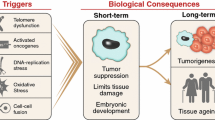
Abstract
Cellular senescence was first reported in human fibroblasts as a state of stable in vitro growth arrest following extended culture. Since that initial observation, a variety of other phenotypic characteristics have been shown to co-associate with irreversible cell cycle exit in senescent fibroblasts. These include (1) a pro-inflammatory secretory response, (2) the up-regulation of immune ligands, (3) altered responses to apoptotic stimuli and (4) promiscuous gene expression (stochastic activation of genes possibly as a result of chromatin remodeling). Many features associated with senescent fibroblasts appear to promote conversion to an immunogenic phenotype that facilitates self-elimination by the immune system. Pro-inflammatory cytokines can attract and activate immune cells, the presentation of membrane bound immune ligands allows for specific recognition and promiscuous gene expression may function to generate an array of tissue restricted proteins that could subsequently be processed into peptides for presentation via MHC molecules. However, the phenotypes of senescent cells from different tissues and species are often assumed to be broadly similar to those seen in senescent human fibroblasts, but the data show a more complex picture in which the growth arrest mechanism, tissue of origin and species can all radically modulate this basic pattern. Furthermore, well-established triggers of cell senescence are often associated with a DNA damage response (DDR), but this may not be a universal feature of senescent cells. As such, we discuss the role of DNA damage in regulating an immunogenic response in senescent cells, in addition to discussing less established “atypical” senescent states that may occur independent of DNA damage.

Similar content being viewed by others
References
Acosta JC, O’Loghlen A, Banito A, Guijarro MV, Augert A, Raguz S, Fumagalli M, Da Costa M, Brown C, Popov N, Takatsu Y, Melamed J, d’Adda diFagagna F, Bernard D, Hernando E, Gil J (2008) Chemokine signaling via the CXCR2 receptor reinforces senescence. Cell 133(6):1006–1018
Acosta JC, Banito A, Wuestefeld T, Georgilis A, Janich P, Morton JP, Athineos D, Kang TW, Lasitschka F, Andrulis M, Pascual G, Morris KJ, Khan S, Jin H, Dharmalingam G, Snijders AP, Carroll T, Capper D, Pritchard C, Inman GJ, Longerich T, Sansom OJ, Benitah SA, Zender L, Gil J (2013) A complex secretory program orchestrated by the inflammasome controls paracrine senescence. Nat Cell Biol 15(8):978–90
Ahmed EK, Rogowska-Wrzesinska A, Roepstorff P, Bulteau AL, Friguet B (2010) Protein modification and replicative senescence of WI-38 human embryonic fibroblasts. Aging Cell 9(2):252–72, 2010 Apr
Bahar R, Hartmann CH, Rodriguez KA, Denny AD, Busuttil RA, Dollé ME, Calder RB, Chisholm GB, Pollock BH, Klein CA, Vijg J (2006) Increased cell-to-cell variation in gene expression in ageing mouse heart. Nature 441(7096):1011–4, 2006 Jun 22
Benhamed M, Herbig U, Ye T, Dejean A, Bischof O (2012) Senescence is an endogenous trigger for microRNA-directed transcriptional gene silencing in human cells. Nat Cell Biol 14(3):266–75, Feb 26
Benedict WF, Weissman BE, Mark C, Stanbridge EJ (1984) Tumorigenicity of human HT1080 fibrosarcoma X normal fibroblast hybrids: chromosome dosage dependency. Cancer Res 44(8):3471–9, 1984 Aug
Biroccio A, Cherfils-Vicini J, Augereau A, Pinte S, Bauwens S, Ye J, Simonet T, Horard B, Jamet K, Cervera L, Mendez-Bermudez A, Poncet D, Grataroli R, de Rodenbeeke CT, Salvati E, Rizzo A, Zizza P, Ricoul M, Cognet C, Kuilman T, Duret H, Lépinasse F, Marvel J, Verhoeyen E, Cosset FL, Peeper D, Smyth MJ, Londoño-Vallejo A, Sabatier L, Picco V, Pages G, Scoazec JY, Stoppacciaro A, Leonetti C, Vivier E, Gilson E (2013) TRF2 inhibits a cell-extrinsic pathway through which natural killer cells eliminate cancer cells. Nat Cell Biol 15(7):818–28, 2013 Jul
Bourougaa K, Naski N, Boularan C, Mlynarczyk C, Candeias MM, Marullo S, Fåhraeus R (2010) Endoplasmic reticulum stress induces G2 cell-cycle arrest via mRNA translation of the p53 isoform p53/47. Mol Cell 38(1):78–88, Apr 9
Braig M, Lee S, Loddenkemper C, Rudolph C, Peters AH, Schlegelberger B, Stein H, Dorken B, Jenuwein T, Schmitt CA (2005) Oncogene-induced senescence as an initial barrier in lymphoma development. Nature 436(7051):660–665
Braumüller H, Wieder T, Brenner E, Aßmann S, Hahn M, Alkhaled M, Schilbach K, Essmann F, Kneilling M, Griessinger C, Ranta F, Ullrich S, Mocikat R, Braungart K, Mehra T, Fehrenbacher B, Berdel J, Niessner H, Meier F, van den Broek M, Häring HU, Handgretinger R, Quintanilla-Martinez L, Fend F, Pesic M, Bauer J, Zender L, Schaller M, Schulze-Osthoff K, Röcken M (2013) T-helper-1-cell cytokines drive cancer into senescence. Nature 494(7437):361–5, Feb 21
Buganim Y, Faddah AD, Jaenisch R (2013) Mechanisms and models of somatic cell reprogramming. Nat Rev Genet 14(6):427–439
Burton DG, Giles PJ, Sheerin AN, Smith SK, Lawton JJ, Ostler EL, Rhys-Williams W, Kipling D, Faragher RG (2009) Microarray analysis of senescent vascular smooth muscle cells: a link to atherosclerosis and vascular calcification. Exp Gerontol 44(10):659–665
Burton DG (2009) Cellular senescence, ageing and disease. Age (Dordr) 31(1):1–9, 2009 Mar
Burton DG, Krizhanovsky V (2014) Physiological and pathological consequences of cellular senescence. Cell Mol Life Sci 71(22):4373–86
Cerboni C, Fionda C, Soriani A, Zingoni A, Doria M, Cippitelli M, Santoni A (2014) The DNA damage response: a common pathway in the regulation of NKG2D and DNAM-1 ligand expression in normal, infected, and cancer cells. Front Immunol 7:4–508, 2014 Jan
Cerwenka A (2009) New twist on the regulation of NKG2D ligand expression. J Exp Med 206(2):265–268
Chen Z, Trotman LC, Shaffer D, Lin HK, Dotan ZA, Niki M, Koutcher JA, Scher HI, Ludwig T, Gerald W, Cordon-Cardo C, Pandolfi PP (2005) Crucial role of p53-dependent cellular senescence in suppression of Pten-deficient tumorigenesis. Nature 436(7051):725–730
Chen JH, Hales CN, Ozanne SE (2007) DNA damage, cellular senescence and organismal ageing: causal or correlative? Nucleic Acids Res 35(22):7417–28
Chien Y, Scuoppo C, Wang X, Fang X, Balgley B, Bolden JE, Premsrirut P, Luo W, Chicas A, Lee CS, Kogan SC, Lowe SW (2011) Control of the senescence-associated secretory phenotype by NF-κB promotes senescence and enhances chemosensitivity. Genes Dev 25(20):2125–36, Oct 15
Chuprin A, Gal H, Biron-Shental T, Biran A, Amiel A, Rozenblatt S, Krizhanovsky V (2013) Cell fusion induced by ERVWE1 or measles virus causes cellular senescence. Genes Dev 27(21):2356–66, Nov 1
Codogno P, Meijer AJ (2005) Autophagy and signaling: their role in cell survival and cell death. Cell Death Differ 12(Suppl 2):1509–18
Collado M, Gil J, Efeyan A, Guerra C, Schuhmacher AJ, Barradas M, Benguria A, Zaballos A, Flores JM, Barbacid M, Beach D, Serrano M (2005) Tumour biology: senescence in premalignant tumours. Nature 436(7051):642
Coppé JP, Patil CK, Rodier F, Sun Y, Muñoz DP, Goldstein J, Nelson PS, Desprez PY, Campisi J (2008) Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol 6(12):2853–68, Dec 2
Coppé JP, Patil CK, Rodier F, Krtolica A, Beauséjour CM, Parrinello S, Hodgson JG, Chin K, Desprez PY, Campisi J (2010) A human-like senescence-associated secretory phenotype is conserved in mouse cells dependent on physiological oxygen. PLoS One 5(2):e9188, Feb 12
Coppé JP, Rodier F, Patil CK, Freund A, Desprez PY, Campisi J (2011) Tumor suppressor and aging biomarker p16(INK4a) induces cellular senescence without the associated inflammatory secretory phenotype. J Biol Chem 286(42):36396–403, Oct 21
Crescenzi E, Pacifico F, Lavorgna A, De Palma R, D’Aiuto E, Palumbo G, Formisano S, Leonardi A (2011) NF-κB-dependent cytokine secretion controls Fas expression on chemotherapy-induced premature senescent tumor cells. Oncogene 30(24):2707–17, Jun 16
Cruickshanks HA, McBryan T, Nelson DM, Vanderkraats ND, Shah PP, van Tuyn J, Singh Rai T, Brock C, Donahue G, Dunican DS, Drotar ME, Meehan RR, Edwards JR, Berger SL, Adams PD (2013) Senescent cells harbour features of the cancer epigenome. Nat Cell Biol 15(12):1495–506
Davalos AR, Kawahara M, Malhotra GK, Schaum N, Huang J, Ved U, Beausejour CM, Coppe JP, Rodier F, Campisi J (2013) p53-dependent release of Alarmin HMGB1 is a central mediator of senescent phenotypes. J Cell Biol 201(4):613–29, May 13
Demaria M, Ohtani N, Youssef SA, Rodier F, Toussaint W, Mitchell JR, Laberge RM, Vijg J, van Steeg H, Dollé MET, Hoeijmakers JHJ, de Bruin A, Hara E, Campisi J (2014) An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev Cell, 2014; doi: 10.1016/j.devcel.2014.11.012
Dengjel J, Schoor O, Fischer R, Reich M, Kraus M, Müller M, Kreymborg K, Altenberend F, Brandenburg J, Kalbacher H, Brock R, Driessen C, Rammensee HG, Stevanovic S (2005) Autophagy promotes MHC class II presentation of peptides from intracellular source proteins. Proc Natl Acad Sci U S A 102(22):7922–7, May 31
Di Micco R, Fumagalli M, Cicalese A, Piccinin S, Gasparini P, Luise C, Schurra C, Garre M, Nuciforo PG, Bensimon A, Maestro R, Pelicci PG, d’ Adda di Fagagna F (2006) Oncogene-induced senescence is a DNA damage response triggered by DNA hyper-replication. Nature 444(7119):638–642
Dolezalova D, Mraz M, Barta T, Plevova K, Vinarsky V, Holubcova Z, Jaros J, Dvorak P, Pospisilova S, Hampl A (2012) MicroRNAs regulate p21(Waf1/Cip1) protein expression and the DNA damage response in human embryonic stem cells. Stem Cells 30(7):1362–72
Effros RB, Dagarag M, Spaulding C, Man J (2005) The role of CD8+ T-cell replicative senescence in human aging. Immunol Rev 205:147–57
Epstein CJ, Martin GM, Schultz AL, Motulsky AG (1966) Werner’s syndrome a review of its symptomatology, natural history, pathologic features, genetics and relationship to the natural aging process. Medicine (Baltimore) 45(3):177–221
Evans RJ, Wyllie FS, Wynford-Thomas D, Kipling D, Jones CJ (2003) A P53-dependent, telomere-independent proliferative life span barrier in human astrocytes consistent with the molecular genetics of glioma development. Cancer Res 63(16):4854–61, Aug 15
d’Adda di Fagagna F (2008) Living on a break: cellular senescence as a DNA-damage response. Nat Rev Cancer 8:512–522
Feliciano A, Sánchez-Sendra B, Kondoh H, Lleonart ME (2011) MicroRNAs regulate key effector pathways of senescence. J Aging Res 2011:205378
Fitzner B, Muller S, Walther M, Fischer M, Engelmann R, Muller-Hilke B, Putzer BM, Kreutzer M, Nizze H, Jaster R (2012) Senescence determines the fate of activated rat pancreatic stellate cells. J Cell Mol Med.
Freund A, Patil CK, Campisi J (2011) p38MAPK is a novel DNA damage response-independent regulator of the senescence-associated secretory phenotype. EMBO J 30(8):1536–48, 2011 Apr 20
Fumagalli M, Rossiello F, Mondello C, d’Adda di Fagagna F (2014) Stable cellular senescence is associated with persistent DDR activation. PLoS One 9(10):e110969, 2014 Oct 23
Garg AD, Kaczmarek A, Krysko O, Vandenabeele P, Krysko DV, Agostinis P (2012) ER stress-induced inflammation: does it aid or impede disease progression? Trends Mol Med 18(10):589–98
Gartel AL, Tyner AL (2002) The role of the cyclin-dependent kinase inhibitor p21 in apoptosis. Mol Cancer Ther 1(8):639–49
Gasser S, Orsulic S, Brown EJ, Raulet DH (2005) The DNA damage pathway regulates innate immune system ligands of the NKG2D receptor. Nature 436(7054):1186–90, Aug 25
Gilbert LA, Hemann MT (2010) DNA damage-mediated induction of a chemoresistant niche. Cell 143(3):355–66, Oct 29
Goldstein S (1969) Lifespan of cultured cells in progeria. Lancet 1(7591):424, 1969 Feb 22
Hampel B, Malisan F, Niederegger H, Testi R, Jansen-Dürr P (2004) Differential regulation of apoptotic cell death in senescent human cells. Exp Gerontol 39(11-12):1713–21, Nov-Dec
Hampel B, Wagner M, Teis D, Zwerschke W, Huber LA, Jansen-Dürr P (2005) Apoptosis resistance of senescent human fibroblasts is correlated with the absence of nuclear IGFBP-3. Aging Cell 4(6):325–30, Dec
Han Y, Randell E, Vasdev S, Gill V, Gadag V, Newhook LA, Grant M, Hagerty D (2007) Plasma methylglyoxal and glyoxal are elevated and related to early membrane alteration in young, complication-free patients with type 1 diabetes. Mol Cell Biochem 305(1-2):123–31, Nov
Han C, Jin L, Mei Y, Wu M (2013) Endoplasmic reticulum stress inhibits cell cycle progression via induction of p27 in melanoma cells. Cell Signal 25(1):144–9, Jan
Hawkins AJ, Golding SE, Khalil A, Valerie K (2011) DNA double-strand break—induced pro-survival signaling. Radiother Oncol 101(1):13–7, Oct
Hayflick L, Moorhead PS (1961) The serial cultivation of human diploid cell strains. Exp Cell Res 25:585–621
Hayflick L (1965) The limited in vitro lifetime of human diploid cell strains. Exp Cell Res 37:614–636
Henson SM, Lanna A, Riddell NE, Franzese O, Macaulay R, Griffiths SJ, Puleston DJ, Watson AS, Simon AK, Tooze SA, Akbar AN (2014) p38 signaling inhibits mTORC1-independent autophagy in senescent human CD8+ T cells. J Clin Invest 124(9):4004–16, 2014 Sep 2
Herbig U, Jobling WA, Chen BP, Chen DJ, Sedivy JM (2004) Telomere shortening triggers senescence of human cells through a pathway involving ATM, p53, and p21(CIP1), but not p16(INK4a). Mol Cell 14(4):501–13, May 21
Herbig U, Ferreira M, Condel L, Carey D, Sedivy JM (2006) Cellular senescence in aging primates. Science 311(5765):1257
Hewitt G, Jurk D, Marques FD, Correia-Melo C, Hardy T, Gackowska A, Anderson R, Taschuk M, Mann J, Passos JF (2012) Telomeres are favoured targets of a persistent DNA damage response in ageing and stress-induced senescence. Nat Commun 3:708, Feb 28
Hoffmann J, Haendeler J, Aicher A, Rössig L, Vasa M, Zeiher AM, Dimmeler S (2001) Aging enhances the sensitivity of endothelial cells toward apoptotic stimuli: important role of nitric oxide. Circ Res 89(8):709–15, Oct 12
House NC, Koch MR, Freudenreich CH (2014) Chromatin modifications and DNA repair: beyond double-strand breaks. Front Genet 5:296, Sep 5
Hubackova S, Krejcikova K, Bartek J, Hodny Z (2012) IL1- and TGFβ-Nox4 signaling, oxidative stress and DNA damage response are shared features of replicative, oncogene-induced, and drug-induced paracrine 'bystander senescence'. Aging (Albany NY) 4(12):932–51, 2012 Dec
Itahana K, Campisi J, Dimri GP (2004) Mechanisms of cellular senescence in human and mouse cells. Biogerontology 5(1):1–10
Janssens S, Tschopp J (2006) Signals from within: the DNA-damage-induced NF-kappaB response. Cell Death Differ 13(5):773–84, May
Jeon H, Boo YC (2013) Senescent endothelial cells are prone to TNF-α-induced cell death due to expression of FAS receptor. Biochem Biophys Res Commun 438(2):277–82, Aug 23
Jun JI, Lau LF (2010) The matricellular protein CCN1 induces fibroblast senescence and restricts fibrosis in cutaneous wound healing. Nat Cell Biol 12(7):676–685
Jurk D, Wang C, Miwa S, Maddick M, Korolchuk V, Tsolou A, Gonos ES, Thrasivoulou C, Saffrey MJ, Cameron K, von Zglinicki T (2012) Postmitotic neurons develop a p21-dependent senescence-like phenotype driven by a DNA damage response. Aging Cell 11(6):996–1004, Dec
Kang TW, Yevsa T, Woller N, Hoenicke L, Wuestefeld T, Dauch D, Hohmeyer A, Gereke M, Rudalska R, Potapova A, Iken M, Vucur M, Weiss S, Heikenwalder M, Khan S, Gil J, Bruder D, Manns M, Schirmacher P, Tacke F, Ott M, Luedde T, Longerich T, Kubicka S, Zender L (2011) Senescence surveillance of pre-malignant hepatocytes limits liver cancer development. Nature 479(7374):547–51, Nov 9
Kalashnik L, Bridgeman CJ, King AR, Francis SE, Mikhalovsky S, Wallis C, Denyer SP, Crossman D, Faragher RG (2000) A cell kinetic analysis of human umbilical vein endothelial cells. Mech Ageing Dev 120(1-3):23–32, Dec 1
Karakasilioti I, Kamileri I, Chatzinikolaou G, Kosteas T, Vergadi E, Robinson AR, Tsamardinos I, Rozgaja TA, Siakouli S, Tsatsanis C, Niedernhofer LJ, Garinis GA (2013) DNA damage triggers a chronic autoinflammatory response, leading to fat depletion in NER progeria. Cell Metab 18(3):403–15, Sep 3
Khalil A, Morgan RN, Adams BR, Golding SE, Dever SM, Rosenberg E, Povirk LF, Valerie K (2011) ATM-dependent ERK signaling via AKT in response to DNA double-strand breaks. Cell Cycle 10(3):481–91, 2011 Feb 1
Kim TW, Kim HJ, Lee C, Kim HY, Baek SH, Kim JH, Kwon KS, Kim JR (2008) Identification of replicative senescence-associated genes in human umbilical vein endothelial cells by an annealing control primer system. Exp Gerontol 43(4):286–95, Apr
Kim KH, Chen CC, Monzon RI, Lau LF (2013) Matricellular protein CCN1 promotes regression of liver fibrosis through induction of cellular senescence in hepatic myofibroblasts. Mol Cell Biol 33(10):2078–2090
Kipling D, Faragher RG (1997) Progeroid syndromes: probing the molecular basis of aging? Mol Pathol 50(5):234–41, 1997 Oct
Kipling D, Jones DL, Smith SK, Giles PJ, Jennert-Burston K, Ibrahim B, Sheerin AN, Evans AJ, Rhys-Willams W, Faragher RG (2009) A transcriptomic analysis of the EK1.Br strain of human fibroblastoid keratocytes: the effects of growth, quiescence and senescence. Exp Eye Res 88(2):277–85, 2009 Feb
Kiyono T, Foster SA, Koop JI, McDougall JK, Galloway DA, Klingelhutz AJ (1998) Both Rb/p16INK4a inactivation and telomerase activity are required to immortalize human epithelial cells. Nature 396(6706):84–8, Nov 5
Korotchkina LG, Demidenko ZN, Gudkov AV, Blagosklonny MV (2009) Cellular quiescence caused by the Mdm2 inhibitor nutlin-3A. Cell Cycle 8(22):3777–81, Nov 15
Kreis NN, Louwen F, Yuan J (2014) Less understood issues: p21Cip1 in mitosis and its therapeutic potential. Oncogene. May 26;0
Krizhanovsky V, Yon M, Dickins RA, Hearn S, Simon J, Miething C, Yee H, Zender L, Lowe SW (2008) Senescence of activated stellate cells limits liver fibrosis. Cell 134(4):657–67, Aug 22
Kuilman T, Michaloglou C, Vredeveld LC, Douma S, van Doorn R, Desmet CJ, Aarden LA, Mooi WJ, Peeper DS (2008) Oncogene-induced senescence relayed by an interleukin dependent inflammatory network. Cell 133(6):1019–1031
Lackner DH, Hayashi MT, Cesare AJ, Karlseder J (2014) A genomics approach identifies senescence-specific gene expression regulation. Aging Cell 13(5):946–50, Oct
Lanna A, Henson SM1, Escors D2, Akbar AN (2014) The kinase p38 activated by the metabolic regulator AMPK and scaffold TAB1 drives the senescence of human T cells. Nat Immunol 15(10):965–72, Oct
Larsen SA, Kassem M, Rattan SI (2012) Glucose metabolite glyoxal induces senescence in telomerase-immortalized human mesenchymal stem cells. Chem Cent J 6(1):18, Mar 17
Lin D, Lavender H, Soilleux EJ, O’Callaghan CA (2012) NF-κB regulates MICA gene transcription in endothelial cell through a genetically inhibitable control site. J Biol Chem 287(6):4299–310, Feb 3
Lindqvist LM, Vaux DL (2014) BCL2 and related prosurvival proteins require BAK1 and BAX to affect autophagy. Autophagy 10(8):1474–5, Aug
Liu FJ, Wen T, Liu L (2012) MicroRNAs as a novel cellular senescence regulator. Ageing Res Rev 11(1):41–50, Jan
Liu J, Huang K, Cai GY, Chen XM, Yang JR, Lin LR, Yang J, Huo BG, Zhan J, He YN (2014) Receptor for advanced glycation end-products promotes premature senescence of proximal tubular epithelial cells via activation of endoplasmic reticulum stress-dependent p21 signaling. Cell Signal 26(1):110–21, Jan
López-Soto A, Huergo-Zapico L, Acebes-Huerta A, Villa-Alvarez M, Gonzalez S (2014) NKG2D signaling in cancer immunosurveillance. Int J Cancer. doi:10.1002/ijc.28775, Feb 8
Lotem J, Sachs L (1999) Cytokines as suppressors of apoptosis. Apoptosis 4(3):187–96, Jun
Luo X, Suzuki M, Ghandhi SA, Amundson SA, Boothman DA (2014) ATM regulates insulin-like growth factor 1-secretory clusterin (IGF-1-sCLU) expression that protects cells against senescence. PLoS One 9(6):e99983, Jun 17
Lujambio A, Akkari L, Simon J, Grace D, Tschaharganeh DF, Bolden JE, Zhao Z, Thapar V, Joyce JA, Krizhanovsky V, Lowe SW (2013) Non-cell-autonomous tumor suppression by p53. Cell 153(2):449–60, Apr 11
Ma K, Qiu L, Mrasek K, Zhang J, Liehr T, Quintana LG, Li Z (2012) Common fragile sites: genomic hotspots of DNA damage and carcinogenesis. Int J Mol Sci 13(9):11974–99
Macieira-Coelho A, Ponten J, Philipson L (1966) The division cycle and RNA-synthesis in diploid human cells at different passage levels in vitro. Exp Cell Res 42:673–684
Marcotte R, Lacelle C, Wang E (2004) Senescent fibroblasts resist apoptosis by downregulating caspase-3. Mech Ageing Dev 125(10-11):777–83, 2004 Oct-Nov
Martin GM, Sprague CA, Epstein CJ (1970) Replicative life-span of cultivated human cells. Effects of donor’s age, tissue, and genotype. Lab Invest 23:86–92
Maya-Mendoza A, Ostrakova J, Kosar M, Hall A, Duskova P, Mistrik M, Merchut-Maya JM, Hodny Z, Bartkova J, Christensen C, Bartek J. (2014) Myc and Ras oncogenes engage different energy metabolism programs and evoke distinct patterns of oxidative and DNA replication stress. Mol Oncol. Nov 15
Mayorga M, Bahi N, Ballester M, Comella JX, Sanchis D (2004) Bcl-2 is a key factor for cardiac fibroblast resistance to programmed cell death. J Biol Chem 279(33):34882–9, Aug 13
Michaloglou C, Vredeveld LC, Soengas MS, Denoyelle C, Kuilman T, van der Horst CM, Majoor DM, Shay JW, Mooi WJ, Peeper DS (2005) BRAFE600-associated senescence-like cell cycle arrest of human naevi. Nature 436(7051):720–724
Minagawa S, Araya J, Numata T, Nojiri S, Hara H, Yumino Y, Kawaishi M, Odaka M, Morikawa T, Nishimura SL, Nakayama K, Kuwano K (2010) Accelerated epithelial cell senescence in IPF and the inhibitory role of SIRT6 in TGF-β-induced senescence of human bronchial epithelial cells. Am J Physiol Lung Cell Mol Physiol 300(3):L391–401, Mar
Munoz-Espin D, Canamero M, Maraver A, Gomez-Lopez G, Contreras J, Murillo-Cuesta S, Rodriguez-Baeza A, Varela-Nieto I, Ruberte J, Collado M, Serrano M (2013) Programmed cell senescence during mammalian embryonic development. Cell 155(5):1104–1118
Norsgaard H, Clark BF, Rattan SI (1996) Distinction between differentiation and senescence and the absence of increased apoptosis in human keratinocytes undergoing cellular aging in vitro. Exp Gerontol 31(5):563–70, Sep-Oct
Ovadya Y, Krizhanovsky V (2014) Senescent cells: SASPected drivers of age-related pathologies. Biogerontology. 2014 Sep 13
Overhoff MG, Garbe JC, Koh J, Stampfer MR, Beach DH, Bishop CL (2013) Cellular senescence mediated by p16INK4A-coupled miRNA pathways. Nucleic Acids Res 42(3):1606–18, Feb
Passos JF, Nelson G, Wang C, Richter T, Simillion C, Proctor CJ, Miwa S, Olijslagers S, Hallinan J, Wipat A, Saretzki G, Rudolph KL, Kirkwood TB, von Zglinicki T (2010) Feedback between p21 and reactive oxygen production is necessary for cell senescence. Mol Syst Biol 6:347, 2010
Pálmai-Pallag T, Bachrati CZ (2014) Inflammation-induced DNA damage and damage-induced inflammation: a vicious cycle. Microbes Infect 16(10):822–832, Oct 22
Park J, Jo YH, Cho CH, Choe W, Kang I, Baik HH, Yoon KS (2013) ATM-deficient human fibroblast cells are resistant to low levels of DNA double-strand break induced apoptosis and subsequently undergo drug-induced premature senescence. Biochem Biophys Res Commun 430(1):429–35, 2013 Jan 4
Perucca P, Cazzalini O, Madine M, Savio M, Laskey RA, Vannini V, Prosperi E, Stivala LA (2009) Loss of p21 CDKN1A impairs entry to quiescence and activates a DNA damage response in normal fibroblasts induced to quiescence. Cell Cycle 8(1):105–14, Jan 1
Piccolo MT, Crispi S (2012) The dual role played by p21 may influence the apoptotic or anti-apoptotic fate in cancer. J Can Res Updates 1(189–202):189
Price BD, D’Andrea AD (2013) Chromatin remodeling at DNA double-strand breaks. Cell 152(6):1344–54, Mar 14
Raciti M, Lotti LV, Valia S, Pulcinelli FM, Di Renzo L (2012) JNK2 is activated during ER stress and promotes cell survival. Cell Death Dis 3:e429, Nov 22
Rajagopalan S, Long EO (2012) Cellular senescence induced by CD158d reprograms natural killer cells to promote vascular remodeling. Proc Natl Acad Sci U S A 109(50):20596–601, Dec 11
Rakhra K, Bachireddy P, Zabuawala T, Zeiser R, Xu L, Kopelman A, Fan AC, Yang Q, Braunstein L, Crosby E, Ryeom S, Felsher DW (2010) CD4(+) T cells contribute to the remodeling of the microenvironment required for sustained tumor regression upon oncogene inactivation. Cancer Cell 18(5):485–98, Nov 16
Reimann M, Lee S, Loddenkemper C, Dörr JR, Tabor V, Aichele P, Stein H, Dörken B, Jenuwein T, Schmitt CA (2010) Tumor stroma-derived TGF-beta limits myc-driven lymphomagenesis via Suv39h1-dependent senescence. Cancer Cell 17(3):262–72, Mar 16
Reuschenbach M, von Knebel Doeberitz M, Wentzensen N (2009) A systematic review of humoral immune responses against tumor antigens. Cancer Immunol Immunother 58(10):1535–44, Oct
Rodier F, Coppe JP, Patil CK, Hoeijmakers WA, Munoz DP, Raza SR, Freund A, Campeau E, Davalos AR, Campisi J (2009) Persistent DNA damage signalling triggers senescence-associated inflammatory cytokine secretion. Nat Cell Biol 11(8):973–979
Robles SJ, Adami GR (1998) Agents that cause DNA double strand breaks lead to p16INK4a enrichment and the premature senescence of normal fibroblasts. Oncogene 16(9):1113–1123
Rodriguez-Rocha H, Garcia-Garcia A, Panayiotidis MI, Franco R (2011) DNA damage and autophagy. Mutat Res 711(1-2):158–66, Jun 3
Rose J, Söder S, Skhirtladze C, Schmitz N, Gebhard PM, Sesselmann S, Aigner T (2012) DNA damage, discoordinated gene expression and cellular senescence in osteoarthritic chondrocytes. Osteoarthr Cartil 20(9):1020–8, Sep
Ryu SJ, Oh YS, Park SC (2007) Failure of stress-induced downregulation of Bcl-2 contributes to apoptosis resistance in senescent human diploid fibroblasts. Cell Death Differ 14(5):1020–8, 2007 May
Sagiv A, Biran A, Yon M, Simon J, Lowe SW, Krizhanovsky V (2013) Granule exocytosis mediates immune surveillance of senescent cells. Oncogene 32(15):1971–7, Apr 11
Sagiv A, Krizhanovsky V (2013) Immunosurveillance of senescent cells: the bright side of the senescence program. Biogerontology 14(6):617–28, Dec
Scherz-Shouval R, Elazar Z (2011) Regulation of autophagy by ROS: physiology and pathology. Trends Biochem Sci 36(1):30–8, Jan
Schröder M (2008) Endoplasmic reticulum stress responses. Cell Mol Life Sci 65(6):862–94, Mar
Seluanov A, Gorbunova V, Falcovitz A, Sigal A, Milyavsky M, Zurer I, Shohat G, Goldfinger N, Rotter V (2001) Change of the death pathway in senescent human fibroblasts in response to DNA damage is caused by an inability to stabilize p53. Mol Cell Biol 21(5):1552–1564
Senturk S, Mumcuoglu M, Gursoy-Yuzugullu O, Cingoz B, Akcali KC, Ozturk M (2010) Transforming growth factor-beta induces senescence in hepatocellular carcinoma cells and inhibits tumor growth. Hepatology 52(3):966–74, Sep
Sejersen, Rattan (2009) Dicarbonyl-induced accelerated aging in vitro in human skin fibroblasts. Biogerontology 10(2):203–11
Serrano M, Lin AW, McCurrach ME, Beach D, Lowe SW (1997) Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell 88(5):593–602
Severino J, Allen RG, Balin S, Balin A, Cristofalo VJ (2000) Is beta-galactosidase staining a marker of senescence in vitro and in vivo? Exp Cell Res 257(1):162–71, 2000 May 25
Shangari O’B (2004) The cytotoxic mechanism of glyoxal involves oxidative stress. Biochem Pharmacol 68(7):1433–1442, 1 October
Shelton DN, Chang E, Whittier PS, Choi D, Funk WD (1999) Microarray analysis of replicative senescence. Curr Biol 9:939–945
Singh K, Matsuyama S, Drazba JA, Almasan A (2012) Autophagy-dependent senescence in response to DNA damage and chronic apoptotic stress. Autophagy 8(2):236–51, Feb 1
Smith SK, Kipling D (2004) The role of replicative senescence in cancer and human ageing: utility (or otherwise) of murine models. Cytogenet Genome Res 105(2–4):455–63
Smith J, Tho LM, Xu N, Gillespie DA (2010) The ATM-Chk2 and ATR-Chk1 pathways in DNA damage signaling and cancer. Adv Cancer Res 108:73–112
Sousa-Victor P, Gutarra S, Garcia-Prat L, Rodriguez-Ubreva J, Ortet L, Ruiz-Bonilla V, Jardi M, Ballestar E, Gonzalez S, Serrano AL, Perdiguero E, Munoz-Canoves P (2014) Geriatric muscle stem cells switch reversible quiescence into senescence. Nature 506(7488):316–321
Soriani A, Zingoni A, Cerboni C, Iannitto ML, Ricciardi MR, Di Gialleonardo V, Cippitelli M, Fionda C, Petrucci MT, Guarini A, Foà R, Santoni A (2009) ATM-ATR-dependent up-regulation of DNAM-1 and NKG2D ligands on multiple myeloma cells by therapeutic agents results in enhanced NK-cell susceptibility and is associated with a senescent phenotype. Blood 113(15):3503–11, Apr 9
Stern-Ginossar N, Gur C, Biton M, Horwitz E, Elboim M, Stanietsky N, Mandelboim M, Mandelboim O (2008) Human microRNAs regulate stress-induced immune responses mediated by the receptor NKG2D. Nat Immunol 9(9):1065–73, Sep
Storer M, Mas A, Robert-Moreno A, Pecoraro M, Ortells MC, Di Giacomo V, Yosef R, Pilpel N, Krizhanovsky V, Sharpe J, Keyes WM (2013) Senescence is a developmental mechanism that contributes to embryonic growth and patterning. Cell 155(5):1119–1130
Sun X, Mao Y, Wang J, Zu L, Hao M, Cheng G, Qu Q, Cui D, Keller ET, Chen X, Shen K, Wang J (2014) IL-6 secreted by cancer-associated fibroblasts induces tamoxifen resistance in luminal breast cancer. Oncogene Jun 9; 0
Suram A, Kaplunov J, Patel PL, Ruan H, Cerutti A, Boccardi V, Fumagalli M, Di Micco R, Mirani N, Gurung RL, Hande MP, d’Adda di Fagagna F, Herbig U (2012) Oncogene-induced telomere dysfunction enforces cellular senescence in human cancer precursor lesions. EMBO J 31(13):2839–2851
Takacova S, Slany R, Bartkova J, Stranecky V, Dolezel P, Luzna P, Bartek J, Divoky V (2012) DNA damage response and inflammatory signaling limit the MLL-ENL-induced leukemogenesis in vivo. Cancer Cell 21(4):517–31, Apr 17
Tang ML, Khan MK, Croxford JL, Tan KW, Angeli V, Gasser S (2014) The DNA damage response induces antigen presenting cell-like functions in fibroblasts. Eur J Immunol 44(4):1108–18
Tepper CG, Seldin MF, Mudryj M (2000) Fas-mediated apoptosis of proliferating, transiently growth-arrested, and senescent normal human fibroblasts. Exp Cell Res 260(1):9–19, Oct 10
Thornalley PJ (2008) Protein and nucleotide damage by glyoxal and methylglyoxal in physiological systems–role in ageing and disease. Drug Metabol Drug Interact 23(1–2):125–50
Tokarsky-Amiel R, Azazmeh N, Helman A, Stein Y, Hassan A, Maly A, Ben-Porath I (2013) Dynamics of senescent cell formation and retention revealed by p14ARF induction in the epidermis. Cancer Res 73(9):2829–39, May 1
Toledo LI, Murga M, Gutierrez-Martinez P, Soria R, Fernandez-Capetillo O (2008) ATR signaling can drive cells into senescence in the absence of DNA breaks. Genes Dev 22(3):297–302, Feb 1
Toso A, Revandkar A, Di Mitri D, Guccini I, Proietti M, Sarti M, Pinton S, Zhang J, Kalathur M, Civenni G, Jarrossay D, Montani E, Marini C, Garcia-Escudero R, Scanziani E, Grassi F, Pandolfi PP, Catapano CV, Alimonti A (2014) Enhancing chemotherapy efficacy in Pten-deficient prostate tumors by activating the senescence-associated antitumor immunity. Cell Rep 9(1):75–89, Oct 9
Toussaint O, Medrano EE, von Zglinicki T (2000) Cellular and molecular mechanisms of stress-induced premature senescence (SIPS) of human diploid fibroblasts and melanocytes. Exp Gerontol 35(8):927–945
Untergasser G, Gander R, Rumpold H, Heinrich E, Plas E, Berger P (2003) TGF-beta cytokines increase senescence-associated beta-galactosidase activity in human prostate basal cells by supporting differentiation processes, but not cellular senescence. Exp Gerontol 38(10):1179–88, Oct
Vasey DB, Wolf CR, Brown K, Whitelaw CB (2011) Spatial p21 expression profile in the mid-term mouse embryo. Transgenic Res 20(1):23–8, Feb
Valés-Gómez M, Chisholm SE, Cassady-Cain RL, Roda-Navarro P, Reyburn HT (2008) Selective induction of expression of a ligand for the NKG2D receptor by proteasome inhibitors. Cancer Res 68(5):1546–54, Mar 1
Vermeij WP, Hoeijmakers JH, Pothof J (2014) Aging: not all DNA damage is equal. Curr Opin Genet Dev 26C:124–130, Jun
Vitiello PF, Wu YC, Staversky RJ, O’Reilly MA (2009) p21(Cip1) protects against oxidative stress by suppressing ER-dependent activation of mitochondrial death pathways. Free Radic Biol Med 46(1):33–41, Jan 1
Wang C, Jurk D, Maddick M, Nelson G, Martin-Ruiz C, von Zglinicki T (2009) DNA damage response and cellular senescence in tissues of aging mice. Aging Cell 8(3):311–323
Wang X, Brégégère F, Soroka Y, Kayat A, Redziniak G, Milner Y (2004) Enhancement of Fas-mediated apoptosis in ageing human keratinocytes. Mech Ageing Dev 125(3):237–49, 2004 Mar
Wang E (1995) Senescent human fibroblasts resist programmed cell death, and failure to suppress bcl2 is involved. Cancer Res 55(11):2284–2292
Wang Y, Taniguchi T (2013) MicroRNAs and DNA damage response: implications for cancer therapy. Cell Cycle 12(1):32–42, Jan 1
West MD, Pereira-Smith OM, Smith JR (1989) Replicative senescence of human skin fibroblasts correlates with a loss of regulation and overexpression of collagenase activity. Exp Cell Res 184:138–147
Weyemi U, Lagente-Chevallier O, Boufraqech M, Prenois F, Courtin F, Caillou B, Talbot M, Dardalhon M, Al Ghuzlan A, Bidart JM, Schlumberger M, Dupuy C (2012) ROS-generating NADPH oxidase NOX4 is a critical mediator in oncogenic H-Ras-induced DNA damage and subsequent senescence. Oncogene 31(9):1117–29, 2012 Mar 1
Wu J, Niu J, Li X, Wang X, Guo Z, Zhang F (2014) TGF-β1 induces senescence of bone marrow mesenchymal stem cells via increase of mitochondrial ROS production. BMC Dev Biol 14:21, May 18
Xu HD, Wu D, Gu JH, Ge JB, Wu JC, Han R, Liang ZQ, Qin ZH (2013) The pro-survival role of autophagy depends on Bcl-2 under nutrition stress conditions. PLoS One 8(5):e63232, May 3
Xue W, Zender L, Miething C, Dickins RA, Hernando E, Krizhanovsky V, Cordon-Cardo C, Lowe SW (2007) Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature 445(7128):656–660
Yu AL, Birke K, Moriniere J, Welge-Lüssen U (2010) TGF-{beta}2 induces senescence-associated changes in human trabecular meshwork cells. Invest Ophthalmol Vis Sci 51(11):5718–23, Nov
Zhang H, Pan KH, Cohen SN (2003) Senescence-specific gene expression fingerprints reveal cell-type-dependent physical clustering of up-regulated chromosomal loci. Proc Natl Acad Sci U S A 100(6):3251–3256
Zhang K, Chen C, Liu Y, Chen H, Liu JP (2014) Cellular senescence occurred widespread to multiple selective sites in the fetal tissues and organs of mice. Clin Exp Pharmacol Physiol 41(12):965–75, Dec
Zhou F, Yang Y, Xing D (2011) Bcl-2 and Bcl-xL play important roles in the crosstalk between autophagy and apoptosis. FEBS J 278(3):403–13, Feb
Zhu J, Woods D, McMahon M, Bishop JM (1998) Senescence of human fibroblasts induced by oncogenic Raf. Genes Dev 12(19):2997–3007
Acknowledgments
We wish to thank Adi Sagiv and Anat Biran for reading our manuscript and for helpful suggestions. RGAF is funded by the Glenn Foundation for Medical Research.
Author information
Authors and Affiliations
Corresponding authors
About this article
Cite this article
Burton, D.G.A., Faragher, R.G.A. Cellular senescence: from growth arrest to immunogenic conversion. AGE 37, 27 (2015). https://doi.org/10.1007/s11357-015-9764-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11357-015-9764-2