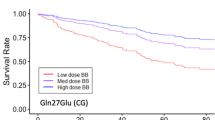
Abstract
The combination of angiotensin-converting enzyme (ACE) inhibitors and β-adrenergic receptor (βAR) blockers remains the essential component of heart failure (HF) pharmacotherapy. However, individual patient responses to these pharmacotherapies vary widely. The variability in response cannot be explained entirely by clinical characteristics, and genetic variation may play a role. The purpose of this chapter is to examine the current knowledge in the field of beta-blocker and ACE inhibitor pharmacogenetics in HF. β-blocker and ACE inhibitor pharmacogenetic studies performed in patients with HF were identified from the PubMed database from 1966 to July 2011. Thirty beta-blocker and 10 ACE inhibitor pharmacogenetic studies in patients with HF were identified.
The ACE deletion variant was associated with greater survival benefit from ACE inhibitors and beta-blockers compared with the ACE insertion. Ser49 in the β1AR, the insertion in the α2CAR, and Gln41 in G protein-coupled receptor (GPCR) kinase (GRK)-5 are associated with greater survival benefit from β-blockers, compared with Gly49, the deletion, and Leu41, respectively. However, many of these associations have not been validated. The HF pharmacogenetic literature is still in its very early stages, but there are promising candidate genetic variants that may identify which HF patients are most likely to benefit from beta-blockers and ACE inhibitors and patients that may require additional therapies.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Heart Failure Society of America (1999) Heart Failure Society of America (HFSA) practice guidelines. HFSA guidelines for management of patients with heart failure caused by left ventricular systolic dysfunction-pharmacological approaches. J Card Fail 5:357–382
Adams KF (2011) Development and implementation of heart failure practice guidelines. In: Mann DL (ed) Heart failure: a companion to Braunwald’s heart disease, 2nd edn. Saunders, Philadelphia, PA
Adams KF (2002) Beta-blockers in heart failure: how far have we progressed? Postgrad Med 112:7–15
MacFadyen RJ, Lee AF, Morton JJ et al (1999) How often are angiotensin II and aldosterone concentrations raised during chronic ACE inhibitor treatment in cardiac failure? Heart 82:57–61
Israili ZH, Hall WD (1992) Cough and angioneurotic edema associated with angiotensin-converting enzyme inhibitor therapy. A review of the literature and pathophysiology. Ann Intern Med 117:234–242
Metra M, Giubbini R, Nodari S et al (2000) Differential effects of beta-blockers in patients with heart failure: a prospective, randomized, double-blind comparison of the long-term effects of metoprolol versus carvedilol. Circulation 102:546–551
Lindenfeld J, Albert NM, Boehmer JP et al (2010) Heart Failure Society of America. HFSA 2010 comprehensive heart failure practice guideline. J Card Fail 16:e1–e194
Troncoso R, Moraga F, Chiong M et al (2009) Gln(27)/Glubeta(2)-adrenergic receptor polymorphism in heart failure patients: differential clinical and oxidative response to carvedilol. Basic Clin Pharmacol Toxicol 104:374–378
Cresci S, Kelly RJ, Cappola TP et al (2009) Clinical and genetic modifiers of long-term survival in heart failure. J Am Coll Cardiol 54:432–444
McNamara DM, Holubkov R, Janosko K et al (2001) Pharmacogenetic interactions between beta-blocker therapy and the angiotensin-converting enzyme deletion polymorphism in patients with congestive heart failure. Circulation 103:1644–1648
McNamara DM, Holubkov R, Postava L et al (2004) Pharmacogenetic interactions between angiotensin-converting enzyme inhibitor therapy and the angiotensin-converting enzyme deletion polymorphism in patients with congestive heart failure. J Am Coll Cardiol 44: 2019–2026
de Groote P, Helbecque N, Lamblin N et al (2004) Beta-adrenergic receptor blockade and the angiotensin-converting enzyme deletion polymorphism in patients with chronic heart failure. Eur J Heart Fail 6:17–21
Levin MC, Marullo S, Muntaner O (2002) The myocardium-protective Gly-49 variant of the beta 1-adrenergic receptor exhibits constitutive activity and increased desensitization and down-regulation. J Biol Chem 277:30429–30435
Rathz DA, Brown KM, Kramer LA et al (2002) Amino acid 49 polymorphisms of the human beta1-adrenergic receptor affect agonist-promoted trafficking. J Cardiovasc Pharmacol 39:155–160
Mann DL, Kent RL, Parsons B et al (1992) Adrenergic effects on the biology of the adult mammalian cardiocyte. Circulation 85:790–804
Terra SG, Hamilton KK, Pauly DF et al (2005) Beta1-adrenergic receptor polymorphisms and left ventricular remodeling changes in response to beta-blocker therapy. Pharmacogenet Genomics 15:227–234
de Groote P, Helbecque N, Lamblin N et al (2005) Association between beta-1 and beta-2 adrenergic receptor gene polymorphisms and the response to beta-blockade in patients with stable congestive heart failure. Pharmacogenet Genomics 15:137–142
Nonen S, Okamoto H, Fujio Y et al (2008) Polymorphisms of norepinephrine transporter and adrenergic receptor alpha1D are associated with the response to beta-blockers in dilated cardiomyopathy. Pharmacogenomics J 8:78–84
Chen L, Meyers D, Javorsky G et al (2007) Arg389Gly-beta1-adrenergic receptors determine improvement in left ventricular systolic function in non-ischemic cardiomyopathy patients with heart failure after chronic treatment with carvedilol. Pharmacogenet Genomics 17:941–949
Borjesson M, Magnusson Y, Hjalmarson A et al (2000) A novel polymorphism in the gene coding for the beta(1)-adrenergic receptor associated with survival in patients with heart failure. Eur Heart J 21:1853–1858
Ioannidis JP, Ntzani EE, Trikalinos TA et al (2001) Replication validity of genetic association studies. Nat Genet 29:306–309
Brodde OE, Michel MC (1999) Adrenergic and muscarinic receptors in the human heart. Pharmacol Rev 51:651–690
Mason DA, Moore JD, Green SA et al (1999) A gain-of-function polymorphism in a G-protein coupling domain of the human beta1-adrenergic receptor. J Biol Chem 274: 12670–12674
Mialet PJ, Rathz DA, Petrashevskaya NN et al (2003) Beta 1-adrenergic receptor polymorphisms confer differential function and predisposition to heart failure. Nat Med 9:1300–1305
Luo M, Bi Y, Xu YX (2007) Effects of metoprolol on beta1-adrenergic receptor polymorphism and receptor density in urban Chinese patients with heart failure. Chin Med J 120:1720–1723
Hu H, Jui HY, Hu FC et al (2007) Predictors of therapeutic response to beta-blockers in patients with heart failure in Taiwan. J Formos Med Assoc 106:641–648
Metra M, Covolo L, Pezzali N et al (2010) Role of beta-adrenergic receptor gene polymorphisms in the long-term effects of beta-blockade with carvedilol in patients with chronic heart failure. Cardiovasc Drugs Ther 24:49–60
Beta-Blocker Evaluation of Survival Trial Investigators (2001) A trial of the beta-blocker bucindolol in patients with advanced chronic heart failure. N Engl J Med 344:1659–1667
Liggett SB, Mialet-Perez J, Thaneemit-Chen S et al (2006) A polymorphism within a conserved beta(1)-adrenergic receptor motif alters cardiac function and beta-blocker response in human heart failure. Proc Natl Acad Sci U S A 103:11288–11293
MERIT-HF Investigators (1999) Effect of metoprolol CR/XL in chronic heart failure: metoprolol CR/XL randomised intervention trial in congestive heart failure (MERIT-HF). Lancet 353:2001–2007
Sehnert AJ, Daniels SE, Elashoff M et al (2008) Lack of association between adrenergic receptor genotypes and survival in heart failure patients treated with carvedilol or metoprolol. J Am Coll Cardiol 52:644–651
Biolo A, Clausell N, Santos KG et al (2008) Impact of beta1-adrenergic receptor polymorphisms on susceptibility to heart failure, arrhythmogenesis, prognosis, and response to beta-blocker therapy. Am J Cardiol 102:726–732
Bristow MR, Ginsburg R, Umans V et al (1986) Beta 1- and beta 2-adrenergic-receptor subpopulations in non-failing and failing human ventricular myocardium: coupling of both receptor subtypes to muscle contraction and selective beta1-receptor down-regulation in heart failure. Circ Res 59:297–309
Green SA, Turki J, Innis M et al (1994) Amino-terminal polymorphisms of the human beta 2-adrenergic receptor impart distinct agonist-promoted regulatory properties. Biochemistry 33:9414–9419
Terra SG, Pauly DF, Lee CR et al (2005) beta-Adrenergic receptor polymorphisms and responses during titration of metoprolol controlled release/extended release in heart failure. Clin Pharmacol Ther 77:127–137
Kaye DM, Smirk B, Williams C et al (2003) Beta-adrenoceptor genotype influences the response to carvedilol in patients with congestive heart failure. Pharmacogenetics 13:379–382
Green SA, Cole G, Jacinto M et al (1993) A polymorphism of the human beta2-adrenergic receptor within the fourth transmembrane domain alters ligand binding and functional properties of the receptor. J Biol Chem 268:23116–23121
Littlejohn MD, Palmer BR, Richards AM et al (2008) Ile164 variant of beta2-adrenoceptor does not influence outcome in heart failure but may interact with beta-blocker treatment. Eur J Heart Fail 10:55–59
Neumeister A, Charney DS, Belfer I et al (2005) Sympathoneural and adrenomedullary functional effects of alpha2C-adrenoreceptor gene polymorphism in healthy humans. Pharmacogenet Genomics 15:143–149
Lobmeyer MT, Gong Y, Terra SG et al (2007) Synergistic polymorphisms of beta1 and alpha2C-adrenergic receptors and the influence on left ventricular ejection fraction response to beta-blocker therapy in heart failure. Pharmacogenet Genomics 17:277–282
Bristow MR, Murphy GA, Krause-Steinrauf H et al (2010) An alpha2C-adrenergic receptor polymorphism alters the norepinephrine-lowering effects and therapeutic response of the beta-blocker bucindolol in chronic heart failure. Circ Heart Fail 3:21–28
Bristow MR, Krause-Steinrauf H, Nuzzo R et al (2004) Effect of baseline or changes in adrenergic activity on clinical outcomes in the beta-blocker evaluation of survival trial. Circulation 110:1437–1442
Liggett SB, Cresci S, Kelly RJ et al (2008) A GRK5 polymorphism that inhibits beta-adrenergic receptor signaling is protective in heart failure. Nat Med 14:510–517
Shekelle PG, Rich MW, Morton SC et al (2003) Efficacy of angiotensin-converting enzyme inhibitors and beta-blockers in the management of left ventricular systolic dysfunction according to race, gender, and diabetic status: a meta-analysis of major clinical trials. J Am Coll Cardiol 41:1529–1538
Rigat B, Hubert C, Alhenc-Gelas F et al (1990) An insertion/deletion polymorphism in the angiotensin I-converting enzyme gene accounting for half the variance of serum enzyme levels. J Clin Invest 86:1343–1346
Holmer SR, Hengstenberg C, Mayer B et al (2001) Marked suppression of renin levels by beta-receptor blocker in patients treated with standard heart failure therapy: a potential mechanism of benefit from beta-blockade. J Intern Med 249:167–172
Packer M (1992) The neurohormonal hypothesis: a theory to explain the mechanism of disease progression in heart failure. J Am Coll Cardiol 20:248–254
O’Toole L, Stewart M, Padfield P et al (1998) Effect of the insertion/deletion polymorphism of the angiotensin-converting enzyme gene on response to angiotensin-converting enzyme inhibitors in patients with heart failure. J Cardiovasc Pharmacol 32:988–994
Cicoira M, Zanolla L, Rossi A et al (2001) Failure of aldosterone suppression despite angiotensin-converting enzyme (ACE) inhibitor administration in chronic heart failure is associated with ACE DD genotype. J Am Coll Cardiol 37:1808–1812
Tiago AD, Badenhorst D, Skudicky D et al (2002) An aldosterone synthase gene variant is associated with improvement in left ventricular ejection fraction in dilated cardiomyopathy. Cardiovasc Res 54:584–589
Cuoco MA, Pereira AC, Mota Gde F et al (2008) Genetic polymorphism, medical therapy and sequential cardiac function in patients with heart failure. Arq Bras Cardiol 90:252–256
Wu CK, Luo JL, Tsai CT et al (2010) Demonstrating the pharmacogenetic effects of angiotensin-converting enzyme inhibitors on long-term prognosis of diastolic heart failure. Pharmacogenomics J 10:46–53
Acknowledgements
Dr. Lymperopoulos is supported by a Scientist Development Grant (SDG) award from the American Heart Association (09SDG2010138, National Center). No other disclosure to report.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this protocol
Cite this protocol
Lymperopoulos, A., French, F. (2014). Pharmacogenomics of Heart Failure. In: Yan, Q. (eds) Pharmacogenomics in Drug Discovery and Development. Methods in Molecular Biology, vol 1175. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-0956-8_10
Download citation
DOI: https://doi.org/10.1007/978-1-4939-0956-8_10
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-0955-1
Online ISBN: 978-1-4939-0956-8
eBook Packages: Springer Protocols