, there is a report of antisynthetase syndrome after treatment with rituximab, a CD20 monoclonal antibody [204]. The patient was presented with recurrent respiratory infections, arthropathy, and interstitial pneumonitis. Immunoglobulin replacement therapy was shown to improve immunodeficiency and pneumonitis. It is also interesting that treatment with 25-OH vitamin D3 has been shown to enhance CD20 levels [205].
3.10 Other Monogenic Defects Associated with Hypogammaglobulinemia (ICOS Deficiency, TACI Deficiency, BAFF Receptor Deficiency, TWEAK Deficiency, and NFKB2 Deficiency)
As above explained, CVID is not usually a single-gene disorder but rather is a complex disorder resulting from the combined effect of several genes. Overall, monogenic defects account for just 2–10% of cases with CVID [206].
3.10.1 ICOS Deficiency
The inducible T-cell co-stimulator (ICOS) is shown to stimulate differentiation of T follicular helper (TFH) cells and development of Treg, Th17, and Th2 cells that can promote autoimmunity and local inflammation [207]. In this manner, myeloid cells (CD11+ cells) expressing this co-stimulator can contribute to the generation of inflammatory processes in lung tissues [207]. What more supports this is increased expression of ICOS in autoimmune conditions (acute graft-versus-host disease) and allergic airway diseases [208]. Also, its ligand ICOS-L display by dendritic cells is capable to improve Treg or Th17 responses [209], whose pathogenic roles in autoimmunity and organ rejection have been well-described [210]. ICOS deficiency in mice could diminish lung tissue fibrosis [211] and delay rejection of the transplanted bronchus by reduction of relative numbers of Th17, Th2, and Treg cells [54]. By contrast, lung fibrosis was exacerbated in isolated ICOS-L deficiency and in double ICOS/ICOS-L deficiency [211].
Loss-of-function mutations in the ICOS gene as seen in patients with ICOS deficiency lead to reduce the numbers of TFH cells [212]. It has been demonstrated that intranasal application of Protollin, a TLR4 ligand adjuvant, can play a protective role against allergic lower airway disease upon stimulation with pollen allergen. This TLR4-dependent protection was accompanied by engagement of CD4+ T-cells expressing ICOS [213]. Additionally, adoptive transfer of CD4+ T-cells expressing ICOS could reverse allergen-induced airway hyper-responsiveness [213]. Whereas, inhibition of TLR4 signaling (TLR4-TRIF pathway) exacerbating allergen-induced airway hyper-responsiveness resulted in a reduction of CD4+ T-cells expressing ICOS [214]. Therefore, ICOS deficiency is expected to increase the risk of allergic airway diseases. As for B-cells expressing CD40, all CVID patients show diminished number of CD4+ T-cells expressing ICOS [215].
As above mentioned, ICOS is also implicated in development of Th2 immunity. Th2 cytokines (IL-5) are essential to the recruitment of eosinophils into the airway [216]. Therefore, it is expected that ICOS-deficient mice exhibit impairment in the generation of antigen-induced airway eosinophilia and inflammation [217]. It should be noted that CD28 also plays a crucial role in this context.
3.10.2 TACI Deficiency
Antiviral antibodies are essential to the generation of antiviral immunity. Animals deficient in transmembrane activator and calcium-modulator and cyclophilin ligand interactor (TACI) were not able to achieve protective titers of antibodies against influenza virus [218]. Even, mice that carried only a single copy of mutation in the TACI gene showed reduction of TACI expression on B-cells and were more susceptible to pneumococcal infections [219]. In this manner, TACI deficiency would predispose the lungs to viral and bacterial infections. TACI deficiency has been observed among patients with PADs who had hypogammaglobulinemia of IgG [220] and IgA [221]. It can cause phenotypes in the spectrum of clinical presentations correlated to CVID or even beyond them, for example, lymphoproliferation and IgG subclass deficiencies [222]. Later in life, it might lead to autoimmune disorders [221, 223].
3.10.3 NFKB2 Deficiency
Germline heterozygous mutations in NFKB2 are known to cause an early-onset type of CVID characterized by B-cell deficiency, T-cell deficiency, hypogammaglobulinemia, central adrenal insufficiency, alopecia totalis or areata, and trachyonychia [224,225,226,227,228,229,230]. They can contribute to changes in NK-cell count or function as well. Autoimmunity is present in the majority of patients with NFKB2 deficiency. Other presentations of this immunodeficiency include recurrent respiratory tract infections [228, 231].
Animal studies provide evidence that NFKB2 deficiency can cause serious lung inflammation by inducing the expression IFN-γ and thereby Th1 cytokines. Due to extensive infiltration of lymphocytes, this long inflammation is considered an autoimmune condition that can be potentially fatal [232, 233].
3.10.4 BAFF Receptor Deficiency
B-cell-activating factor of the TNF family (BAFF) emerged as an innate mediator that is involved in antiviral immunity. Upon exposure to viral dsRNA, Ig class switching is induced by TLR3-expressing B-cells of the upper respiratory tract. NFKB activation and TLR3 signaling are central to Ig class switching, which are in turn required for production of IgA and IgG antibodies in response to viral antigens [234]. Patients in intensive care unit (ICU) show increased susceptibility to hospital-acquired pneumonia. This is suggested to be caused by pulmonary IgA deficiency secondary to antibiotic therapy. Patients in this condition often reveal low expression of BAFF [235]. BAFF neutralization in mice has been associated with reduction in the number of antibody-secreting cells (ASC) that contribute to antiviral immunity [218]. Interestingly, immunoglobulin D has been shown to improve immunity in basophils of the upper respiratory tract. Its role is mediated by increasing the expression of pro-inflammatory in addition to B-cell stimulating factors, for example, BAFF [236].
On the other hand, BAFF acts to facilitate the cross talk between IL-1β and Th17 cell development, whereby Th1 and Th17 responses which mainly contribute to inflammatory and autoimmune processes are augmented [237]. Overexpression of BAFF is implicated in lung fibrosis [238], and therefore its inhibition is proposed as a targeted therapy for conditions associated with long fibrosis such as scleroderma [239].
3.10.5 TWEAK Deficiency
Tumor necrosis factor-like weak inducer of apoptosis (TWEAK) binding to fibroblast growth factor-inducible 14 (Fn14) leads to activation of intracellular signaling cascades that are likely to contribute to progression of a variety of tumors favorably non-small cell lung cancer (NSCLC) [240].
To the knowledge of the authors, there are not any respiratory manifestations documented as complications of other monogenic defects associated with hypogammaglobulinemia such as MOGS deficiency, TRNT1 deficiency, and TTC37 deficiency.
3.11 Immunoglobulin Class Switch Recombination Deficiencies Affecting B-Cells (AICDA Deficiency, UNG Deficiency, MMR Deficiency, and INO80 Deficiency)
3.11.1 AICDA Deficiency
The activation-induced cytidine deaminase (AICDA) gene encodes the enzyme activation-induced cytidine deaminase (AID) required to CSR in addition to Ig gene somatic hypermutation (SHM) [241]. More importantly, AID plays a crucial role in DNA methylation and reprogramming of somatic cells into induced pluripotent stem cells (iPSC) [242]. Besides the role of activated B-cell CD40 signaling, bacterial and viral antigens can lead to the expression of AID by engaging IL-4-secreting CD4+ T cells or TLR signaling [241].
Patients with AICDA deficiency often present with lymphoid hyperplasia [243]. Also, they frequently develop sinopulmonary infections (sinusitis and pneumonia), and finally their CT scan may reveal evidences of bronchiectasis [244]. Immunological findings include hypogammaglobulinemia of IgA, IgG, and IgE in addition to hypergammaglobulinemia of IgM [244].
The early clearance is an important component in engendering protective immunity against pneumococci [245]. Mice deficient in AID failed to effectively clear pneumococcal bacteria from pulmonary tissues at early time points [246]. The issue was resolved by adoptive transfer of AID+ B1a cells.
3.11.2 UNG Deficiency
The uracil-DNA glycosylase (UNG) gene encodes a base excision repair (BER) enzyme that functions to exclude misincorporated uracil. Cancerous cells deficient in UNG will develop hypersensitivity to pemetrexed (a chemotherapy drug). On the contrary, lung cancer cells carrying high expression of UNG reveal resistance to pemetrexed [247]. Moreover, it has been shown that viral UNG aids replication and dissemination of murine gammaherpesvirus 68 [248]. It seems that viral UNG expression is induced as a compensatory mechanism to increase UNG activity in host lung cells, which typically express low values of UNG [248]. As for AICDA deficiency, UNG deficiency is characterized with hypergammaglobulinemia of IgM along with hypogammaglobulinemia of other immunoglobulins. Also, lymphoid hyperplasia is commonly seen in patients with this immunodeficiency.
3.11.3 MMR Deficiency
DNA mismatch repair (MMR) is a mechanism of DNA repair conserved during evolution from bacteria to humans. It provides a compensatory pathway for correcting mismatched bases during DNA replication. Therefore, patients with MMR deficiency are prone to genomic instability and ultimately malignant transformation. MMR defects have been discovered in cancers of different primary sites such as the colon, rectum [249, 250], stomach [251], prostate, esophagus, endometrium, oral cavity [252], skin, head and neck, and brain [253]. Due to their role in class switch DNA recombination (CSR) and somatic hypermutation (SHM), MMR components are required for the regulation of antibody response as well [254]. Mice deficient in MMR components exhibit spontaneous development of premalignant and malignant lung lesions [255]. It is consistent with some observations of MMR defects in human lung cancers [256,257,258]. Moreover, defective mismatch repair system can contribute to chronic lung infection with Pseudomonas aeruginosa [259].
3.11.4 INO80 Deficiency
The INO80 ATPase takes part in formation of the ATP-dependent chromatin remodeling complex, which is involved in nuclear transactions, e.g., DNA replication, DNA repair, and transcription [260]. Energy-dependent metabolic pathways are accompanied by increased expression of the INO80 complex. While, defective INO80 complex correlates to decreased glycolysis, increased oxidative stress, and increased oxygen consumption. In this manner, the INO80 complex acts as a regulator of respiratory capacity [261]. On the other hand, INO80 is also generated as a nuclear protein to assist with genome replication of herpes simplex virus [262] and with oncogenic transcription and tumorigenesis of non-small cell lung cancer (NSCLC) [263]. However, study of NSCLC cells has recently revealed that the increased activity of chemotherapeutic agents (cisplatin) is underpinned by increased expression of more than 100 genes among which is INO80 [264].
Two patients with nonsynonymous, compound heterozygous single-nucleotide variants in INO80 have been described in [143]. The first one presented with recurrent bacterial infections at 5 years of age and the second with severe and recurrent respiratory infections at 18 years of age. The second also progressed to chronic obstructive pulmonary disease (of course with a history of 35 years of smoking).
3.12 Transient Hypogammaglobulinemia of Infancy
The physiologic hypogammaglobulinemia is what is typically happening in the first 3–6 months of life. Along with IgG subclass deficiency, partial antibody deficiency with impaired polysaccharide responsiveness, and selective IgA deficiency, transient hypogammaglobulinemia of infancy (THI) is one of the common immune-deficiency conditions of children [265], where the certain period of the physiologic hypogammaglobulinemia is extended. The way to diagnose THI is not straightforward; but its diagnosis requires a retrospective look of the patient characteristics: early hypogammaglobulinemia of at least one immunoglobulin isotype, later achieving normal levels for all immunoglobulin isotypes, and no diagnosis more likely than THI is suspected [266]. Overall, recurrent respiratory tract (pneumonia) infections, ENT (otitis media) infections, bronchitis, and asthma are the most common presentations of THI [267,268,269]. However, other infectious complications that may affect patients with THI include sinusitis, enteritis, lymphadenitis, meningitis, mastoiditis, impetigo [267], urinary tract infections, and sepsis [266]. Moreover, atopic reactions in THI are common and mainly include bronchial hyperreactivity, food allergy, and atopic dermatitis [266]. Study of a single center in Jordan estimated that the average age of onset and diagnosis for THI (n = 10) are 1 and 1.6 years [267]. So the diagnostic delay was less than 1 year [267]. In other studies from Turkey and the United States, the mean age of onset was less than 1 year of age [266, 270]. Often it is spontaneously resolved by 3–5 years of age [270], and more than 90% of patients achieve normal immunoglobulin levels by 10 years of age. However, some patients continue to have hypogammaglobulinemia, the so-called undefined/unclassified hypogammaglobulinemia. Of note is that both conditions, i.e., THI and undefined/unclassified hypogammaglobulinemia, share common clinical characteristics [268]. However, patients with persistent antibody deficiency are less able to produce IgA responses against pneumococcal polysaccharide (PnPS) vaccine compared to patients with transient antibody deficiency [271].
3.12.1 Respiratory Tract Infections
Infections affecting the upper and lower respiratory tract are commonly seen among patients with THI. Overall patients with THI have a benign clinical outcome [272]. They usually do not develop life-threatening infections [269] requiring long-term immunoglobulin replacement therapy [267]. Immediate treatment is, however, indicated in severe respiratory tract infections. Also, additional treatments may be warranted according to the type and severity of infection. For example, extracorporeal membrane oxygenation (ECMO), ribavirin, and steroid (albeit along with immunoglobulin replacement therapy) benefited a THI case with severe human parainfluenza viruses (HPIVs) [273]. Moreover there are reports of Pneumocystis carinii pneumonia in patients with THI [274,275,276]. It is a life-threatening infection [277], and despite high rates of drug adverse reactions, drug therapy should be started once the diagnosis of Pneumocystis carinii pneumonia is suspected.
3.12.2 Asthma
Studies estimate that about 27–55% of patients with THI suffer from asthma [268, 269]. The history of recurrent upper respiratory tract infections showed a negative association to asthma. Whereas, patients with THI who had history of recurrent lower respiratory tract infections were more likely to suffer from asthma [268].
3.12.3 Potential Mechanisms of Respiratory Manifestations in Transient Hypogammaglobulinemia of Infancy
Patients with THI are categorized into three according to their initial immunoglobulin levels: isolated IgG deficiency, isolated IgA deficiency, isolated IgM deficiency, IgG and IgA deficiency, IgG and IgM deficiency, and IgG, IgA, and IgM deficiency. The majority of patients show IgG and IgA deficiency [266], whereas isolated deficiency of IgA or IgM are relatively rare in patients with THI [269]. It has been well-appreciated that hypogammaglobulinemia either in primary immunodeficiency disorders or secondary to other conditions (malignancies and immunosuppressive drugs) increases the risk of infections.
It has been shown that antibody response to specific respiratory viral antigens (influenzas A and B, adenovirus, mycoplasma, respiratory syncytial virus, and parainfluenzas 1, 2, and 3) is more likely to be impaired in patients with THI than in age-matched controls without THI [270]. This is reflected in increased risk of viral infections in these patients.
3.13 Selective IgA Deficiency
Selective IgA deficiency (SIgAD) is the most common PID where B-cell switching to IgA-producing cells is impaired [278]. For children aged 1–18 years with recurrent infections and warning signs of immunodeficiency, it is the most commonly diagnosed disease [279]. Compared to other PIDs, patients with SIgAD appear to less prone to lower respiratory tract infection, sepsis, skin infections, mucocutaneous candidiasis, dental alterations, cardiovascular malformations, angioedema, hospitalizations, and death [280]. The world’s epidemiology data indicate that the incidence of SIgAD varies by a family history of SIgAD and ethnicity ranging from 1:18,500 in Japan to 1:143 in the Arabian Peninsula [278, 281]. Intrinsic factors causing IgA deficiency include both monogenic mutations (TNFRSF13B/TACI) [282, 283, 220] and chromosomal abnormalities mainly involving chromosome 18 [278, 1]. SIgAD is indicated by low levels of serum IgA (below 7 mg/dl) in presence of normal levels of IgG and IgM after the age of 4 years in whom other causes of hypogammaglobulinemia have not been identified. Clinical presentations may range from asymptomatic to clinically overt complications including recurrent sinopulmonary infections [284], autoimmune diseases [285], allergies, atopic disorders [286], gastrointestinal disorders, and malignancies [278]. Fortunately, about 65–90% of patients with SIgAD are asymptomatic [1, 287]. Only the minority of patients suffer from severe disease. They are those who may progress to CVID at later time points [278]. Follow-up study reveals that unlike patients with partial IgA deficiency and patients with partial IgA + IgG subclass deficiency, patients with selective IgA deficiency never can achieve normal levels of IgA during study [288]. This indicates the importance of periodic monitoring in these patients.
After IgG, IgA is the second most abundant immunoglobulin in the blood circulation [281]. It is secreted into the circulation and mucosal secretions. Two subclasses of IgA are present in humans: IgA1 and IgA2. Circulating IgA which is mainly comprised of IgA1 (80–90%) may help to regulate immune response via activation of the phagocyte system and inhibition of neutrophil chemotaxis. Mucosal secretions mainly include IgA2, which is more resistant to bacterial proteases present in the respiratory and gastrointestinal tract than IgA1. Secretory IgA contributes to the coating of mucosa-associated bacteria and therefore plays an important role to prevent penetration of bacteria into the mucosa. In addition, patients with SIgAD demonstrated altered cytokine/chemokine system in terms of increased levels of CXCL10/IP-10, IL-10, and G-CSF and decreased levels of IL-9 and IL-12 (p70) [289]. In this manner, SIgAD is regarded as a risk factor of recurrent respiratory infections and bronchial hyper-responsiveness [290].
About 40–90% of patients with symptomatic SIgAD report recurrent sinopulmonary infections as the most important manifestations leading to diagnosis of SIgAD. In particular, adults with SIgAD are more prone to upper respiratory tract infections (e.g., infectious conjunctivitis, common viral cold, pharyngitis, and laryngitis) and lower respiratory tract infections (e.g., bronchitis and pneumonia) [287]. Overall, the main pathogens involved are encapsulated bacteria including Haemophilus influenzae and Streptococcus pneumoniae. Bronchiectasis and obliterative bronchiolitis represent the most severe respiratory conditions experienced by patients with SIgAD. Studies demonstrate that deficiency of any of IgG subclasses is also present in about 20% of patients with SIgAD [291], rendering them more susceptible to sinopulmonary infections and respiratory insufficiency [292]. Moreover, patients with a low percentage of switched memory B-cells have a higher incidence of pneumonia and bronchiectasis compared to patients with a high percentage of switched memory B-cells [293]. On the contrary, an increased number of IgM-producing B-cells as a compensatory mechanism are commonly found in SIgAD. Because of the functional overlap between IgA and IgM, high levels of secretory IgM may protect patients against infections and help to maintain them asymptomatic [281]. When compared to controls without SIgAD, adults with SIgAD are more likely to be diagnosed with allergic rhinoconjunctivitis, but not asthma [287].
Rarely, SIgAD has been observed concurrent with other respiratory conditions including severe asthma, chronic granulomatous disease [294,295,296], chronic pulmonary aspergillosis [297], tracheobronchopathia osteochondroplastica [298], pulmonary nodules, disseminated cat-scratch disease [299], pleuropulmonary blastoma [300, 301], eosinophilic pneumonia [302], cryptococcal pneumonia [303], and idiopathic pulmonary hemosiderosis [304]. Also, SIgAD can mimic Churg-Strauss syndrome and hypereosinophilic syndrome [302].
As reviewed in [278], the management of patients with SIgAD includes education and periodic monitoring (every 4–6 months), treatment of allergic and autoimmune disorders, prophylactic antibiotics (especially for patients with chronic and recurrent sinopulmonary infections), pneumococcal vaccine (especially for patients with IgG subclass deficiency), and intravenous or subcutaneous immunoglobulin replacement therapy (especially for patients with recurrent infections). Immunoglobulin products low in IgA that can be administered to those patients include lyophilized Gammagard, Gammaplex, Vigam, Iveegam, Polygam, and Nanogam with IgA <10 mg/ml. Also lobectomy can be considered in cases of bronchiectasis [305]. Monoclonal antibodies such as rituximab (anti-CD20) might be effective in treating autoimmune disorders associated with SIgAD such as autoimmune thrombocytopenia [296].
References
Fried AJ, Bonilla FA. Pathogenesis, diagnosis, and management of primary antibody deficiencies and infections. Clin Microbiol Rev. 2009;22(3):396–414.
Maarschalk-Ellerbroek LJ, Hoepelman IM, Ellerbroek PM. Immunoglobulin treatment in primary antibody deficiency. Int J Antimicrob Agents. 2011;37(5):396–404.
Shillitoe B, Gennery A. X-linked agammaglobulinaemia: outcomes in the modern era. Clin Immunol (Orlando, Fla). 2017;183:54–62.
Jolles S, Sanchez-Ramon S, Quinti I, Soler-Palacin P, Agostini C, Florkin B, et al. Screening protocols to monitor respiratory status in primary immunodeficiency disease: findings from a European survey and subclinical infection working group. Clin Exp Immunol. 2017;190(2):226–34.
Hendriks RW, Bredius RGM, Pike-Overzet K, Staal FJT. Biology and novel treatment options for XLA, the most common monogenetic immunodeficiency in man. Expert Opin Ther Targets. 2011;15(8):1003–21.
Justiz Vaillant AA, Qurie A. Immunodeficiency. StatPearls. Treasure Island: StatPearls Publishing LLC; 2018.
Morales-Aza B, Glennie SJ, Garcez TP, Davenport V, Johnston SL, Williams NA, et al. Impaired maintenance of naturally acquired T-cell memory to the meningococcus in patients with B-cell immunodeficiency. Blood. 2009;113(18):4206–12.
Smith CIE, Berglof A. X-Linked Agammaglobulinemia. In: Adam MP, Ardinger HH, Pagon RA, Wallace SE, LJH B, Stephens K, et al., editors. GeneReviews((R)). Seattle: University of Washington, Seattle; 1993. GeneReviews is a registered trademark of the University of Washington, Seattle. All rights reserved.
Winkelstein JA, Marino MC, Lederman HM, Jones SM, Sullivan K, Burks AW, et al. X-linked agammaglobulinemia: report on a United States registry of 201 patients. Medicine. 2006;85(4):193–202.
Moin M, Aghamohammadi A, Farhoudi A, Pourpak Z, Rezaei N, Movahedi M, et al. X-linked agammaglobulinemia: a survey of 33 Iranian patients. Immunol Investig. 2004;33(1):81–93.
Buckley RH. Pulmonary complications of primary immunodeficiencies. Paediatr Respir Rev. 2004;5:S225–S33.
Ishida F, Kobayashi H, Saito H, Futatani T, Miyawaki T, Kiyosawa K. The oldest case with X-linked agammaglobulinemia in Japan lacking Bruton-type tyrosine kinase protein detected by flow cytometry. [Rinsho ketsueki]. Jpn J Clin Hematol. 1998;39(1):44–7.
Sweinberg SK, Wodell RA, Grodofsky MP, Greene JM, Conley ME. Retrospective analysis of the incidence of pulmonary disease in hypogammaglobulinemia. J Allergy Clin Immunol. 1991;88(1):96–104.
Quinti I, Soresina A, Guerra A, Rondelli R, Spadaro G, Agostini C, et al. Effectiveness of immunoglobulin replacement therapy on clinical outcome in patients with primary antibody deficiencies: results from a multicenter prospective cohort study. J Clin Immunol. 2011;31(3):315–22.
Chen Y, Stirling RG, Paul E, Hore-Lacy F, Thompson BR, Douglass JA. Longitudinal decline in lung function in patients with primary immunoglobulin deficiencies. J Allergy Clin Immunol. 2011;127(6):1414.
Basile N, Danielian S, Oleastro M, Rosenzweig S, Prieto E, Rossi J, et al. Clinical and molecular analysis of 49 patients with X-linked agammaglobulinemia from a single center in Argentina. J Clin Immunol. 2009;29(1):123–9.
He JX, Zhao SY, Jiang ZF. Clinical features of 17 cases of X-linked agammaglobulinemia. Zhongguo dang dai er ke za zhi= Chinese Journal of Contemporary Pediatrics. 2008;10(2):139–42.
Plebani A, Soresina A, Rondelli R, Amato GM, Azzari C, Cardinale F, et al. Clinical, immunological, and molecular analysis in a large cohort of patients with X-linked agammaglobulinemia: an Italian multicenter study. Clin Immunol. 2002;104(3):221–30.
Rip J, Van Der Ploeg EK, Hendriks RW, Corneth OBJ. The role of Bruton’s tyrosine kinase in immune cell signaling and systemic autoimmunity. Crit Rev Immunol. 2018;38(1):17–62.
Pal Singh S, Dammeijer F, Hendriks RW. Role of Bruton’s tyrosine kinase in B cells and malignancies. Mol Cancer. 2018;17(1):57.
Bao Y, Zheng J, Han C, Jin J, Han H, Liu Y, et al. Tyrosine kinase Btk is required for NK cell activation. J Biol Chem. 2012;287:23769–78. https://doi.org/10.1074/jbc.M112.372425.
Ochs HD, Davis SD, Wedgwood RJ. Immunologic responses to bacteriophage ϕX 174 in immunodeficiency diseases. J Clin Invest. 1971;50(12):2559–68.
Hendriks RW, Yuvaraj S, Kil LP. Targeting Bruton’s tyrosine kinase in B cell malignancies. Nat Rev Cancer. 2014;14(4):219–32.
Burger JA, Wiestner A. Targeting B cell receptor signalling in cancer: preclinical and clinical advances. Nat Rev Cancer. 2018;18(3):148–67.
Lee J, Rhee M, Min TK, Bang HI, Jang MA, Kang ES, et al. A novel BTK gene mutation, c.82delC (p.Arg28 Alafs(*)5), in a Korean family with X-linked agammaglobulinemia. Korean J Pediatr. 2016;59(Suppl 1):S49–s52.
Xu Y, Qing Q, Liu X, Chen S, Chen Z, Niu X, et al. Bruton’s agammaglobulinemia in an adult male due to a novel mutation: a case report. J Thorac Dis. 2016;8(10):E1207–e12.
Wang S, Lu Y, Li H, Wang Z, Mo X, Chai Z, et al. A novel deletion mutation and structural abnormality in the Bruton’s tyrosine kinase gene identified in a Chinese patient with X-linked agammaglobulinemia. Clin Lab. 2014;60(5):859–62.
Graziani S, Di Matteo G, Benini L, Di Cesare S, Chiriaco M, Chini L, et al. Identification of a Btk mutation in a dysgammaglobulinemic patient with reduced B cells: XLA diagnosis or not? Clin Immunol. 2008;128(3):322–8.
Aghamohammadi A, Fiorini M, Moin M, Parvaneh N, Teimourian S, Yeganeh M, et al. Clinical, immunological and molecular characteristics of 37 Iranian patients with X-linked agammaglobulinemia. Int Arch Allergy Immunol. 2006;141(4):408–14.
Jansen AG, Noordzij JG, Bröcker-Vriends AH, Bredius RG. Widely divergent clinical phenotype of X-linked agammaglobulinemia in two cousins. Ned Tijdschr Geneeskd. 2004;148(21):1053–6.
Abolhassani H, Vitali M, Lougaris V, Giliani S, Parvaneh N, Parvaneh L, et al. Cohort of Iranian patients with congenital agammaglobulinemia: mutation analysis and novel gene defects. Expert Rev Clin Immunol. 2016;12(4):479–86.
Lederman HM, Winkelstein JA. X-linked agammaglobulinemia: an analysis of 96 patients. Medicine. 1985;64(3):145–56.
Stubbs A, Bangs C, Shillitoe B, Edgar JD, Burns SO, Thomas M, et al. Bronchiectasis and deteriorating lung function in agammaglobulinaemia despite immunoglobulin replacement therapy. Clin Exp Immunol. 2018;191(2):212–9.
Quezada A, Norambuena X, Bravo A, Castro-Rodriguez JA. Recurrent pneumonia as warning manifestation for suspecting primary immunodeficiencies in children. J Investig Allergol Clin Immunol. 2001;11(4):295–9.
Usui K, Sasahara Y, Tazawa R, Hagiwara K, Tsukada S, Miyawaki T, et al. Recurrent pneumonia with mild hypogammaglobulinemia diagnosed as X-linked agammaglobulinemia in adults. Respir Res. 2001;2(3):188.
Schussler E, Beasley MB, Maglione PJ. Lung disease in primary antibody deficiencies. J Allergy Clin Immunol Pract. 2016;4(6):1039–52.
Quartier P, Debré M, De Blic J, de Sauverzac R, Sayegh N, Jabado N, et al. Early and prolonged intravenous immunoglobulin replacement therapy in childhood agammaglobulinemia: a retrospective survey of 31 patients. J Pediatr. 1999;134(5):589–96.
Bryan BA, Battersby A, Shillitoe BM, Barge D, Bourne H, Flood T, et al. Respiratory health and related quality of life in patients with congenital agammaglobulinemia in the northern region of the UK. J Clin Immunol. 2016;36(5):472–9.
Kainulainen L, Nikoskelainen J, Vuorinen T, Tevola K, Liippo K, Ruuskanen O. Viruses and bacteria in bronchial samples from patients with primary hypogammaglobulinemia. Am J Respir Crit Care Med. 1999;159(4):1199–204.
Nishi K, Kawai T, Kubota M, Ishiguro A, Onodera M. X-linked agammaglobulinemia complicated with pulmonary aspergillosis. Pediatr Int Off J Jpn Pediatr Soc. 2018;60(1):90–2.
Kawakami C, Inoue A, Takitani K, Kanegane H, Miyawaki T, Tamai H. X-linked agammaglobulinemia complicated with endobronchial tuberculosis. Acta Paediatr. 2011;100(3):466–8.
Dogru D, Kiper N, Ozcelik U, Yalcin E, Tezcan I. Tuberculosis in children with congenital immunodeficiency syndromes. Tuberkuloz ve toraks. 2010;58(1):59–63.
Alibrahim A, Lepore M, Lierl M, Filipovich A, Assa’ad A. Pneumocystis carinii pneumonia in an infant with X-linked agammaglobulinemia. J Allergy Clin Immunol. 1998;101(4):552–3.
Howard V, Greene JM, Pahwa S, Winkelstein JA, Boyle JM, Kocak M, et al. The health status and quality of life of adults with X-linked agammaglobulinemia. Clin Immunol. 2006;118(2–3):201–8.
Slade CA, Bosco JJ, Binh Giang T, Kruse E, Stirling RG, Cameron PU, et al. Delayed diagnosis and complications of predominantly antibody deficiencies in a cohort of Australian adults. Front Immunol. 2018;9:694.
Kainulainen L, Varpula M, Liippo K, Svedström E, Nikoskelainen J, Ruuskanen O. Pulmonary abnormalities in patients with primary hypogammaglobulinemia. J Allergy Clin Immunol. 1999;104(5):1031–6.
Funato M, Kaneko H, Ohkusu K, Sasai H, Kubota K, Ohnishi H, et al. Refractory chronic pleurisy caused by helicobacter equorum-like bacterium in a patient with X-linked agammaglobulinemia. J Clin Microbiol. 2011;49(9):3432–5.
Krupa A, Fol M, Rahman M, Stokes KY, Florence JM, Leskov IL, et al. Silencing Bruton’s tyrosine kinase in alveolar neutrophils protects mice from LPS/immune complex-induced acute lung injury. Am J Physiol Lung Cell Mol Physiol. 2014;307(6):L435–48.
Krupa A, Fudala R, Florence JM, Tucker T, Allen TC, Standiford TJ, et al. Bruton’s tyrosine kinase mediates FcgammaRIIa/toll-like receptor-4 receptor crosstalk in human neutrophils. Am J Respir Cell Mol Biol. 2013;48(2):240–9.
Verstak B, Nagpal K, Bottomley SP, Golenbock DT, Hertzog PJ, Mansell A. MyD88 adapter-like (Mal)/TIRAP interaction with TRAF6 is critical for TLR2- and TLR4-mediated NF-kappaB proinflammatory responses. J Biol Chem. 2009;284(36):24192–203.
Florence JM, Krupa A, Booshehri LM, Gajewski AL, Kurdowska AK. Disrupting the Btk pathway suppresses COPD-like lung alterations in Atherosclerosis Prone ApoE−/− mice following regular exposure to cigarette smoke. Int J Mol Sci. 2018;19(2):343.
Florence JM, Krupa A, Booshehri LM, Davis SA, Matthay MA, Kurdowska AK. Inhibiting Bruton’s tyrosine kinase rescues mice from lethal influenza-induced acute lung injury. Am J Physiol Lung Cell Mol Physiol. 2018;315(1):L52–l8.
Phillips JE, Renteria L, Burns L, Harris P, Peng R, Bauer CM, et al. Btk inhibitor RN983 delivered by dry powder nose-only aerosol inhalation inhibits bronchoconstriction and pulmonary inflammation in the ovalbumin allergic mouse model of asthma. J Aerosol Med Pulm Drug Deliv. 2016;29(3):233–41.
Zhou P, Ma B, Xu S, Zhang S, Tang H, Zhu S, et al. Knockdown of Burton’s tyrosine kinase confers potent protection against sepsis-induced acute lung injury. Cell Biochem Biophys. 2014;70(2):1265–75.
Grommes C, Younes A. Ibrutinib in PCNSL: the curious cases of clinical responses and aspergillosis. Cancer Cell. 2017;31(6):731–3.
Gu Y, Huang B, Yang Y, Qi M, Lu G, Xia D, et al. Ibrutinib exacerbates bleomycin-induced pulmonary fibrosis via promoting inflammation. Inflammation. 2018;41(3):904–13.
Arthurs B, Wunderle K, Hsu M, Kim S. Invasive aspergillosis related to ibrutinib therapy for chronic lymphocytic leukemia. Respir Med Case Rep. 2017;21:27–9.
Szymczak WA, Davis MJ, Lundy SK, Dufaud C, Olszewski M, Pirofski LA. X-linked immunodeficient mice exhibit enhanced susceptibility to cryptococcus neoformans Infection. MBio. 2013;4(4):e00265–13.
Kondratieva TK, Rubakova EI, Linge IA, Evstifeev VV, Majorov KB, Apt AS. B cells delay neutrophil migration toward the site of stimulus: tardiness critical for effective bacillus Calmette-Guerin vaccination against tuberculosis infection in mice. J Immunol. 2010;184(3):1227–34.
Marron TU, Rohr K, Martinez-Gallo M, Yu J, Cunningham-Rundles C. TLR signaling and effector functions are intact in XLA neutrophils. Clin Immunol. 2010;137(1):74–80.
Forman HJ, Torres M. Reactive oxygen species and cell signaling: respiratory burst in macrophage signaling. Am J Respir Crit Care Med. 2002;166(Supplement_1):S4–8.
Lundy SK, Berlin AA, Martens TF, Lukacs NW. Deficiency of regulatory B cells increases allergic airway inflammation. Inflamm Res. 2005;54(12):514–21.
Shabestari MS, Rezaei N. Asthma and allergic rhinitis in a patient with BTK deficiency. J Investig Allergol Clin Immunol. 2008;18(4):300–4.
Liese JG, Wintergerst U, Tympner KD, Belohradsky BH. High-vs low-dose immunoglobulin therapy in the long-term treatment of X-linked agammaglobulinemia. Am J Dis Child. 1992;146(3):335–9.
Suri D, Bhattad S, Sharma A, Gupta A, Rawat A, Sehgal S, et al. Serial serum immunoglobulin G (IgG) trough levels in patients with X-linked agammaglobulinemia on replacement therapy with intravenous immunoglobulin: its correlation with infections in Indian children. J Clin Immunol. 2017;37(3):311–8.
Morales P, Hernandez D, Vicente R, Sole A, Moreno I, Torres JJ, Lanuza A. Lung transplantation in patients with x-linked agammaglobulinemia. In: Transplantation proceedings. Elsevier; 2003. Vol. 35, No. 5, pp. 1942–43.
Barnes S, Kotecha S, Douglass JA, Paul E, Hore-Lacy F, Stirling R, et al. Evolving practice: X-linked agammaglobulinemia and lung transplantation. Am J Transpl Off J Am Soc Transpl Am Soc Transpl Surg. 2015;15(4):1110–3.
Conley ME, Rohrer J, Rapalus L, Boylin EC, Minegishi Y. Defects in early B-cell development: comparing the consequences of abnormalities in pre-BCR signaling in the human and the mouse. Immunol Rev. 2000;178:75–90.
Mårtensson I-L, Almqvist N, Grimsholm O, Bernardi AI. The pre-B cell receptor checkpoint. FEBS Lett. 2010;584(12):2572–9.
Yel L, Minegishi Y, Coustan-Smith E, Buckley RH, Trübel H, Pachman LM, et al. Mutations in the mu heavy-chain gene in patients with agammaglobulinemia. N Engl J Med. 1996;335(20):1486–93.
Granados EL, Porpiglia AS, Hogan MB, Matamoros N, Krasovec S, Pignata C, et al. Clinical and molecular analysis of patients with defects in μ heavy chain gene. J Clin Invest. 2002;110(7):1029–35.
Ferrari S, Zuntini R, Lougaris V, Soresina A, Sourkova V, Fiorini M, et al. Molecular analysis of the pre-BCR complex in a large cohort of patients affected by autosomal-recessive agammaglobulinemia. Genes Immun. 2007;8(4):325–33.
Mohammadzadeh I, Yeganeh M, Aghamohammadi A, Parvaneh N, Behniafard N, Abolhassani H, et al. Severe primary antibody deficiency due to a novel mutation of mu heavy chain. J Investig Allergol Clin Immunol. 2012;22(1):78–9.
Silva P, Justicia A, Regueiro A, Farina S, Couselo JM, Loidi L. Autosomal recessive agammaglobulinemia due to defect in mu heavy chain caused by a novel mutation in the IGHM gene. Genes Immun. 2017;18(3):197–9.
Schuh W, Meister S, Roth E, Jack HM. Cutting edge: signaling and cell surface expression of a mu H chain in the absence of lambda 5: a paradigm revisited. J Immunol. 2003;171(7):3343–7.
Karasuyama H, Rolink A, Shinkal Y, Young F, Alt FW, Melchers F. The expression of Vpre-B/λ5 surrogate light chain in early bone marrow precursor B cells of normal and B cell-deficient mutant mice. Cell. 1994;77(1):133–43.
Minegishi Y, Coustan-Smith E, Wang YH, Cooper MD, Campana D, Conley ME. Mutations in the human lambda5/14.1 gene result in B cell deficiency and agammaglobulinemia. J Exp Med. 1998;187(1):71–7.
Minegishi Y, Coustan-Smith E, Wang Y-H, Cooper MD, Campana D, Conley ME. Mutations in the human λ5/14.1 gene result in B cell deficiency and agammaglobulinemia. J Exp Med. 1998;187(1):71–7.
Wang Y, Kanegane H, Sanal O, Tezcan I, Ersoy F, Futatani T, et al. Novel Igalpha (CD79a) gene mutation in a Turkish patient with B cell-deficient agammaglobulinemia. Am J Med Genet. 2002;108(4):333–6.
Ferrari S, Lougaris V, Caraffi S, Zuntini R, Yang J, Soresina A, et al. Mutations of the Igβ gene cause agammaglobulinemia in man. J Exp Med. 2007;204(9):2047–51.
Meffre E, Nussenzweig MC. Deletion of immunoglobulin beta in developing B cells leads to cell death. Proc Natl Acad Sci U S A. 2002;99(17):11334–9.
Dobbs AK, Yang T, Farmer D, Kager L, Parolini O, Conley ME. Cutting edge: a hypomorphic mutation in Igβ (CD79b) in a patient with immunodeficiency and a leaky defect in B cell development. J Immunol. 2007;179(4):2055–9.
Lougaris V, Vitali M, Baronio M, Moratto D, Tampella G, Biasini A, et al. Autosomal recessive agammaglobulinemia: the third case of Igbeta deficiency due to a novel non-sense mutation. J Clin Immunol. 2014;34(4):425–7.
Pappu R, Cheng AM, Li B, Gong Q, Chiu C, Griffin N, et al. Requirement for B cell linker protein (BLNK) in B cell development. Science. 1999;286(5446):1949–54.
Jin G, Hamaguchi Y, Matsushita T, Hasegawa M, Le Huu D, Ishiura N, et al. B-cell linker protein expression contributes to controlling allergic and autoimmune diseases by mediating IL-10 production in regulatory B cells. J Allergy Clin Immunol. 2013;131(6):1674–82.
Minegishi Y, Rohrer J, Coustan-Smith E, Lederman HM, Pappu R, Campana D, et al. An essential role for BLNK in human B cell development. Science. 1999;286(5446):1954–7.
Van Zelm MC, Geertsema C, Nieuwenhuis N, De Ridder D, Conley ME, Schiff C, et al. Gross deletions involving IGHM, BTK, or Artemis: a model for genomic lesions mediated by transposable elements. Am J Hum Genet. 2008;82(2):320–32.
Lagresle-Peyrou C, Millili M, Luce S, Boned A, Sadek H, Rouiller J, et al. The BLNK adaptor protein has a nonredundant role in human B-cell differentiation. J Allergy Clin Immunol. 2014;134(1):145–54.
NaserEddin A, Shamriz O, Keller B, Alzyoud RM, Unger S, Fisch P, et al. Enteroviral infection in a patient with BLNK adaptor protein deficiency. J Clin Immunol. 2015;35(4):356–60.
Bain G, Maandag ECR, Izon DJ, Amsen D, Kruisbeek AM, Weintraub BC, et al. E2A proteins are required for proper B cell development and initiation of immunoglobulin gene rearrangements. Cell. 1994;79(5):885–92.
Zhuang Y, Soriano P, Weintraub H. The helix-loop-helix gene E2A is required for B cell formation. Cell. 1994;79(5):875–84.
Sun X-H. Constitutive expression of the Id1 gene impairs mouse B cell development. Cell. 1994;79(5):893–900.
Ben-Ali M, Yang J, Chan KW, Ben-Mustapha I, Mekki N, Benabdesselem C, et al. Homozygous transcription factor 3 gene <em>(TCF3)</em> mutation is associated with severe hypogammaglobulinemia and B-cell acute lymphoblastic leukemia. J Allergy Clin Immunol. 2017;140(4):1191–4.e4.
Boisson B, Wang Y-D, Bosompem A, Ma CS, Lim A, Kochetkov T, et al. A recurrent dominant negative E47 mutation causes agammaglobulinemia and BCR–B cells. J Clin Invest. 2013;123(11):4781–5.
Dobbs AK, Bosompem A, Coustan-Smith E, Tyerman G, Saulsbury FT, Conley ME. Agammaglobulinemia associated with BCR- B cells and enhanced expression of CD19. Blood. 2011;118(7):1828–37.
Sawada A, Takihara Y, Kim JY, Matsuda-Hashii Y, Tokimasa S, Fujisaki H, et al. A congenital mutation of the novel gene LRRC8 causes agammaglobulinemia in humans. J Clin Invest. 2003;112(11):1707–13.
Tarr PE, Sneller MC, Mechanic LJ, Economides A, Eger CM, Strober W, et al. Infections in patients with immunodeficiency with thymoma (Good syndrome). Report of 5 cases and review of the literature. Medicine. 2001;80(2):123–33.
Miyakis S, Pefanis A, Passam FH, Christodulakis GR, Roussou PA, Mountokalakis TD. Thymoma with immunodeficiency (Good’s syndrome): review of the literature apropos three cases. Scand J Infect Dis. 2006;38(4):314–20.
Miyakis S, Pefanis A, Passam FH, Christodulakis GR, Roussou PA, Mountokalakis TD. Thymoma with immunodeficiency (Good’s syndrome): review of the literature apropos three cases. Scand J Infect Dis. 2006;38(4):314–9.
Masci AM, Palmieri G, Vitiello L, Montella L, Perna F, Orlandi P, et al. Clonal expansion of CD8+ BV8 T lymphocytes in bone marrow characterizes thymoma-associated B lymphopenia. Blood. 2003;101(8):3106–8.
Leibovitz I, Zamir D, Polychuck I, Reitblat T, Gheorghiu D. Recurrent pneumonia post-thymectomy as a manifestation of Good syndrome. Eur J Intern Med. 2003;14(1):60–2.
Ohuchi M, Inoue S, Hanaoka J, Igarashi T, Tezuka N, Ozaki Y, et al. Good syndrome coexisting with leukopenia. Ann Thorac Surg. 2007;84(6):2095–7.
Ho JK, Wong MM, Tai TK, Tse DM. A rare combination of recurrent pneumonia, diarrhoea, and visual loss in a patient after thymectomy: good syndrome. Hong Kong medical journal = Xianggang yi xue za zhi. 2010;16(6):493–6.
Pena C, Intriago M, Munoz P, Gray AM, Cabrera ME. Association of Good syndrome with pure red cell aplasia: report of one case. Revista medica de Chile. 2012;140(8):1050–2.
Qu J, Lu X, Gao Q, Zhang Y. Good syndrome, a rare cause of refractory chronic diarrhea and recurrent pneumonia in a Chinese patient after thymectomy. Clin Vaccine Immunol CVI. 2013;20(7):1097–8.
Jeandel C, Gastin I, Blain H, Jouanny P, Laurain MC, Penin F, et al. Thymoma with immunodeficiency (Good’s syndrome) associated with selective cobalamin malabsorption and benign IgM-kappa gammopathy. J Intern Med. 1994;235(2):179–82.
Di Renzo M, Pasqui AL, Voltolini L, Gotti G, Pompella G, Auteri A. Myelodysplasia and Good syndrome. A case report. Clin Exp Med. 2008;8(3):171–3.
Jorge LP, Pereira CE, Jorge E. Cytomegalovirus retinitis in good syndrome. Revista Brasileira de Oftalmologia. 2018;77(3):153–5.
Numakura T, Matsuura Y, Takiguchi H, Hara Y, Ameku K. A Good syndrome associated with pure red cell aplasia. Nihon Kokyuki Gakkai zasshi = J Jpn Respir Soc. 2011;49(9):647–50.
Arnold SJ, Hodgson T, Misbah SA, Patel SY, Cooper SM, Venning VA. Three difficult cases: the challenge of autoimmunity, immunodeficiency and recurrent infections in patients with Good syndrome. Br J Dermatol. 2015;172(3):774–7.
Jansen A, van Deuren M, Miller J, Litzman J, de Gracia J, Saenz-Cuesta M, et al. Prognosis of Good syndrome: mortality and morbidity of thymoma associated immunodeficiency in perspective. Clin Immunol (Orlando, Fla). 2016;171:12–7.
Takai S, Tagawa A, Ogawa T, Kato H, Saito N, Okada S. Thymoma with immunodeficiency/good syndrome associated with myasthenia gravis. Rinsho shinkeigaku = Clin Neurol. 2017;57(5):208–13.
Chen X, Zhang JX, Shang WW, Xie WP, Jin SX, Wang F. Aberrant peripheral immune function in a good syndrome patient. J Immunol Res. 2018;2018:6212410.
Aouadi S, Ghrairi N, Braham E, Kaabi M, Maalej S, Elgharbi LD. Acquired hypogammaglobulinemia associated with thymoma: Good syndrome. Pan Afr Med J. 2017;28:253.
Makinson A, Corne P, Pelle C, Ben Hadj Salem M, Pouessel D, Landreau L, et al. Good syndrome and cytomegalovirus pneumonia. La Revue de medecine interne. 2003;24(5):330–1.
Jian L, Bin D, Haiyun W. Fatal pneumocystis pneumonia with good syndrome and pure red cell aplasia. Clin Infect Dis Off Publ Infect Dis Soc Am. 2004;39(11):1740–1.
Katsuki Y, Suzuki S, Takahashi Y, Satoh T, Nogawa S, Tanaka K, et al. A case of good syndrome accompanied by myasthenia gravis: immunological evaluations. Nihon Rinsho Men’eki Gakkai kaishi = Jpn J Clin Immunol. 2006;29(2):102–6.
Fernandez-Villar A, Garcia-Tejedor JL, Leiro Fernandez V, Botana Rial M, Mendez Garrido A, Rivo Vazquez E, et al. Tracheobronchial wall thickening secondary to herpesvirus infection in a patient with Goods syndrome. An Med Interna (Madrid, Spain : 1984). 2008;25(5):234–6.
Inomata T, Honda M, Murakami A. Atypical VZV retinitis in a patient with Good syndrome. Ocul Immunol Inflamm. 2018;26(2):194–8.
Fukushima A, Ichimura Y, Obata S, Kinoshita-Ise M, Fujio Y, Takeno M, et al. Thymoma-associated multi-organ autoimmunity: a case of graft-versus-host disease-like erythroderma complicated by good syndrome successfully treated by thymectomy. J Dermatol. 2017;44(7):830–5.
Thongngarm T, Boonyasiri A, Pradubpongsa P, Tesavibul N, Anekpuritanang T, Kreetapirom P, Sompornrattanaphan M. Features and outcomes of immunoglobulin therapy in patients with good syndrome at Thailand’s largest tertiary referral hospital. Asian Pac J Allergy Immunol. 2018 Jun 1.
Chijimatsu Y, Nakazato Y, Homma H, Mizuguchi K. A case report of Good syndrome complicated by diffuse panbronchiolitis. Nihon Kyobu Shikkan Gakkai Zasshi. 1982;20(7):803–8.
Tsuburai T, Ikehara K, Suzuki S, Shinohara T, Mishima W, Tagawa A, et al. Hypogammaglobulinemia associated with thymoma (Good syndrome) similar to diffuse panbronchiolitis. Nihon Kokyuki Gakkai zasshi = J Jpn Respir Soc. 2003;41(6):421–5.
Ogoshi T, Ishimoto H, Yatera K, Oda K, Akata K, Yamasaki K, et al. A case of good syndrome with pulmonary lesions similar to diffuse panbronchiolitis. Intern Med (Tokyo, Japan). 2012;51(9):1087–91.
Lee SH, Lee SM, Yang SC, Yoo CG, Kim YW, Shim YS, et al. A case of granulomatous lung disease in a patient with Good’s syndrome. Korean J Intern Med. 2008;23(4):219–22.
Maccari ME, Abolhassani H, Aghamohammadi A, Aiuti A, Aleinikova O, Bangs C, Baris S, Barzaghi F, Baxendale H, Buckland M, Burns SO. Disease evolution and response to rapamycin in activated phosphoinositide 3-kinase δ syndrome: the European Society for Immunodeficiencies-activated phosphoinositide 3-kinase δ syndrome registry. Front Immunol. 2018;9:543.
Angulo I, Vadas O, Garcon F, Banham-Hall E, Plagnol V, Leahy TR, et al. Phosphoinositide 3-kinase delta gene mutation predisposes to respiratory infection and airway damage. Science. 2013;342(6160):866–71.
Elkaim E, Neven B, Bruneau J, Mitsui-Sekinaka K, Stanislas A, Heurtier L, et al. Clinical and immunologic phenotype associated with activated phosphoinositide 3-kinase delta syndrome 2: a cohort study. J Allergy Clin Immunol. 2016;138(1):210–8.e9.
Stokes CA, Condliffe AM. Phosphoinositide 3-kinase delta (PI3Kdelta) in respiratory disease. Biochem Soc Trans. 2018;46(2):361–9.
Southworth T, Mason S, Bell A, Ramis I, Calbet M, Domenech A, et al. PI3K, p38 and JAK/STAT signalling in bronchial tissue from patients with asthma following allergen challenge. Biomark Res. 2018;6(14):018–0128.
Marwick JA, Adcock IM, Chung KF. Overcoming reduced glucocorticoid sensitivity in airway disease: molecular mechanisms and therapeutic approaches. Drugs. 2010;70(8):929–48.
Marwick JA, Caramori G, Casolari P, Mazzoni F, Kirkham PA, Adcock IM, et al. A role for phosphoinositol 3-kinase delta in the impairment of glucocorticoid responsiveness in patients with chronic obstructive pulmonary disease. J Allergy Clin Immunol. 2010;125(5):1146–53.
Xia M, Xu H, Dai W, Zhu C, Wu L, Yan S, et al. The role of HDAC2 in cigarette smoke-induced airway inflammation in a murine model of asthma and the effect of intervention with roxithromycin. J Asthma. 2018;55(4):337–44.
Kim SR, Lee KS, Park HS, Park SJ, Min KH, Moon H, et al. HIF-1alpha inhibition ameliorates an allergic airway disease via VEGF suppression in bronchial epithelium. Eur J Immunol. 2010;40(10):2858–69.
Lee KS, Jeong JS, Kim SR, Cho SH, Kolliputi N, Ko YH, et al. Phosphoinositide 3-kinase-delta regulates fungus-induced allergic lung inflammation through endoplasmic reticulum stress. Thorax. 2016;71(1):52–63.
Jeong JS, Lee KB, Kim SR, Kim DI, Park HJ, Lee HK, et al. Airway epithelial phosphoinositide 3-kinase-delta contributes to the modulation of fungi-induced innate immune response. Thorax. 2018;73(8):758–68.
Kan-o K, Matsumoto K, Asai-Tajiri Y, Fukuyama S, Hamano S, Seki N, et al. PI3K-delta mediates double-stranded RNA-induced upregulation of B7-H1 in BEAS-2B airway epithelial cells. Biochem Biophys Res Commun. 2013;435(2):195–201.
Dinavahi SS, Nyayapathy S, Perumal Y, Dharmarajan S, Viswanadha S. Combined inhibition of PDE4 and PI3Kdelta modulates the inflammatory component involved in the progression of chronic obstructive pulmonary disease. Drug Res. 2014;64(4):214–9.
Doukas J, Eide L, Stebbins K, Racanelli-Layton A, Dellamary L, Martin M, et al. Aerosolized phosphoinositide 3-kinase gamma/delta inhibitor TG100-115 [3-[2,4-diamino-6-(3-hydroxyphenyl)pteridin-7-yl]phenol] as a therapeutic candidate for asthma and chronic obstructive pulmonary disease. J Pharmacol Exp Ther. 2009;328(3):758–65.
Sun XJ, Li ZH, Zhang Y, Zhou G, Zhang JQ, Deng JM, et al. Combination of erythromycin and dexamethasone improves corticosteroid sensitivity induced by CSE through inhibiting PI3K-delta/Akt pathway and increasing GR expression. Am J Physiol Lung Cell Mol Physiol. 2015;309(2):8.
Lee KS, Park SJ, Kim SR, Min KH, Jin SM, Puri KD, et al. Phosphoinositide 3-kinase-delta inhibitor reduces vascular permeability in a murine model of asthma. J Allergy Clin Immunol. 2006;118(2):403–9.
To Y, Ito K, Kizawa Y, Failla M, Ito M, Kusama T, et al. Targeting phosphoinositide-3-kinase-delta with theophylline reverses corticosteroid insensitivity in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2010;182(7):897–904.
Kracker S, Di Virgilio M, Schwartzentruber J, Cuenin C, Forveille M, Deau MC, et al. An inherited immunoglobulin class-switch recombination deficiency associated with a defect in the INO80 chromatin remodeling complex. J Allergy Clin Immunol. 2015;135(4):998–1007.e6.
Wiestner A. The role of B-cell receptor inhibitors in the treatment of patients with chronic lymphocytic leukemia. Haematologica. 2015;100(12):1495–507.
Hammadi SA, Almarzooqi S, Abdul-Kader HM, Saraswathiamma D, Souid AK. The PI3Kdelta inhibitor idelalisib suppresses liver and lung cellular respiration. Int J Physiol Pathophysiol Pharmacol. 2015;7(3):115–25.
Azizi G, Abolhassani H, Mahdaviani SA, Chavoshzadeh Z, Eshghi P, Yazdani R, et al. Clinical, immunologic, molecular analyses and outcomes of iranian patients with LRBA deficiency: a longitudinal study. Pediatr Allergy Immunol. 2017;28(5):478–84.
Shamriz O, Shadur B, NaserEddin A, Zaidman I, Simanovsky N, Elpeleg O, et al. Respiratory manifestations in LPS-responsive beige-like anchor (LRBA) protein-deficient patients. Eur J Pediatr. 2018;177(8):1163–72.
Al Sukaiti N, AbdelRahman K, AlShekaili J, Al Oraimi S, Al Sinani A, Al Rahbi N, Cho V, Field M, Cook MC. Agammaglobulinaemia despite terminal B‐cell differentiation in a patient with a novel LRBA mutation. Clin Transl Immunology. 2017;6(5):e144.
Shokri S, Nabavi M, Hirschmugl T, Aghamohammadi A, Arshi S, Bemanian MH, et al. LPS-responsive beige-like anchor gene mutation associated with possible bronchiolitis obliterans organizing pneumonia associated with hypogammaglobulinemia and normal IgM phenotype and low number of B cells. Acta Med Iran. 2016;54(10):620–3.
Alkhairy OK, Abolhassani H, Rezaei N, Fang M, Andersen KK, Chavoshzadeh Z, et al. Spectrum of phenotypes associated with mutations in LRBA. J Clin Immunol. 2016;36(1):33–45.
Schreiner F, Plamper M, Dueker G, Schoenberger S, Gamez-Diaz L, Grimbacher B, et al. Infancy-onset T1DM, short stature, and severe immunodysregulation in two siblings with a homozygous LRBA mutation. J Clin Endocrinol Metab. 2016;101(3):898–904.
Liu Y, Yang J, Wu Q, Han R, Yan W, Yuan J, Ji X, Li Y, Yao W, Ni C. LRBA gene polymorphisms and risk of coal workers’ pneumoconiosis: a case–control study from China. Int J Environ Res Public Health. 2017;14(10):1138.
Tedder TF, Inaoki M, Sato S. The CD19–CD21 complex regulates signal transduction thresholds governing humoral immunity and autoimmunity. Immunity. 1997;6(2):107–18.
Skendros P, Rondeau S, Chateil JF, Bui S, Bocly V, Moreau JF, et al. Misdiagnosed CD19 deficiency leads to severe lung disease. Pediatr Allergy Immunol Off Publ Eur Soc Pediatr Allergy Immunol. 2014;25(6):603–6.
van Zelm MC, Reisli I, van der Burg M, Castaño D, van Noesel CJM, van Tol MJD, et al. An antibody-deficiency syndrome due to mutations in the CD19 gene. N Engl J Med. 2006;354(18):1901–12.
Kanegane H, Agematsu K, Futatani T, Sira MM, Suga K, Sekiguchi T, et al. Novel mutations in a Japanese patient with CD19 deficiency. Genes Immun. 2007;8(8):663.
Artac H, Reisli I, Kara R, Pico-Knijnenburg I, Adin-Cinar S, Pekcan S, et al. B-cell maturation and antibody responses in individuals carrying a mutated CD19 allele. Genes Immun. 2010;11(7):523.
Vince N, Boutboul D, Mouillot G, Just N, Peralta M, Casanova J-L, et al. Defects in the CD19 complex predispose to glomerulonephritis, as well as IgG1 subclass deficiency. J Allergy Clin Immunol. 2011;127(2):538.
van Zelm MC, Smet J, van der Burg M, Ferster A, Le PQ, Schandené L, et al. Antibody deficiency due to a missense mutation in CD19 demonstrates the importance of the conserved tryptophan 41 in immunoglobulin superfamily domain formation. Hum Mol Genet. 2011;20(9):1854–63.
van Zelm MC, Smet J, van der Burg M, Ferster A, Le PQ, Schandene L, et al. Antibody deficiency due to a missense mutation in CD19 demonstrates the importance of the conserved tryptophan 41 in immunoglobulin superfamily domain formation. Hum Mol Genet. 2011;20(9):1854–63.
Kelic S, Levy S, Suarez C, Weinstein DE. CD81 regulates neuron-induced astrocyte cell-cycle exit. Mol Cell Neurosci. 2001;17(3):551–60.
Geisert EE Jr, Williams RW, Geisert GR, Fan L, Asbury AM, Maecker HT, et al. Increased brain size and glial cell number in CD81-null mice. J Comp Neurol. 2002;453(1):22–32.
Rubinstein E, Ziyyat A, Prenant M, Wrobel E, Wolf JP, Levy S, et al. Reduced fertility of female mice lacking CD81. Dev Biol. 2006;290(2):351–8.
Song BK, Levy S, Geisert EE Jr. Increased density of retinal pigment epithelium in cd81−/− mice. J Cell Biochem. 2004;92(6):1160–70.
van Zelm MC, Smet J, Adams B, Mascart F, Schandené L, Janssen F, et al. CD81 gene defect in humans disrupts CD19 complex formation and leads to antibody deficiency. J Clin Invest. 2010;120(4):1265–74.
Takeda Y, He P, Tachibana I, Zhou B, Miyado K, Kaneko H, et al. Double deficiency of tetraspanins CD9 and CD81 alters cell motility and protease production of macrophages and causes chronic obstructive pulmonary disease-like phenotype in mice. J Biol Chem. 2008;283(38):26089–97.
Churg A, Zhou S, Wright JL. Matrix metalloproteinases in COPD. Eur Respir J. 2012;39(1):197.
Deng J, Yeung VP, Tsitoura D, DeKruyff RH, Umetsu DT, Levy S. Allergen-induced airway hyperreactivity is diminished in CD81-deficient mice. J Immunol. 2000;165(9):5054–61.
Roberts ML, Luxembourg AT, Cooper NR. Epstein-Barr virus binding to CD21, the virus receptor, activates resting B cells via an intracellular pathway that is linked to B cell infection. J Gen Virol. 1996;77(Pt 12):3077–85.
Del Nagro CJ, Kolla RV, Rickert RC. A critical role for complement C3d and the B cell coreceptor (CD19/CD21) complex in the initiation of inflammatory arthritis. J Immunol. 2005;175(8):5379–89.
Thiel J, Kimmig L, Salzer U, Grudzien M, Lebrecht D, Hagena T, et al. Genetic CD21 deficiency is associated with hypogammaglobulinemia. J Allergy Clin Immunol. 2012;129(3):801–10.e6.
Wentink MW, Lambeck AJ, van Zelm MC, Simons E, van Dongen JJ, IJspeert H, et al. CD21 and CD19 deficiency: two defects in the same complex leading to different disease modalities. Clin Immunol (Orlando, Fla). 2015;161(2):120–7.
Rosain J, Miot C, Lambert N, Rousselet MC, Pellier I, Picard C. CD21 deficiency in 2 siblings with recurrent respiratory infections and hypogammaglobulinemia. J Allergy Clin Immunol Pract. 2017;5(6):1765–7.
Farrington M, Grosmaire LS, Nonoyama S, Fischer SH, Hollenbaugh D, Ledbetter JA, et al. CD40 ligand expression is defective in a subset of patients with common variable immunodeficiency. Proc Natl Acad Sci. 1994;91(3):1099–103.
Sneller MC. Common variable immunodeficiency. Am J Med Sci. 2001;321(1):42–8.
Grimbacher B, Hutloff A, Schlesier M, Glocker E, Warnatz K, Dräger R, et al. Homozygous loss of ICOS is associated with adult-onset common variable immunodeficiency. Nat Immunol. 2003;4(3):261.
Salzer U, Maul-Pavicic A, Cunningham-Rundles C, Urschel S, Belohradsky BH, Litzman J, et al. ICOS deficiency in patients with common variable immunodeficiency. Clin Immunol. 2004;113(3):234–40.
Castigli E, Wilson SA, Garibyan L, Rachid R, Bonilla F, Schneider L, et al. TACI is mutant in common variable immunodeficiency and IgA deficiency. Nat Genet. 2005;37(8):829.
Salzer U, Chapel HM, Webster ADB, Pan-Hammarström Q, Schmitt-Graeff A, Schlesier M, et al. Mutations in TNFRSF13B encoding TACI are associated with common variable immunodeficiency in humans. Nat Genet. 2005;37(8):820.
Gathmann B, Mahlaoui N, Gérard L, Oksenhendler E, Warnatz K, Schulze I, et al. Clinical picture and treatment of 2212 patients with common variable immunodeficiency. J Allergy Clin Immunol. 2014;134(1):116–26.
Oksenhendler E, Gérard L, Fieschi C, Malphettes M, Mouillot G, Jaussaud R, et al. Infections in 252 patients with common variable immunodeficiency. Clin Infect Dis. 2008;46(10):1547–54.
Garcia MAM, De Rojas MDHF, Manzur MDN, Pamplona MPM, Torrero LC, Macian V, et al. Respiratory disorders in common variable immunodeficiency. Respir Med. 2001;95(3):191–5.
Cunningham-Rundles C. Clinical and immunologic analyses of 103 patients with common variable immunodeficiency. J Clin Immunol. 1989;9(1):22–33.
Cunningham-Rundles C. Common variable immunodeficiency. Curr Allergy Asthma Rep. 2001;1(5):421–9.
Bates CA, Ellison MC, Lynch DA, Cool CD, Brown KK, Routes JM. Granulomatous-lymphocytic lung disease shortens survival in common variable immunodeficiency. J Allergy Clin Immunol. 2004;114(2):415–21.
Quinti I, Soresina A, Spadaro G, Martino S, Donnanno S, Agostini C, et al. Long-term follow-up and outcome of a large cohort of patients with common variable immunodeficiency. J Clin Immunol. 2007;27(3):308–16.
Thickett KM, Kumararatne DS, Banerjee AK, Dudley R, Stableforth DE. Common variable immune deficiency: respiratory manifestations, pulmonary function and high-resolution CT scan findings. QJM. 2002;95(10):655–62.
Maglione PJ, Overbey JR, Radigan L, Bagiella E, Cunningham-Rundles C. Pulmonary radiologic findings in common variable immunodeficiency: clinical and immunological correlations. Ann Allergy Asthma Immunol. 2014;113(4):452–9.
Park JH, Levinson AI. Granulomatous-lymphocytic interstitial lung disease (GLILD) in common variable immunodeficiency (CVID). Clin Immunol. 2010;134(2):97–103.
Levinson AI, Hopewell PC, Stites DP, Spitler LE, Fudenberg HH. Coexistent lymphoid interstitial pneumonia, pernicious anemia, and agammaglobulinemia: comment on autoimmune pathogenesis. Arch Intern Med. 1976;136(2):213–6.
Kohler PF, Cook RD, Brown WR, Manguso RL. Common variable hypogammaglobulinemia with T-cell nodular lymphoid interstitial pneumonitis and B-cell nodular lymphoid hyperplasia: different lymphocyte populations with a similar response to prednisone therapy. J Allergy Clin Immunol. 1982;70(4):299–305.
Dukes RJ, Rosenow EC, Hermans PE. Pulmonary manifestations of hypogammaglobulinaemia. Thorax. 1978;33(5):603–7.
Wheat WH, Cool CD, Morimoto Y, Rai PR, Kirkpatrick CH, Lindenbaum BA, et al. Possible role of human herpesvirus 8 in the lymphoproliferative disorders in common variable immunodeficiency. J Exp Med. 2005;202(4):479–84.
Jolles S, Carne E, Brouns M, El-Shanawany T, Williams P, Marshall C, et al. FDG PET-CT imaging of therapeutic response in granulomatous lymphocytic interstitial lung disease (GLILD) in common variable immunodeficiency (CVID). Clin Exp Immunol. 2017;187(1):138–45.
Lucas M, Lee M, Lortan J, Lopez-Granados E, Misbah S, Chapel H. Infection outcomes in patients with common variable immunodeficiency disorders: relationship to immunoglobulin therapy over 22 years. J Allergy Clin Immunol. 2010;125(6):1354–60.
de Gracia J, Vendrell M, Álvarez A, Pallisa E, Rodrigo M-J, de la Rosa D, et al. Immunoglobulin therapy to control lung damage in patients with common variable immunodeficiency. Int Immunopharmacol. 2004;4(6):745–53.
Busse PJ, Razvi S, Cunningham-Rundles C. Efficacy of intravenous immunoglobulin in the prevention of pneumonia in patients with common variable immunodeficiency. J Allergy Clin Immunol. 2002;109(6):1001–4.
Kuijpers TW, Bende RJ, Baars PA, Grummels A, Derks IAM, Dolman KM, et al. CD20 deficiency in humans results in impaired T cell–independent antibody responses. J Clin Invest. 2010;120(1):214–22.
Morsy DED, Sanyal R, Zaiss AK, Deo R, Muruve DA, Deans JP. Reduced T-dependent humoral immunity in CD20-deficient mice. J Immunol. 2013;191:3112–8. https://doi.org/10.4049/jimmunol.1202098.
Breitling S, Hui Z, Zabini D, Hu Y, Hoffmann J, Goldenberg NM, et al. The mast cell-B cell axis in lung vascular remodeling and pulmonary hypertension. Am J Physiol Lung Cell Mol Physiol. 2017;312(5):L710–L21.
Alvarez B, Arcos J, Fernandez-Guerrero ML. Pulmonary infectious diseases in patients with primary immunodeficiency and those treated with biologic immunomodulating agents. Curr Opin Pulm Med. 2011;17(3):172–9.
Elsegeiny W, Eddens T, Chen K, Kolls JK. Anti-CD20 antibody therapy and susceptibility to pneumocystis pneumonia. Infect Immun. 2015;83(5):2043–52.
Sato M, Ito S, Ogura M, Kamei K, Miyairi I, Miyata I, et al. Atypical pneumocystis jiroveci pneumonia with multiple nodular granulomas after rituximab for refractory nephrotic syndrome. Pediatr Nephrol. 2013;28(1):145–9.
Cherin P, de Jaeger C, Crave JC, Delain JC, Tadmouri A, Amoura Z. Subcutaneous immunoglobulins for the treatment of a patient with antisynthetase syndrome and secondary chronic immunodeficiency after anti-CD20 treatment: a case report. J Med Case Rep. 2017;11(1):017–1211.
Dogan M, Erol M, Cesur Y, Yuca SA, Doğan Ş. The effect of 25-hydroxyvitamin D3 on the immune system. J Pediatr Endocrinol Metab. 2009;22(10):929–36.
Bogaert DJ, Dullaers M, Lambrecht BN, Vermaelen KY, De Baere E, Haerynck F. Genes associated with common variable immunodeficiency: one diagnosis to rule them all? J Med Genet. 2016;53(9):575–90.
Teichmann LL, Cullen JL, Kashgarian M, Dong C, Craft J, Shlomchik MJ. Local triggering of the ICOS coreceptor by CD11c(+) myeloid cells drives organ inflammation in lupus. Immunity. 2015;42(3):552–65.
Li Z, Lu H, Gu J, Liu J, Zhu Q, Lu Y, et al. Chitinase 3-Like-1-deficient splenocytes deteriorated the pathogenesis of acute graft-versus-host disease via regulating differentiation of Tfh cells. Inflammation. 2017;40(5):1576–88.
Gao X, Zhao L, Wang S, Yang J, Yang X. Enhanced inducible costimulator ligand (ICOS-L) expression on dendritic cells in interleukin-10 deficiency and its impact on T-cell subsets in respiratory tract infection. Mol Med. 2013;19:346–56.
Afzali B, Lombardi G, Lechler RI, Lord GM. The role of T helper 17 (Th17) and regulatory T cells (Treg) in human organ transplantation and autoimmune disease. Clin Exp Immunol. 2007;148(1):32–46.
Tanaka C, Fujimoto M, Hamaguchi Y, Sato S, Takehara K, Hasegawa M. Inducible costimulator ligand regulates bleomycin-induced lung and skin fibrosis in a mouse model independently of the inducible costimulator/inducible costimulator ligand pathway. Arthritis Rheum. 2010;62(6):1723–32.
Ma CS, Wong N, Rao G, Avery DT, Torpy J, Hambridge T, et al. Monogenic mutations differentially affect the quantity and quality of T follicular helper cells in patients with human primary immunodeficiencies. J Allergy Clin Immunol. 2015;136(4):993–1006.e1.
Shalaby KH, Jo T, Nakada E, Allard-Coutu A, Tsuchiya K, Hirota N, et al. ICOS-expressing CD4 T cells induced via TLR4 in the nasal mucosa are capable of inhibiting experimental allergic asthma. J Immunol (Baltimore, Md : 1950). 2012;189(6):2793–804.
Shalaby KH, Al Heialy S, Tsuchiya K, Farahnak S, McGovern TK, Risse PA, et al. The TLR4-TRIF pathway can protect against the development of experimental allergic asthma. Immunology. 2017;152(1):138–49.
Berron-Ruiz L, Lopez-Herrera G, Vargas-Hernandez A, Mogica-Martinez D, Garcia-Latorre E, Blancas-Galicia L, et al. Lymphocytes and B-cell abnormalities in patients with common variable immunodeficiency (CVID). Allergol Immunopathol. 2014;42(1):35–43.
Tamachi T, Watanabe N, Oya Y, Kagami S, Hirose K, Saito Y, et al. B and T lymphocyte attenuator inhibits antigen-induced eosinophil recruitment into the airways. Int Arch Allergy Immunol. 2007;143(Suppl 1):50–5.
Shilling RA, Clay BS, Tesciuba AG, Berry EL, Lu T, Moore TV, et al. CD28 and ICOS play complementary non-overlapping roles in the development of Th2 immunity in vivo. Cell Immunol. 2009;259(2):177–84.
Wolf AI, Mozdzanowska K, Quinn WJ 3rd, Metzgar M, Williams KL, Caton AJ, et al. Protective antiviral antibody responses in a mouse model of influenza virus infection require TACI. J Clin Invest. 2011;121(10):3954–64.
Jabara HH, Lee JJ, Janssen E, Ullas S, Liadaki K, Garibyan L, et al. Heterozygosity for transmembrane activator and calcium modulator ligand interactor A144E causes haploinsufficiency and pneumococcal susceptibility in mice. J Allergy Clin Immunol. 2017;139(4):1293–301.e4.
Speletas M, Mamara A, Papadopoulou-Alataki E, Iordanakis G, Liadaki K, Bardaka F, et al. TNFRSF13B/TACI alterations in Greek patients with antibody deficiencies. J Clin Immunol. 2011;31(4):550–9.
Ameratunga R, Koopmans W, Woon ST, Leung E, Lehnert K, Slade CA, et al. Epistatic interactions between mutations of TACI (TNFRSF13B) and TCF3 result in a severe primary immunodeficiency disorder and systemic lupus erythematosus. Clin Transl Immunol. 2017;6(10):e159.
Speletas M, Salzer U, Florou Z, Petinaki E, Daniil Z, Bardaka F, et al. Heterozygous alterations of TNFRSF13B/TACI in tonsillar hypertrophy and sarcoidosis. Clin Dev Immunol. 2013;2013:532437.
Barroeta Seijas AB, Graziani S, Cancrini C, Finocchi A, Ferrari S, Miniero R, et al. The impact of TACI mutations: from hypogammaglobulinemia in infancy to autoimmunity in adulthood. Int J Immunopathol Pharmacol. 2012;25(2):407–14.
Chen K, Coonrod EM, Kumanovics A, Franks ZF, Durtschi JD, Margraf RL, et al. Germline mutations in NFKB2 implicate the noncanonical NF-kappaB pathway in the pathogenesis of common variable immunodeficiency. Am J Hum Genet. 2013;93(5):812–24.
Lee CE, Fulcher DA, Whittle B, Chand R, Fewings N, Field M, et al. Autosomal-dominant B-cell deficiency with alopecia due to a mutation in NFKB2 that results in nonprocessable p100. Blood. 2014;124(19):2964–72.
Lindsley AW, Qian Y, Valencia CA, Shah K, Zhang K, Assa’ad A. Combined immune deficiency in a patient with a novel NFKB2 mutation. J Clin Immunol. 2014;34(8):910–5.
Brue T, Quentien MH, Khetchoumian K, Bensa M, Capo-Chichi JM, Delemer B, et al. Mutations in NFKB2 and potential genetic heterogeneity in patients with DAVID syndrome, having variable endocrine and immune deficiencies. BMC Med Genet. 2014;15:139.
Lougaris V, Tabellini G, Vitali M, Baronio M, Patrizi O, Tampella G, et al. Defective natural killer-cell cytotoxic activity in NFKB2-mutated CVID-like disease. J Allergy Clin Immunol. 2015;135(6):1641–3.
Liu Y, Hanson S, Gurugama P, Jones A, Clark B, Ibrahim MA. Novel NFKB2 mutation in early-onset CVID. J Clin Immunol. 2014;34(6):686–90.
Shi C, Wang F, Tong A, Zhang X-Q, Song H-M, Liu Z-Y, et al. NFKB2 mutation in common variable immunodeficiency and isolated adrenocorticotropic hormone deficiency: a case report and review of literature. Medicine. 2016;95(40):e5081.
Shi C, Wang F, Tong A, Zhang XQ, Song HM, Liu ZY, et al. NFKB2 mutation in common variable immunodeficiency and isolated adrenocorticotropic hormone deficiency: a case report and review of literature. Medicine. 2016;95(40):e5081.
Yang L, Cui H, Wang Z, Zhang B, Ding J, Liu L, et al. Loss of negative feedback control of nuclear factor-kappaB2 activity in lymphocytes leads to fatal lung inflammation. Am J Pathol. 2010;176(6):2646–57.
Low JT, Hughes P, Lin A, Siebenlist U, Jain R, Yaprianto K, et al. Impact of loss of NF-kappaB1, NF-kappaB2 or c-REL on SLE-like autoimmune disease and lymphadenopathy in Fas(lpr/lpr) mutant mice. Immunol Cell Biol. 2016;94(1):66–78.
Xu W, Santini PA, Matthews AJ, Chiu A, Plebani A, He B, et al. Viral double-stranded RNA triggers Ig class switching by activating upper respiratory mucosa B cells through an innate TLR3 pathway involving BAFF. J Immunol. 2008;181(1):276–87.
Lohmeyer J, Morty RE, Herold S. Antibiotic therapy-induced collateral damage: IgA takes center stage in pulmonary host defense. J Clin Invest. 2018;128(8):3234–6.
Chen K, Xu W, Wilson M, He B, Miller NW, Bengten E, et al. Immunoglobulin D enhances immune surveillance by activating antimicrobial, proinflammatory and B cell-stimulating programs in basophils. Nat Immunol. 2009;10(8):889–98.
Sutherland AP, Ng LG, Fletcher CA, Shum B, Newton RA, Grey ST, et al. BAFF augments certain Th1-associated inflammatory responses. J Immunol. 2005;174(9):5537–44.
Francois A, Gombault A, Villeret B, Alsaleh G, Fanny M, Gasse P, et al. B cell activating factor is central to bleomycin- and IL-17-mediated experimental pulmonary fibrosis. J Autoimmun. 2015;56:1–11.
Matsushita T, Kobayashi T, Mizumaki K, Kano M, Sawada T, Tennichi M, et al. BAFF inhibition attenuates fibrosis in scleroderma by modulating the regulatory and effector B cell balance. Sci Adv. 2018;4(7):eaas9944.
Whitsett TG, Cheng E, Inge L, Asrani K, Jameson NM, Hostetter G, et al. Elevated expression of Fn14 in non-small cell lung cancer correlates with activated EGFR and promotes tumor cell migration and invasion. Am J Pathol. 2012;181(1):111–20.
Xu Z, Pone EJ, Al-Qahtani A, Park SR, Zan H, Casali P. Regulation of aicda expression and AID activity: relevance to somatic hypermutation and class switch DNA recombination. Crit Rev Immunol. 2007;27(4):367–97.
Bhutani N, Brady JJ, Damian M, Sacco A, Corbel SY, Blau HM. Reprogramming towards pluripotency requires AID-dependent DNA demethylation. Nature. 2010;463(7284):1042–7.
Aghamohammadi A, Parvaneh N, Rezaei N, Moazzami K, Kashef S, Abolhassani H, et al. Clinical and laboratory findings in hyper-IgM syndrome with novel CD40L and AICDA mutations. J Clin Immunol. 2009;29(6):769–76.
Mahdaviani SA, Hirbod-Mobarakeh A, Wang N, Aghamohammadi A, Hammarstrom L, Masjedi MR, et al. Novel mutation of the activation-induced cytidine deaminase gene in a Tajik family: special review on hyper-immunoglobulin M syndrome. Expert Rev Clin Immunol. 2012;8(6):539–46.
Askenase PW, Bryniarski K, Paliwal V, Redegeld F, Groot Kormelink T, Kerfoot S, et al. A subset of AID-dependent B-1a cells initiates hypersensitivity and pneumococcal pneumonia resistance. Ann N Y Acad Sci. 2015;1362:200–14.
Yamamoto N, Kerfoot SM, Hutchinson AT, Dela Cruz CS, Nakazawa N, Szczepanik M, et al. Expression of activation-induced cytidine deaminase enhances the clearance of pneumococcal pneumonia: evidence of a subpopulation of protective anti-pneumococcal B1a cells. Immunology. 2016;147(1):97–113.
Weeks LD, Fu P, Gerson SL. Uracil-DNA glycosylase expression determines human lung cancer cell sensitivity to pemetrexed. Mol Cancer Ther. 2013;12(10):2248–60.
Minkah N, Macaluso M, Oldenburg DG, Paden CR, White DW, McBride KM, et al. Absence of the uracil DNA glycosylase of murine gammaherpesvirus 68 impairs replication and delays the establishment of latency in vivo. J Virol. 2015;89(6):3366–79.
Poulogiannis G, Frayling IM, Arends MJ. DNA mismatch repair deficiency in sporadic colorectal cancer and lynch syndrome. Histopathology. 2010;56(2):167–79.
Haraldsdottir S, Roth R, Pearlman R, Hampel H, Arnold CA, Frankel WL. Mismatch repair deficiency concordance between primary colorectal cancer and corresponding metastasis. Familial Cancer. 2016;15(2):253–60.
Giampieri R, Maccaroni E, Mandolesi A, Del Prete M, Andrikou K, Faloppi L, et al. Mismatch repair deficiency may affect clinical outcome through immune response activation in metastatic gastric cancer patients receiving first-line chemotherapy. Gastric Cancer Off J Int Gastric Cancer Assoc Jpn Gastric Cancer Assoc. 2017;20(1):156–63.
Spetsotaki KN, Tsiambas E, Stamatelopoulos A, Fotiades PP, Kastanioudakis I, Tomos P, et al. DNA mismatch repair deficiency in lung and oral cavity carcinomas: the role of histogenetic origin. J BUON Off J Balkan Union Oncol. 2017;22(3):606–9.
Bouffet E, Larouche V, Campbell BB, Merico D, de Borja R, Aronson M, et al. Immune checkpoint inhibition for hypermutant glioblastoma multiforme resulting from germline biallelic mismatch repair deficiency. J Clin Oncol Off J Am Soc Clin Oncol. 2016;34(19):2206–11.
Wu X, Tsai CY, Patam MB, Zan H, Chen JP, Lipkin SM, et al. A role for the MutL mismatch repair Mlh3 protein in immunoglobulin class switch DNA recombination and somatic hypermutation. J Immunol (Baltimore, Md : 1950). 2006;176(9):5426–37.
Downey CM, Jirik FR. DNA mismatch repair deficiency accelerates lung neoplasm development in K-ras(LA1/+) mice: a brief report. Cancer Med. 2015;4(6):897–902.
Takamochi K, Takahashi F, Suehara Y, Sato E, Kohsaka S, Hayashi T, et al. DNA mismatch repair deficiency in surgically resected lung adenocarcinoma: microsatellite instability analysis using the Promega panel. Lung Cancer (Amsterdam, Netherlands). 2017;110:26–31.
Chung C, Christianson M. Predictive and prognostic biomarkers with therapeutic targets in breast, colorectal, and non-small cell lung cancers: a systemic review of current development, evidence, and recommendation. J Oncol Pharm Pract Off Publ Int Soc Oncol Pharm Pract. 2014;20(1):11–28.
Warth A, Korner S, Penzel R, Muley T, Dienemann H, Schirmacher P, et al. Microsatellite instability in pulmonary adenocarcinomas: a comprehensive study of 480 cases. Virchows Archiv Int J Pathol. 2016;468(3):313–9.
Moyano AJ, Feliziani S, Di Rienzo JA, Smania AM. Simple sequence repeats together with mismatch repair deficiency can bias mutagenic pathways in Pseudomonas aeruginosa during chronic lung infection. PLoS One. 2013;8(11):e80514.
Conaway RC, Conaway JW. The INO80 chromatin remodeling complex in transcription, replication and repair. Trends Biochem Sci. 2009;34(2):71–7.
Yao W, King DA, Beckwith SL, Gowans GJ, Yen K, Zhou C, et al. The INO80 complex requires the Arp5-Ies6 subcomplex for chromatin remodeling and metabolic regulation. Mol Cell Biol. 2016;36(6):979–91.
Dembowski JA, DeLuca NA. Selective recruitment of nuclear factors to productively replicating herpes simplex virus genomes. PLoS Pathog. 2015;11(5):e1004939.
Zhang S, Zhou B, Wang L, Li P, Bennett BD, Snyder R, et al. INO80 is required for oncogenic transcription and tumor growth in non-small cell lung cancer. Oncogene. 2017;36(10):1430–9.
Leung AW, Hung SS, Backstrom I, Ricaurte D, Kwok B, Poon S, et al. Combined use of gene expression modeling and siRNA screening identifies genes and pathways which enhance the activity of cisplatin when added at no effect levels to non-small cell lung Cancer cells in vitro. PLoS One. 2016;11(3):e0150675.
Stiehm RE. The four most common pediatric immunodeficiencies. Adv Exp Med Biol. 2007;601:15–26.
Karaca NE, Aksu G, Gulez N, Yildiz B, Azarsiz E, Kutukculer N. New laboratory findings in Turkish patients with transient hypogammaglobulinemia of infancy. Iran J Allergy Asthma Immunol. 2010;9(4):237–43.
Habahbeh ZM, Abu-Shukair ME, Almutereen MA, Alzyoud RM, Wahadneh AM. Primary antibody deficiencies at queen Rania children Hospital in Jordan: single center experience. Iran J Immunol. 2014;11(1):49–58.
Keles S, Artac H, Kara R, Gokturk B, Ozen A, Reisli I. Transient hypogammaglobulinemia and unclassified hypogammaglobulinemia: ‘similarities and differences’. Pediatr Allergy Immunol. 2010;21(5):843–51.
Kilic SS, Tezcan I, Sanal O, Metin A, Ersoy F. Transient hypogammaglobulinemia of infancy: clinical and immunologic features of 40 new cases. Pediatr Int. 2000;42(6):647–50.
Cano F, Mayo DR, Ballow M. Absent specific viral antibodies in patients with transient hypogammaglobulinemia of infancy. J Allergy Clin Immunol. 1990;85(2):510–3.
Janssen WJ, Nierkens S, Sanders EA, Boes M, van Montfrans JM. Antigen-specific IgA titres after 23-valent pneumococcal vaccine indicate transient antibody deficiency disease in children. Vaccine. 2015;33(46):6320–6.
Kidon MI, Handzel ZT, Schwartz R, Altboum I, Stein M, Zan-Bar I. Symptomatic hypogammaglobulinemia in infancy and childhood – clinical outcome and in vitro immune responses. BMC Fam Pract. 2004;5(23):1471–2296.
Cotugno N, Manno EC, Stoppa F, Sinibaldi S, Saffirio C, D’Argenio P, et al. Severe parainfluenza pneumonia in a case of transient hypogammalobulinemia of infancy. BMJ Case Rep. 2013. pii: bcr2013009959.
Smart JM, Kemp AS, Armstrong DS. Pneumocystis carinii pneumonia in an infant with transient hypogammaglobulinaemia of infancy. Arch Dis Child. 2002;87(5):449–50.
Walzer PD, Schultz MG, Western KA, Robbins JB. Pneumocystis carinii pneumonia and primary immune deficiency diseases of infancy and childhood. J Pediatr. 1973;82(3):416–22.
Burke EC, Brown AL Jr, Weed LA. Pneumocystis carinii pneumonia: report of case in infant with hypogammaglo-bulinemia. Proc Staff Meet Mayo Clin. 1962;37:129–36.
Kramer RI, Cirone VC, Moore H. Interstitial pneumonia due to pneumocystis carinii, cytomegalic inclusion disease and hypogammaglobulinemia occurring simultaneously in an infant. Pediatrics. 1962;29:816–27.
Yazdani R, Azizi G, Abolhassani H, Aghamohammadi A. Selective IgA deficiency: epidemiology, pathogenesis, clinical phenotype, diagnosis, prognosis and management. Scand J Immunol. 2017;85(1):3–12.
Aldirmaz S, Yucel E, Kiykim A, Cokugras H, Akcakaya N, Camcioglu Y. Profile of the patients who present to immunology outpatient clinics because of frequent infections. Turk pediatri arsivi. 2014;49(3):210–6.
Lozano NA, Lozano A, Sasia LV, Saranz RJ, Agresta MF, del Pilar Bovina Martijena M, et al. Clinical comparison between patients with selective immunoglobulin A deficiency and other primary immunodeficiencies. Arch Argent Pediatr. 2015;113(2):141–5.
Yel L. Selective IgA deficiency. J Clin Immunol. 2010;30(1):10–6.
Karaca NE, Severcan EU, Guven B, Azarsiz E, Aksu G, Kutukculer N. TNFRSF13B/TACI alterations in Turkish patients with common variable immunodeficiency and IgA deficiency. Avicenna J Med Biotechnol. 2018;10(3):192–5.
Santos-Valente E, Reisli I, Artac H, Ott R, Sanal O, Boztug K. A novel mutation in the complement component 3 gene in a patient with selective IgA deficiency. J Clin Immunol. 2013;33(1):127–33.
Justiz Vaillant AAQA. Immunodeficiency. [Updated 2018 May 2]. In: StatPearls [Internet]. Treasure Island: StatPearls Publishing; 2018.
Singh K, Chang C, Gershwin ME. IgA deficiency and autoimmunity. Autoimmun Rev. 2014;13(2):163–77.
Azizi G, Bagheri Y, Tavakol M, Askarimoghaddam F, Porrostami K, Rafiemanesh H, et al. The clinical and immunological features of patients with primary antibody deficiencies. Endocr Metab Immune Disord Drug Targets. 2018;18(5):537–45.
Jorgensen GH, Gardulf A, Sigurdsson MI, Sigurdardottir ST, Thorsteinsdottir I, Gudmundsson S, et al. Clinical symptoms in adults with selective IgA deficiency: a case-control study. J Clin Immunol. 2013;33(4):742–7.
Kutukculer N, Karaca NE, Demircioglu O, Aksu G. Increases in serum immunoglobulins to age-related normal levels in children with IgA and/or IgG subclass deficiency. Pediatr Allergy Immunol Off Publ Eur Soc Pediatr Allergy Immunol. 2007;18(2):167–73.
Hel Z, Huijbregts RP, Xu J, Nechvatalova J, Vlkova M, Litzman J. Altered serum cytokine signature in common variable immunodeficiency. J Clin Immunol. 2014;34(8):971–8.
Papadopoulou A, Mermiri D, Taousani S, Triga M, Nicolaidou P, Priftis KN. Bronchial hyper-responsiveness in selective IgA deficiency. Pediatr Allergy Immunol Off Publ Eur Soc Pediatr Allergy Immunol. 2005;16(6):495–500.
Santaella ML, Peredo R, Disdier OM. IgA deficiency: clinical correlates with IgG subclass and mannan-binding lectin deficiencies. Puerto Rico Health Sci J. 2005;24(2):107–10.
Aghamohammadi A, Mohammadi J, Parvaneh N, Rezaei N, Moin M, Espanol T, et al. Progression of selective IgA deficiency to common variable immunodeficiency. Int Arch Allergy Immunol. 2008;147(2):87–92.
Aghamohammadi A, Abolhassani H, Biglari M, Abolmaali S, Moazzami K, Tabatabaeiyan M, et al. Analysis of switched memory B cells in patients with IgA deficiency. Int Arch Allergy Immunol. 2011;156(4):462–8.
Jindal AK, Rawat A, Suri D, Sharma M, Shandilya JK, Goel S, et al. Severe aspergillus pneumonia and pulmonary artery hypertension in a child with autosomal recessive chronic granulomatous disease and selective IgA deficiency. J Clin Immunol. 2017;37(4):333–5.
Patiroglu T, Gungor HE, Lazaroski S, Unal E. Chronic granulomatous disease with markedly elevated IgE levels mimicking hyperimmunoglobulin E syndrome. Acta Microbiol Immunol Hung. 2013;60(2):155–62.
Shamsian BS, Mansouri D, Pourpak Z, Rezaei N, Chavoshzadeh Z, Jadali F, et al. Autosomal recessive chronic granulomatous disease, IgA deficiency and refractory autoimmune thrombocytopenia responding to anti-CD20 monoclonal antibody. Iran J Allergy Asthma Immunol. 2008;7(3):181–4.
Ragesh R, Ray A, Mian A, Vyas S, Sharma SK. Cavitary lung lesions in a difficult-to-treat asthma patient. J Assoc Physicians India. 2016;64(4):73–6.
Dincer HE, Dunitz JM. Tracheobronchopathia osteochondroplastica and selective IgA deficiency. J Bronchol Interv Pulmonol. 2012;19(1):54–6.
Rohr A, Ash R, Vadaparampil J, Hill J, Wetzel L. Disseminated cat-scratch disease in an adult with selective IgA deficiency. Radiol Case Rep. 2016;11(2):54–7.
Stewart DR, Givens SS, Harris AK, Williams GM, Messinger YH, Schultz KA, et al. Comment on: DICER1-negative pleuropulmonary blastoma in a patient with selective IgA deficiency. Pediatr Blood Cancer. 2016;63(10):1869–70.
Chen Y, Jin M, Zhao W, Li S, Wang X, Cai S, et al. DICER1-negative pleuropulmonary blastoma in a patient with selective IgA deficiency. Pediatr Blood Cancer. 2016;63(4):757–8.
Takahashi N, Kondo T, Fukuta M, Takemoto A, Takami Y, Sato M, et al. Selective IgA deficiency mimicking Churg-Strauss syndrome and hypereosinophilic syndrome: a case report. Nagoya J Med Sci. 2013;75(1–2):139–46.
Hage JE, Wu J, Cunha BA. Cryptococcal pneumonia in a patient with presumptive sarcoidosis and selective immunoglobulin a deficiency. Heart Lung J Crit Care. 2012;41(4):398–400.
Erkocoglu M, Civelek E, Kocabas CN. Unusual presentation: concurrent IgA deficiency and idiopathic pulmonary hemosiderosis. Pediatr Pulmonol. 2016;51(10):E34–e6.
Dominguez O, Giner MT, Alsina L, Martin MA, Lozano J, Plaza AM. Clinical phenotypes associated with selective IgA deficiency: a review of 330 cases and a proposed follow-up protocol. An Pediatr (Barcelona, Spain: 2003). 2012;76(5):261–7.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Saghazadeh, A., Rezaei, N. (2019). Pulmonary Manifestations of Predominantly Antibody Deficiencies. In: Mahdaviani, S., Rezaei, N. (eds) Pulmonary Manifestations of Primary Immunodeficiency Diseases . Springer, Cham. https://doi.org/10.1007/978-3-030-00880-2_3
Download citation
DOI: https://doi.org/10.1007/978-3-030-00880-2_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-00879-6
Online ISBN: 978-3-030-00880-2
eBook Packages: MedicineMedicine (R0)