Abstract
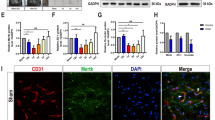
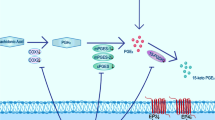
Hemoglobin (Hb) released from extravasated erythrocytes may have a critical role in the process of blood–brain barrier (BBB) disruption and subsequent edema formation after intracerebral hemorrhage (ICH). Excessive nitric oxide (NO) production synthesized by nitric oxide synthase (NOS) has been well documented to contribute to BBB disruption. However, considerably less attention has been focused on the role of NO in Hb-induced BBB disruption. This study was designed to examine the hypothesis that Hb-induced NOS overexpression and excessive NO production may contribute to the changes of tight junction (TJ) proteins and subsequent BBB dysfunction. Hemoglobin was infused with stereotactic guidance into the right caudate nucleus of male Sprague Dawley rats. Then, we investigated the effect of Hb on the BBB permeability, changes of TJ proteins (claudin-5, occludin, zonula occludens-1 (ZO-1), and junctional adhesion molecule-1 (JAM-1)), iron deposition, expression of inducible NOS (iNOS) and endothelial NOS (eNOS), as well as NO production. Hb injection caused a significant increase in BBB permeability. Significant reduction of claudin-5, ZO-1, and JAM-1 was observed after Hb injection as evidenced by PCR and immunofluorescence. After a decrease at early stage, occludin showed a fivefold increase in mRNA level at 7 days. Significant iron deposition was detectable from 48 h to 7 days in a time-dependent manner. The iNOS and eNOS levels dramatically increased after Hb injection concomitantly with large quantities of NO released. Furthermore, enhanced iNOS or eNOS immunoreactivity was co-localized with diffused or diminished claudin-5 staining. We concluded that overexpressed NOS and excessive NO production induced by Hb may contribute to BBB disruption, which may provide an important potential therapeutic target in the treatment of ICH.





Similar content being viewed by others
References
Abbott NJ, Patabendige AA, Dolman DE, Yusof SR, Begley DJ (2010) Structure and function of the blood–brain barrier. Neurobiol Dis 37:13–25
Balami JS, Buchan AM (2012) Complications of intracerebral haemorrhage. Lancet Neurol 11:101–118
Bao X, Wu G, Hu S, Huang F (2008) Poly(ADP-ribose) polymerase activation and brain edema formation by hemoglobin after intracerebral hemorrhage in rats. Acta Neurochir Suppl 105:23–27
Bazzoni G, Dejana E (2004) Endothelial cell-to-cell junctions: molecular organization and role in vascular homeostasis. Physiol Rev 84:869–901
Beauchesne E, Desjardins P, Hazell AS, Butterworth RF (2009) eNOS gene deletion restores blood–brain barrier integrity and attenuates neurodegeneration in the thiamine-deficient mouse brain. J Neurochem 111:452–459
Beckman JS, Koppenol WH (1996) Nitric oxide, superoxide, and peroxynitrite: the good, the bad, and ugly. Am J Physiol 271:C1424–C1437
Belayev L, Busto R, Zhao W, Ginsberg MD (1996) Quantitative evaluation of blood–brain barrier permeability following middle cerebral artery occlusion in rats. Brain Res 739:88–96
Benarroch EE (2011) Nitric oxide: a pleiotropic signal in the nervous system. Neurology 77:1568–1576
Bishop GM, Robinson SR (2001) Quantitative analysis of cell death and ferritin expression in response to cortical iron: implications for hypoxia–ischemia and stroke. Brain Res 907:175–187
Bove PF, Wesley UV, Greul AK, Hristova M, Dostmann WR, van der Vliet A (2007) Nitric oxide promotes airway epithelial wound repair through enhanced activation of MMP-9. Am J Respir Cell Mol Biol 36:138–146
Bulnes S, Argandona EG, Bengoetxea H, Leis O, Ortuzar N, Lafuente JV (2010) The role of eNOS in vascular permeability in ENU-induced gliomas. Acta Neurochir Suppl 106:277–282
Buster BL, Weintrob AC, Townsend GC, Scheld WM (1995) Potential role of nitric oxide in the pathophysiology of experimental bacterial meningitis in rats. Infect Immun 63:3835–3839
Butt OI, Buehler PW, D'Agnillo F (2011) Blood–brain barrier disruption and oxidative stress in guinea pig after systemic exposure to modified cell-free hemoglobin. Am J Pathol 178:1316–1328
Cera MR, Fabbri M, Molendini C, Corada M, Orsenigo F, Rehberg M, Reichel CA, Krombach F, Pardi R, Dejana E (2009) JAM-A promotes neutrophil chemotaxis by controlling integrin internalization and recycling. J Cell Sci 122:268–277
Dulak J, Jozkowicz A (2003) Regulation of vascular endothelial growth factor synthesis by nitric oxide: facts and controversies. Antioxid Redox Signal 5:123–132
Gebel JJ, Jauch EC, Brott TG, Khoury J, Sauerbeck L, Salisbury S, Spilker J, Tomsick TA, Duldner J, Broderick JP (2002) Relative edema volume is a predictor of outcome in patients with hyperacute spontaneous intracerebral hemorrhage. Stroke 33:2636–2641
Gonzalez-Gay MA, Garcia-Unzueta MT, Berja A, Vazquez-Rodriguez TR, Miranda-Filloy JA, Gonzalez-Juanatey C, de Matias JM, Martin J, Dessein PH, Llorca J (2009) Short-term effect of anti-TNF-alpha therapy on nitric oxide production in patients with severe rheumatoid arthritis. Clin Exp Rheumatol 27:452–458
Gursoy-Ozdemir Y, Bolay H, Saribas O, Dalkara T (2000) Role of endothelial nitric oxide generation and peroxynitrite formation in reperfusion injury after focal cerebral ischemia. Stroke 31(8):1974–1980; discussion 1981
Han YJ, Kwon YG, Chung HT, Lee SK, Simmons RL, Billiar TR, Kim YM (2001) Antioxidant enzymes suppress nitric oxide production through the inhibition of NF-kappa B activation: role of H(2)O(2) and nitric oxide in inducible nitric oxide synthase expression in macrophages. Nitric Oxide 5:504–513
Han F, Shirasaki Y, Fukunaga K (2006) Microsphere embolism-induced endothelial nitric oxide synthase expression mediates disruption of the blood–brain barrier in rat brain. J Neurochem 99:97–106
Huang FP, Xi G, Keep RF, Hua Y, Nemoianu A, Hoff JT (2002) Brain edema after experimental intracerebral hemorrhage: role of hemoglobin degradation products. J Neurosurg 96:287–293
Katsu M, Niizuma K, Yoshioka H, Okami N, Sakata H, Chan PH (2010) Hemoglobin-induced oxidative stress contributes to matrix metalloproteinase activation and blood–brain barrier dysfunction in vivo. J Cereb Blood Flow Metab 30:1939–1950
Keep RF, Xiang J, Ennis SR, Andjelkovic A, Hua Y, Xi G, Hoff JT (2008) Blood–brain barrier function in intracerebral hemorrhage. Acta Neurochir Suppl 105:73–77
Keita M, Vincendeau P, Buguet A, Cespuglio R, Vallat JM, Dumas M, Bouteille B (2000) Inducible nitric oxide synthase and nitrotyrosine in the central nervous system of mice chronically infected with Trypanosoma brucei brucei. Exp Parasitol 95:19–27
Kidd GA, Hong H, Majid A, Kaufman DI, Chen AF (2005) Inhibition of brain GTP cyclohydrolase I and tetrahydrobiopterin attenuates cerebral infarction via reducing inducible NO synthase and peroxynitrite in ischemic stroke. Stroke 36:2705–2711
Kim DW, Im SH, Kim JY, Kim DE, Oh GT, Jeong SW (2009) Decreased brain edema after collagenase-induced intracerebral hemorrhage in mice lacking the inducible nitric oxide synthase gene. Laboratory investigation. J Neurosurg 111:995–1000
Koeppen AH, Dickson AC, McEvoy JA (1995) The cellular reactions to experimental intracerebral hemorrhage. J Neurol Sci 134:102–112
Laird MD, Wakade C, Alleyne CJ, Dhandapani KM (2008) Hemin-induced necroptosis involves glutathione depletion in mouse astrocytes. Free Radic Biol Med 45:1103–1114
Li N, Liu YF, Ma L, Worthmann H, Wang YL, Wang YJ, Gao YP, Raab P, Dengler R, Weissenborn K, Zhao XQ (2013) Association of molecular markers with perihematomal edema and clinical outcome in intracerebral hemorrhage. Stroke 44(3):658–663
Mandi Y, Ocsovszki I, Szabo D, Nagy Z, Nelson J, Molnar J (1998) Nitric oxide production and MDR expression by human brain endothelial cells. Anticancer Res 18:3049–3052
Mayhan WG (1995) Role of nitric oxide in disruption of the blood–brain barrier during acute hypertension. Brain Res 686:99–103
Mayhan WG (2000) Nitric oxide donor-induced increase in permeability of the blood–brain barrier. Brain Res 866:101–108
McCarthy SM, Bove PF, Matthews DE, Akaike T, van der Vliet A (2008) Nitric oxide regulation of MMP-9 activation and its relationship to modifications of the cysteine switch. Biochemistry 47:5832–5840
Misra HP, Fridovich I (1972) The generation of superoxide radical during the autoxidation of hemoglobin. J Biol Chem 247:6960–6962
Nag S, Picard P, Stewart DJ (2000) Increased immunolocalization of nitric oxide synthases during blood–brain barrier breakdown and cerebral edema. Acta Neurochir Suppl 76:65–68
Parathath SR, Parathath S, Tsirka SE (2006) Nitric oxide mediates neurodegeneration and breakdown of the blood–brain barrier in tPA-dependent excitotoxic injury in mice. J Cell Sci 119:339–349
Park HY, Lee MH, Kang SU, Hwang HS, Park K, Choung YH, Kim CH (2012) Nitric oxide mediates TNF-alpha-induced apoptosis in the auditory cell line. Laryngoscope 122:2256–2264
Qing WG, Dong YQ, Ping TQ, Lai LG, Fang LD, Min HW, Xia L, Heng PY (2009) Brain edema after intracerebral hemorrhage in rats: the role of iron overload and aquaporin 4. J Neurosurg 110:462–468
Qureshi AI, Mendelow AD, Hanley DF (2009) Intracerebral haemorrhage. Lancet 373:1632–1644
Ryu JK, McLarnon JG (2006) Minocycline or iNOS inhibition block 3-nitrotyrosine increases and blood–brain barrier leakiness in amyloid beta-peptide-injected rat hippocampus. Exp Neurol 198:552–557
Sehba FA, Bederson JB (2011) Nitric oxide in early brain injury after subarachnoid hemorrhage. Acta Neurochir Suppl 110:99–103
Shukla A, Dikshit M, Srimal RC (1995) Nitric oxide modulates blood–brain barrier permeability during infections with an inactivated bacterium. Neuroreport 6:1629–1632
Song S, Hua Y, Keep RF, He Y, Wang J, Wu J, Xi G (2008) Deferoxamine reduces brain swelling in a rat model of hippocampal intracerebral hemorrhage. Acta Neurochir Suppl 105:13–18
Tan KH, Harrington S, Purcell WM, Hurst RD (2004) Peroxynitrite mediates nitric oxide-induced blood–brain barrier damage. Neurochem Res 29:579–587
Tsou HK, Su CM, Chen HT, Hsieh MH, Lin CJ, Lu DY, Tang CH, Chen YH (2010) Integrin-linked kinase is involved in TNF-alpha-induced inducible nitric-oxide synthase expression in myoblasts. J Cell Biochem 109:1244–1253
Won SM, Lee JH, Park UJ, Gwag J, Gwag BJ, Lee YB (2011) Iron mediates endothelial cell damage and blood–brain barrier opening in the hippocampus after transient forebrain ischemia in rats. Exp Mol Med 43:121–128
Xi G, Keep RF, Hoff JT (1998) Erythrocytes and delayed brain edema formation following intracerebral hemorrhage in rats. J Neurosurg 89:991–996
Xi G, Hua Y, Bhasin RR, Ennis SR, Keep RF, Hoff JT (2001) Mechanisms of edema formation after intracerebral hemorrhage: effects of extravasated red blood cells on blood flow and blood–brain barrier integrity. Stroke 32:2932–2938
Xu J, Lan D, Li T, Yang G, Liu L (2012) Angiopoietins regulate vascular reactivity after haemorrhagic shock in rats through the Tie2-nitric oxide pathway. Cardiovasc Res 96:308–319
Yoon JS, Lee JH, Tweedie D, Mughal MR, Chigurupati S, Greig NH, Mattson MP (2013) 3,6'-dithiothalidomide improves experimental stroke outcome by suppressing neuroinflammation. J Neurosci Res. doi:10.1002/jnr.23190
Zacharek A, Chen J, Zhang C, Cui X, Roberts C, Jiang H, Teng H, Chopp M (2006) Nitric oxide regulates Angiopoietin1/Tie2 expression after stroke. Neurosci Lett 404:28–32
Acknowledgments
This study was supported by the National Natural Science Foundation of China (no. 81271314), Natural Science Foundation of Guangdong (no. 5300468), and Special Project on the Integration of Industry, Education and Research of Guangdong Province and Ministry of Education (no. 2012B091100154).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, S., Chen, Y., Deng, X. et al. Hemoglobin-Induced Nitric Oxide Synthase Overexpression and Nitric Oxide Production Contribute to Blood–Brain Barrier Disruption in the Rat. J Mol Neurosci 51, 352–363 (2013). https://doi.org/10.1007/s12031-013-9990-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-013-9990-y