Abstract
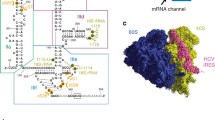
The internal ribosome entry site (IRES) in the 5′ untranslated region (UTR) of the hepatitis C virus (HCV) RNA genome is responsible for initiation of viral protein synthesis. The IRES RNA contains autonomously folding domains that are potential targets for antiviral translation inhibitors. Here, we describe the experimental crystal structure determination of the IRES subdomain IIa in complex with a previously discovered benzimidazole translation inhibitor. The structure of an inhibitor complex of the highly conserved IRES subdomain IIa holds promise for structure-based design of new anti-HCV drugs.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Davis DR, Seth PP (2011) Therapeutic targeting of HCV internal ribosomal entry site RNA. Antivir Chem Chemother 21:117–128
Kieft JS, Grech A, Adams P, Doudna JA (2001) Mechanisms of internal ribosome entry in translation initiation. Cold Spring Harb Symp Quant Biol 66:277–283
Lukavsky PJ, Kim I, Otto GA, Puglisi JD (2003) Structure of HCV IRES domain II determined by NMR. Nat Struct Biol 10:1033–1038
Dibrov SM, Johnston-Cox H, Weng YH, Hermann T (2007) Functional architecture of HCV IRES domain II stabilized by divalent metal ions in the crystal and in solution. Angew Chem Int Ed Engl 46:226–229
Seth PP, Miyaji A, Jefferson EA, Sannes-Lowery KA, Osgood SA, Propp SS, Ranken R, Massire C, Sampath R, Ecker DJ, Swayze EE, Griffey RH (2005) SAR by MS: discovery of a new class of RNA-binding small molecules for the hepatitis C virus: internal ribosome entry site IIA subdomain. J Med Chem 48:7099–7102
Parsons J, Castaldi MP, Dutta S, Dibrov SM, Wyles DL, Hermann T (2009) Conformational inhibition of the hepatitis C virus internal ribosome entry site RNA. Nat Chem Biol 5:823–825
Dibrov SM, Ding K, Brunn ND, Parker MA, Bergdahl BM, Wyles DL, Hermann T (2012) Structure of a hepatitis C virus RNA domain in complex with a translation inhibitor reveals a binding mode reminiscent of riboswitches. Proc Natl Acad Sci U S A 109:5223–5228
Parker MA, Satkiewicz E, Hermann T, Bergdahl BM (2011) An efficient new route to dihydropyranobenzimidazole inhibitors of HCV replication. Molecules 16:281–290
Otwinowski Z, Minor W (1997) Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol 276:307–326
Adams PD, Afonine PV, Bunkoczi G, Chen VB, Davis IW, Echols N, Headd JJ, Hung LW, Kapral GJ, Grosse-Kunstleve RW, McCoy AJ, Moriarty NW, Oeffner R, Read RJ, Richardson DC, Richardson JS, Terwilliger TC, Zwart PH (2010) PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Cryst D 66:213–221
Adams PD, Afonine PV, Bunkoczi G, Chen VB, Echols N, Headd JJ, Hung LW, Jain S, Kapral GJ, Grosse Kunstleve RW, McCoy AJ, Moriarty NW, Oeffner RD, Read RJ, Richardson DC, Richardson JS, Terwilliger TC, Zwart PH (2011) The Phenix software for automated determination of macromolecular structures. Methods 55:94–106
Collaborative Computational Project, Number 4 (1994) The CCP4 suite: programs for protein crystallography. Acta Cryst D 50:760–763
Murshudov GN, Vagin AA, Dodson EJ (1997) Refinement of macromolecular structures by the maximum-likelihood method. Acta Cryst D 53:240–255
Acknowledgement
This work was supported by the National Institutes of Health, grant AI072012.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Science+Business Media New York
About this protocol
Cite this protocol
Dibrov, S.M., Hermann, T. (2016). Structure of the HCV Internal Ribosome Entry Site Subdomain IIa RNA in Complex with a Viral Translation Inhibitor. In: Ennifar, E. (eds) Nucleic Acid Crystallography. Methods in Molecular Biology, vol 1320. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-2763-0_21
Download citation
DOI: https://doi.org/10.1007/978-1-4939-2763-0_21
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-2762-3
Online ISBN: 978-1-4939-2763-0
eBook Packages: Springer Protocols